KANAMYCIN- kanamycin injection, solution
Fresenius Kabi USA, LLC
----------
Kanamycin Injection, USP
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Kanamycin Injection, USP and other antibacterial drugs, Kanamycin Injection, USP should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria.
BOXED WARNING
Patients treated with aminoglycosides by any route should be under close clinical observation because of the potential toxicity associated with their use. As with other aminoglycosides, the major toxic effects of kanamycin are its action on the auditory and vestibular branches of the eighth nerve and the renal tubules. Neurotoxicity is manifested by bilateral auditory toxicity which often is permanent and, sometimes, by vestibular ototoxicity. Loss of high frequency perception usually occurs before there is noticeable clinical hearing loss and can be detected by audiometric testing. There may not be clinical symptoms to warn of developing cochlear damage. Vertigo may occur and may be evidence of vestibular injury. Other manifestations of neurotoxicity may include numbness, skin tingling, muscle twitching, and convulsions. The risk of hearing loss increases with the degree of exposure to either high peak or high trough serum concentrations and continues to progress after drug withdrawal.
Renal impairment may be characterized by decreased creatinine clearance, the presence of cells or casts, oliguria, proteinuria, decreased urine specific gravity, or evidence of increasing nitrogen retention (increasing BUN, NPN, or serum creatinine).
The risks of severe ototoxic and nephrotoxic reactions are sharply increased in patients with impaired renal function and in those with normal renal function who receive high doses or prolonged therapy.
Renal and eighth nerve function should be closely monitored, especially in patients with known or suspected reduced renal function at the onset of therapy, and also in those whose renal function is initially normal but who develop signs of renal dysfunction during therapy. Serum concentrations of parenterally administered aminoglycosides should be monitored when feasible to assure adequate levels and to avoid potentially toxic levels. Urine should be examined for decreased specific gravity, increased excretion of protein, and the presence of cells or casts. Blood urea nitrogen, serum creatinine, or creatinine clearance should be measured periodically. Serial audiograms should be obtained when feasible in patients old enough to be tested; particularly high risk patients. Evidence of ototoxicity (dizziness, vertigo, tinnitus, roaring in the ears, and hearing loss) or nephrotoxicity requires dosage adjustment or discontinuance of the drug.
Neuromuscular blockade with respiratory paralysis may occur when kanamycin is instilled intraperitoneally concomitantly with anesthesia and muscle-relaxing drugs. Neuromuscular blockade has been reported following parenteral injection and the oral use of aminoglycosides. The possibility of the occurrence of neuromuscular blockade and respiratory paralysis should be considered if aminoglycosides are administered by any route, especially in patients receiving anesthetics, neuromuscular-blocking agents such as tubocurarine, succinylcholine, decamethonium, or in patients receiving massive transfusions of citrate-anticoagulated blood. If blockage occurs, calcium salts may reduce these phenomena but mechanical respiratory assistance may be necessary.
The concurrent and/or sequential systemic, oral, or topical use of kanamycin and other potentially nephrotoxic, and/or neurotoxic drugs, particularly polymyxin B, bacitracin, colistin, amphotericin B, cisplatin, vancomycin, and all other aminoglycosides (including paromomycin) should be avoided because the toxicity may be additive. Other factors which may increase patient risk of toxicity are advanced age and dehydration.
Kanamycin should not be given concurrently with potent diuretics (ethacrynic acid, furosemide, meralluride sodium, sodium mercaptomerin, or mannitol). Some diuretics themselves cause ototoxicity, and intravenously administered diuretics may enhance aminoglycoside toxicity by altering antibiotic concentrations in serum and tissue.
DESCRIPTION:
Kanamycin sulfate is an aminoglycoside antibiotic produced by Streptomyces kanamyceticus. It is D-Streptamine, 0-3-amino-3-deoxy-α-D-glucopyranosyl - (1→6)-0- [6-amino-6-deoxy-α-D-glucopyranosyl - (1→4)]-2-deoxy, sulfate 1:1 (salt). It consists of two amino sugars glycosidically linked to deoxystreptamine.
