ORPHENADRINE CITRATE- orphenadrine citrate tablet, extended release
Bryant Ranch Prepack
----------
Orphenadrine Citrate Extended-Release Tablets, USP
DESCRIPTION
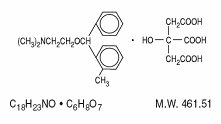
Orphenadrine citrate, USP is the citrate salt of orphenadrine (2-dimethyl-aminoethyl 2-methylbenzhydryl ether citrate). It occurs as a white, crystalline powder having a bitter taste. It is practically odorless; sparingly soluble in water, slightly soluble in alcohol and has a molecular weight of 461.51. The molecular formula C18H23NO • C6H8O7 is represented by the following structural formula:
Each orphenadrine citrate extended-release tablet, USP contains 100 mg orphenadrine citrate, USP. Orphenadrine citrate extended-release tablets, USP also contain: calcium stearate, ethylcellulose and lactose monohydrate.
CLINICAL PHARMACOLOGY
The mode of therapeutic action has not been clearly identified, but may be related to its analgesic properties. Orphenadrine citrate does not directly relax tense skeletal muscles in man. Orphenadrine citrate also possesses anti-cholinergic actions.
INDICATIONS AND USAGE
Orphenadrine citrate extended-release tablets are indicated as an adjunct to rest, physical therapy and other measures for the relief of discomfort associated with acute painful musculo skeletal conditions.
CONTRAINDICATIONS
Contraindicated in patients with glaucoma, pyloric or duodenal obstruction, stenosing peptic ulcers, prostatic hypertrophy or obstruction of the bladder neck, cardio-spasm (megaesophagus) and myasthenia gravis.
Contraindicated in patients who have demonstrated a previous hypersensitivity to the drug.
WARNINGS
Some patients may experience transient episodes of light-headedness, dizziness or syncope. Orphenadrine citrate may impair the ability of the patient to engage in potentially hazardous activities such as operating machinery or driving a motor vehicle; ambulatory patients should therefore be cautioned accordingly.
PRECAUTIONS
Confusion, anxiety and tremors have been reported in few patients receiving propoxyphene and orphenadrine concomitantly. As these symptoms may be simply due to an additive effect, reduction of dosage and/or discontinuation of one or both agents is recommended in such cases.
Orphenadrine citrate should be used with caution in patients with tachycardia, cardiac decompensation, coronary insufficiency, cardiac arrhythmias.
Safety of continuous long-term therapy with orphenadrine citrate has not been established. Therefore, if orphenadrine citrate is prescribed for prolonged use, periodic monitoring of blood, urine and liver function values is recommended.
PREGNANCY
Animal reproduction studies have not been conducted with orphenadrine citrate. It is also not known whether orphenadrine citrate can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Orphenadrine citrate should be given to a pregnant woman only if clearly needed.
ADVERSE REACTIONS
Adverse reactions of orphenadrine citrate are mainly due to the mild anti-cholinergic action of orphenadrine citrate and are usually associated with higher dosage. Dryness of the mouth is usually the first adverse effect to appear. When the daily dose is increased, possible adverse effects include tachycardia, palpitation, urinary hesitancy or retention, blurred vision, dilatation of pupils, increased ocular tension, weakness, nausea, vomiting, headache, dizziness, constipation, drowsiness, hypersensitivity reactions, pruritus, hallucinations, agitation, tremor, gastric irritation and rarely urticaria and other dermatoses. Infrequently, an elderly patient may experience some degree of mental confusion. These adverse reactions can usually be eliminated by reduction in dosage. Very rare cases of aplastic anemia associated with the use of orphenadrine tablets have been reported. No causal relationship has been established.
To report SUSPECTED ADVERSE REACTIONS, contact Sandoz Inc. at 1-800-525-8747 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG ABUSE AND DEPENDENCE
Orphenadrine citrate has been chronically abused for its euphoric effects.[1] The mood elevating effects may occur at therapeutic doses of orphenadrine.[2]
OVERDOSAGE
Orphenadrine citrate is toxic when overdosed and typically induces anticholinergic effects.[3] In a review of orphenadrine toxicity, the minimum lethal dose was found to be 2 to 3 grams for adults; however, the range of toxicity is variable and unpredictable.[4] Treatment for orphenadrine citrate overdose is evacuation of stomach contents (when necessary), charcoal at repeated doses, intensive monitoring and appropriate supportive treatment of any emergent anticholinergic effects.[5]
HOW SUPPLIED
NDC: 71335-0117-1: 20 Tablets in a BOTTLE
NDC: 71335-0117-2: 60 Tablets in a BOTTLE
NDC: 71335-0117-3: 100 Tablets in a BOTTLE
NDC: 71335-0117-4: 90 Tablets in a BOTTLE
NDC: 71335-0117-5: 30 Tablets in a BOTTLE
NDC: 71335-0117-6: 28 Tablets in a BOTTLE
NDC: 71335-0117-7: 45 Tablets in a BOTTLE
NDC: 71335-0117-8: 14 Tablets in a BOTTLE
NDC: 71335-0117-9: 120 Tablets in a BOTTLE
NDC: 71335-0117-0: 56 Tablets in a BOTTLE
| ORPHENADRINE CITRATE
orphenadrine citrate tablet, extended release |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Bryant Ranch Prepack (171714327) |
| Registrant - Bryant Ranch Prepack (171714327) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Bryant Ranch Prepack | 171714327 | REPACK(71335-0117) , RELABEL(71335-0117) | |