| MEDICATION GUIDE
Buprenorphine and Naloxone (bue” pre nor’ feen) and (nal ox’ one) Sublingual Film, CIII |
||
|
IMPORTANT: Keep buprenorphine and naloxone sublingual film in a secure place away from children. Accidental use by a child is a medical emergency and can result in death. If a child accidentally takes buprenorphine and naloxone sublingual film, get emergency help or call 911 right away. Tell your healthcare provider if you are living in a household where there are small children. |
||
What is the most important information I should know about buprenorphine and naloxone sublingual film?
|
||
What is buprenorphine and naloxone sublingual film?
|
||
| Who should not take buprenorphine and naloxone sublingual film?
Do not take buprenorphine and naloxone sublingual film if you are allergic to buprenorphine or naloxone. |
||
Before taking buprenorphine and naloxone, tell your healthcare provider about all your medical conditions, including if you have
|
||
Tell your healthcare provider if you are:
Tell your healthcare provider about all the medicines you take, including prescription and over‐the‐counter medicines, vitamins, or herbal supplements. |
||
| How should I take buprenorphine and naloxone sublingual film?
Read the Instructions for Use at the end of this Medication Guide for detailed instructions on how to take buprenorphine and naloxone.
|
||
What should I avoid while taking buprenorphine and naloxone sublingual film?
|
||
| What are the possible side effects of buprenorphine and naloxone sublingual film?
Buprenorphine and naloxone sublingual film can cause serious side effects, including:
These are not all the possible side effects of buprenorphine and naloxone. |
||
| General information about the safe and effective use of buprenorphine and naloxone sublingual film.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not take buprenorphine and naloxone sublingual film for a condition for which it was not prescribed. Do not give buprenorphine and naloxone sublingual film to other people, even if they have the same symptoms you have. It may harm them and it is against the law. You can ask your doctor or pharmacist for information that is written for healthcare professionals. |
||
| This Medication Guide has been approved by the U.S. Food and Drug Administration. To reorder additional Medication Guides, please contact Dr. Reddy's Customer Service at 1-866-733-3952. |
||
|
Rx only Manufactured By: Lohmann Therapy Systems, (LTS) West Caldwell, NJ 07006 Manufactured For: Dr. Reddy’s Laboratories Inc., Princeton, NJ 08540, USA Revised: 01/2024 |
||
INSTRUCTIONS FOR USE
Buprenorphine and Naloxone
(bue” pre nor’ feen) and (nal ox’ one)
Sublingual Film, CIII
This “Instructions for Use” contains information on how to correctly take buprenorphine and naloxone sublingual film.
Important Information You Need to Know Before Taking Buprenorphine and Naloxone Sublingual Film:
- Your healthcare provider should show you how to take buprenorphine and naloxone sublingual film the right way.
- Each buprenorphine and naloxone sublingual film comes in a sealed child‐resistant foil pouch. Do not open the foil pouch until you are ready to take it.
Preparing to take buprenorphine and naloxone sublingual film:
- To open your buprenorphine and naloxone sublingual film foil pouch, fold along the dotted line (see Figure1)

Figure 1
- Tear down at slit or cut with scissors along the arrow (see Figure 2)

Figure 2
-
- Before taking buprenorphine and naloxone sublingual film, drink water to moisten your mouth. This helps the film dissolve more easily.
To take buprenorphine and naloxone sublingual film under your tongue (sublingual administration):
- Hold the film between two fingers by the outside edges.
- Place the buprenorphine and naloxone sublingual film under your tongue, close to the base either to the left or right of the center (see Figure 3).
-
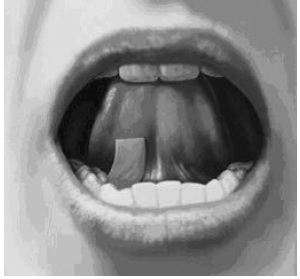
-
Figure 3
- If your healthcare provider tells you to take 2 films at a time, place the second film under your tongue on the opposite side. Avoid letting the films touch.