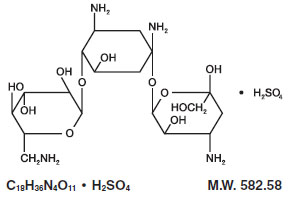
Kanamycin Injection, USP, sterile solution for parenteral administration, contains kanamycin sulfate equivalent to 1 g kanamycin; sodium bisulfite, an antioxidant, 0.66% and 0.45%; and sodium citrate, 2.2% and 2.2% with pH adjusted to 4.5 with sulfuric acid.
CLINICAL PHARMACOLOGY:
The drug is rapidly absorbed after intramuscular injection and peak serum levels are generally reached within approximately one hour. Doses of 7.5 mg/kg give mean peak levels of 22 mcg/mL. At 8 hours following a 7.5 mg/kg dose, mean serum levels are 3.2 mcg/mL. The serum half-life is 2 1/2 hours. Intravenous administration of kanamycin over a period of one hour resulted in serum concentrations similar to those obtained by intramuscular administration.
Kanamycin diffuses rapidly into most body fluids including synovial and peritoneal fluids and bile. Significant levels of the drug appear in cord blood and amniotic fluid following intramuscular administration to pregnant patients. Spinal fluid concentrations in normal infants are approximately 10 to 20 percent of serum levels and may reach 50 percent when the meninges are inflamed.
Studies in normal adult patients have shown only trace levels of kanamycin in spinal fluid. No data are available on adults with meningitis.
The drug is excreted almost entirely by glomerular filtration and is not reabsorbed by the renal tubules. Hence, high concentrations are attained in the nephron, and the urine may contain levels 10 to 20 times higher than those in serum. Little, if any, metabolic transformation occurs. Renal excretion is extremely rapid. In patients with normal renal function, approximately one-half of the administered dose is cleared within 4 hours and excretion is complete within 24 to 48 hours. Patients with impaired renal function or with diminished glomerular filtration pressure excrete kanamycin more slowly. Such patients may build up excessively high blood levels which greatly increase the risk of ototoxic reactions. In severely burned patients the half-life may be significantly decreased and resulting serum concentrations may be lower than anticipated from the mg per kg dose.
Microbiology
Mechanism of Action
Kanamycin, an aminoglycoside, acts by inhibiting the synthesis of protein in susceptible microorganisms. It is bactericidal in vitro against Gram-negative bacteria and certain Gram-positive bacteria.
Mechanism of Resistance
Aminoglycosides are known to be ineffective against
Salmonella and
Shigella species in patients. Therefore,
in vitro susceptibility test results should not be reported.
Enzymatic inactivation of deoxystreptamine is the principal mechanism of resistance to kanamycin.
Aminoglycosides in general have a low order of activity against Gram-positive organisms other than Staphylococcal isolates.
Interactions with Other Antimicrobials
In vitro studies have demonstrated that an aminoglycoside combined with an antibiotic which interferes with cell wall synthesis (i.e., Penicillin G or ampicillin) affects some Group D streptococcal strains synergistically. Bacteriological testing and tests for antibiotic synergism are necessary.
Antibacterial Activity
Kanamycin has been shown to be active against the following bacteria, both in vitro and in clinical infections (see INDICATIONS AND USAGE).
Gram-positive Bacteria
Staphylococcus species
Gram-negative Bacteria
Escherichia coli
Proteus species (indole-positive and indole-negative)
Enterobacter aerogenes
Klebsiella pneumoniae
Serratia marcescens
Acinetobacter species
Kanamycin has demonstrated in vitro activity against the following bacteria. The safety and effectiveness of kanamycin in treating clinical infections due to these bacteria have not been established in adequate and well-controlled trials.
Staphylococcus aureus (including penicillinase and non penicillinase-producing strains)
Staphylococcus epidermidis
N. gonorrhoeae
H. influenzae
Providencia species
Citrobacter freundii
Citrobacter species
Susceptibility Test Methods
When available, the clinical microbiology laboratory should provide cumulative results of the in vitro susceptibility tests for antimicrobial drugs used in local hospitals and practice areas to the physician as periodic reports that describe the susceptibility profile of nosocomial and community-acquired pathogens. These reports should aid the physician in selecting the most effective antimicrobial.
Dilution Techniques
Quantitative methods are used to determine antimicrobial minimal inhibitory concentrations (MICs). These MICs provide estimates of the susceptibility of bacteria to antimicrobial compounds. The MICs should be determined using a standardized test method. 1, 3 Standardized procedures are based on a dilution method (broth or agar) or equivalent with standardized inoculum concentrations and standardized concentrations of kanamycin powder. The MIC values should be interpreted according to the criteria provided in Table 1.
Diffusion Techniques
Quantitative methods that require measurement of zone diameters also provide reproducible estimates of the susceptibility of bacteria to antimicrobial compounds. One such standardized procedure requires the use of standardized inoculum concentrations and paper disks impregnated with 30 mcg of kanamycin. 2, 3 The disk diffusion values should be interpreted according to the criteria provided in Table 1.
Table 1 : Susceptibility Interpretive Criteria for Kanamycin
|
Pathogen |
Susceptibility Interpretive Criteria |
|||||
|
Minimal Inhibitory Concentration (mcg/mL) |
Zone Diameter (mm) |
|||||
|
(S) |
(I) |
(R) |
(S) |
(I) |
(R) |
|
| Enterobacteriaceae
a
|
≤ 16 |
32 |
≥ 64 |
≥ 18 |
14 to 17 |
≤ 13 |
| Staphylococcus spp.
b
|
≤ 16 |
32 |
≥ 64 |
≥ 18 |
14 to 17 |
≤ 13 |
S = Susceptible, I = Intermediate, R = Resistant
a For Salmonella and Shigella spp., aminoglycosides may appear active in vitro but are not effective
clinically; the results should not be reported as susceptible
b For staphylococci that test susceptible, aminoglycosides are used only in combination with other
active agents that test susceptible
A report of "Susceptible" indicates that the antimicrobial is likely to inhibit growth of the pathogen if the antimicrobial compound reaches the concentration at the infection site necessary to inhibit growth of the pathogen. A report of "Intermediate" indicates that the result should be considered equivocal, and if the microorganism is not fully susceptible to alternative clinically feasible drugs, the test should be repeated. This category implies possible clinical applicability in body sites where the drug is physiologically concentrated. This category also provides a buffer zone that prevents small uncontrolled technical factors from causing major discrepancies in interpretation. A report of "Resistant" indicates that the antimicrobial is not likely to inhibit growth of the pathogen if the antimicrobial compound reaches the concentrations usually achievable at the infection site; other therapy should be selected.
Quality Control
Standardized susceptibility test procedures require the use of laboratory controls to monitor and ensure the accuracy and precision of supplies and reagents used in the assay, and the techniques of the individuals performing the test. 1, 2, 3 Standard kanamycin powder should provide the following range of MIC values provided in Table 2. For the diffusion technique using the 30-mcg kanamycin disk the criteria provided in Table 2 should be achieved.
Table 2 : Acceptance Quality Control Ranges for Susceptibility Testing
|
Quality Control Organism |
Minimum Inhibitory Concentrations (mcg/mL) |
Zone Diameter (mm) |
| Escherichia coli
ATCC 25922 |
1 to 4 |
17 to 25 |
| Staphylococcus aureus
ATCC 25923 |
Not Applicable |
19 to 26 |
| Staphylococcus aureus
ATCC 29213 |
1 to 4 |
Not Applicable |
INDICATIONS AND USAGE:
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Kanamycin Injection and other antibacterial drugs, Kanamycin Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
Kanamycin injection is indicated in the short-term treatment of serious infections caused by susceptible strains of the designated microorganisms below. Bacteriological studies to identify the causative organisms and to determine their susceptibility to kanamycin should be performed. Therapy may be instituted prior to obtaining the results of susceptibility testing.