- Keep the films in place until they have completely dissolved.
- If your healthcare provider tells you to take a third film, place it under your tongue on either side after the first 2 films have dissolved.
To take buprenorphine and naloxone sublingual film on the inside of your cheek(buccal administration): - Hold the film between two fingers by the outside edges.
- Place one film on the inside of your right or left cheek (see Figure 4).
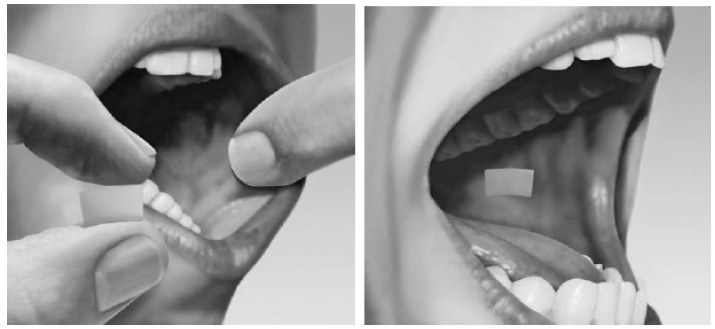
Figure 4
- If your healthcare provider tells you to take 2 films at a time, place the other film on the inside of the opposite cheek.
- Keep the films in place until they have completely dissolved.
- If your healthcare provider tells you to take a third film, place it on the inside of your right or left cheek after the first 2 films have dissolved.
- While buprenorphine and naloxone sublingual film is dissolving, do not chew or swallow the film because the medicine will not work as well.
- Talking while the film is dissolving can affect how well the medicine in buprenorphine and naloxone sublingual film is absorbed.
- After buprenorphine and naloxone sublingual film is completely dissolved, rinse your mouth with water and swallow. Wait for at least one hour before brushing teeth.
- If you miss a dose of buprenorphine and naloxone sublingual film, take your medicine when you remember. If it is almost time for your next dose, skip the missed dose and take the next dose at your regular time. Do not take 2 doses at the same time unless your healthcare provider tells you to. If you are not sure about your dosing, call your healthcare provider.
- Do not stop taking buprenorphine and naloxone sublingual film suddenly. You could become sick and have withdrawal symptoms because your body has become used to the medicine. Physical dependence is not the same as drug addiction. Your healthcare provider can tell you more about the differences between physical dependence and drug addiction. To have fewer withdrawal symptoms, ask your healthcare provider how to stop using buprenorphine and naloxone sublingual film the right way.
If you take too much buprenorphine and naloxone sublingual film or overdose, call Poison Control or get emergency medical help right away.
Storing buprenorphine and naloxone Sublingual Film: - Store buprenorphine and naloxone sublingual film at room temperature between 68°F to 77°F (20°C to 25°C).
- Keep buprenorphine and naloxone sublingual film in a safe place, out of the sight and reach of children.
Disposing of Buprenorphine and Naloxone Sublingual Film: - Dispose of unused buprenorphine and naloxone sublingual film as soon as you no longer need them.
- Dispose of expired, unwanted or unused buprenorphine and naloxone sublingual film by removing the buprenorphine and naloxone sublingual film from the foil packaging, and promptly flushing down the toilet (if a drug take‐back option is not readily available). Do not flush the buprenorphine and naloxone sublingual film foil pouch down the toilet. Visit www.fda.gov/drugdisposal for additional information on disposal of unused medicines.
If you need help with disposal of buprenorphine and naloxone sublingual film, call 1-888-375-3784.
Rx only
Manufactured By:
Lohmann Therapy Systems, (LTS)
West Caldwell, NJ 07006
Manufactured For:
Dr. Reddy’s Laboratories Inc.,
Princeton, NJ 08540, USA
This “Instructions for Use” has been approved by the U.S. Food and Drug Administration.
Revised: 01/2024