Kanamycin may be considered as initial therapy in the treatment of infections where one or more of the following are the known or suspected pathogens: E. coli, Proteus species (both indole-positive and indole-negative), Enterobacter aerogenes, Klebsiella pneumoniae, Serratia marcescens, Acinetobacter species. The decision to continue therapy with the drug should be based on results of the susceptibility tests, the response of the infection to therapy, and the important additional concepts contained in the BOXED WARNING above.
In serious infections when the causative organisms are unknown, kanamycin injection, may be administered as initial therapy in conjunction with a penicillin- or cephalosporin-type drug before obtaining results of susceptibility testing. If anaerobic organisms are suspected, consideration should be given to using other suitable antimicrobial therapy in conjunction with kanamycin.
Although kanamycin is not the drug of choice for staphylococcal infections, it may be indicated under certain conditions for the treatment of known or suspected staphylococcal disease. These situations include the initial therapy of severe infections where the organism is thought to be either a Gram-negative bacterium or a staphylococcus, infections due to susceptible strains of staphylococci in patients allergic to other antibiotics, and mixed staphylococcal/Gram-negative infections.
CONTRAINDICATIONS:
A history of hypersensitivity or toxic reaction to one aminoglycoside may also contraindicate the use of any other aminoglycoside, because of the known cross-sensitivity and cumulative effects of drugs in this category.
THIS DRUG IS NOT INDICATED IN LONG-TERM THERAPY (e.g., Tuberculosis) BECAUSE OF THE TOXIC HAZARD ASSOCIATED WITH EXTENDED ADMINISTRATION.
WARNINGS:
See BOXED WARNING above.
Aminoglycosides can cause fetal harm when administered to pregnant women. Aminoglycoside antibiotics cross the placenta and there have been several reports of total, irreversible, bilateral congenital deafness in children whose mothers received streptomycin during pregnancy. Although serious side effects to fetus or newborn have not been reported in treatment of pregnant women with other aminoglycosides, the potential for harm exists.
Reproductive studies have been performed in rats and rabbits and have revealed no evidence of impaired fertility or teratogenic effects. Dosages of 200 mg/kg/day in pregnant rats and pregnant guinea pigs led to hearing impairment in the off-spring. There are no well-controlled studies in pregnant women but clinical experience does not include any positive evidence of adverse effects on the fetus. However, if the drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard on the fetus.
Contains sodium bisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in nonasthmatic people.
PRECAUTIONS:
General
Prescribing Kanamycin Injection in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Neurotoxic and nephrotoxic antibiotics may be almost completely absorbed from body surfaces (except the urinary bladder) after local irrigation and after topical application during surgical procedures. The potential toxic effects of antibiotics administered in this fashion (oto- and nephrotoxicity, neuromuscular blockade, respiratory paralysis) should be considered (see BOXED WARNING).
Increased nephrotoxicity has been reported following concomitant administration of aminoglycoside antibiotics and with some cephalosporins.
Aminoglycosides should be used with caution in patients with neuromuscular disorders such as myasthenia gravis, Parkinsonism, or infant botulism, since these drugs may aggravate muscle weakness because of their potential curare-like effect on neuromuscular function.
Elderly patients may have a decrease in renal function which may not be evident in the results of routine screening tests, such as BUN or serum creatinine levels. Measurement of creatinine clearance or an estimate based on published nomograms or equations may be more useful. Monitoring of renal function during treatment with kanamycin, as with other aminoglycosides, is particularly important in such patients.
Because of high concentrations of kanamycin in the urinary excretory system, patients should be well hydrated before treatment to prevent irritation of the renal tubules.
NOTE: The risk of toxic reactions is low in well-hydrated patients with normal kidney function,
who receive a total dose of 15 g of kanamycin or less.
Treatment with kanamycin may result in overgrowth of nonsusceptible organisms. If this occurs kanamycin should be discontinued and appropriate therapy initiated.
Information for Patients
Patients should be counseled that antibacterial drugs including Kanamycin Injection should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When Kanamycin Injection is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by Kanamycin Injection or other antibacterial drugs in the future.
Laboratory Tests
Tests of eighth cranial nerve functions: Serial audiometric tests are suggested, particularly when renal function is impaired and/or prolonged aminoglycoside therapy is required; such tests should also be repeated periodically after treatment if there is evidence of a hearing deficit or vestibular abnormalities before or during therapy, or when consecutive or concomitant use of other potentially ototoxic drug is unavoidable.
Test of renal function: It should be emphasized that since renal function may alter appreciably during therapy, renal function should be tested daily or more frequently. Urine should be examined for increased excretion of protein and for presence of cells and casts, keeping in mind the effects of the primary illness on these tests. One or more of the following laboratory measurements should be obtained at the onset of therapy, frequently during therapy, and at, or shortly after, the end of therapy:
Creatinine clearance rate (either carefully measured or estimated from published nomograms or equations based on patient’s age, sex, body weight, and serial creatinine concentrations) (preferred over BUN).
Serum creatinine concentration (preferred over BUN).
Blood urea nitrogen (BUN).
More frequent testing is desirable if renal function is changing. If signs of renal irritation appear, such as casts, white or red cells, and albumin, hydration should be increased and a reduction in dosage may be desirable (see DOSAGE AND ADMINISTRATION). These signs usually disappear when treatment is completed. However, if azotemia or a progressive decrease of urine output occurs, treatment should be stopped.
Drug Interactions
In vitro mixing of an aminoglycoside with beta-lactam-type antibiotics (penicillins or cephalosporins) may result in a significant mutual inactivation. Even when an aminoglycoside and a penicillin-type drug are administered separately by different routes, a reduction in aminoglycoside serum half-life or serum levels has been reported in patients with impaired renal function and in some patients with normal renal function. Usually, such inactivation of the aminoglycoside is clinically significant only in patients with severely impaired renal function (see also Laboratory Test Interactions). See BOXED WARNING regarding concurrent use of potent diuretics, concurrent and/or sequential use of other neurotoxic and/or nephrotoxic antibiotics, and for other essential information.
Laboratory Test Interactions
Concomitant cephalosporin therapy may spuriously elevate creatinine determinations.
The inactivation between aminoglycosides and beta-lactam antibiotics described in Drug Interactions may continue in specimens of body fluids collected for assay, resulting in inaccurate, false low aminoglycoside readings. Such specimens should be properly handled, i.e., assayed promptly, frozen, or treated with beta-lactamase.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies have not been performed with kanamycin to determine its effect in carcinogenesis, mutagenesis, or impairment of fertility.
Nursing Mothers
Kanamycin is excreted in minute amounts in human milk. Because of the potential for serious adverse reactions from aminoglycosides in nursing infants, a decision should be made whether to discontinue nursing or to discontinue the drug taking into account the importance of the drug to the mother.
ADVERSE REACTIONS:
Kanamycin has the potential to induce auditory and sometimes vestibular toxicity, renal toxicity, and neuromuscular blockade. The risks are higher for patients with a present or past history of renal impairment (especially if hemodialysis is required): for those receiving concomitant or sequential treatment with other ototoxic or nephrotoxic drugs or rapid acting diuretic agents given intravenously (ethacrynic acid, furosemide, and mannitol), and for patients treated for longer periods and/or with higher doses than recommended.
Ototoxicity
Toxic effects of kanamycin on the eighth cranial nerve can result in partially reversible or irreversible bilateral loss of hearing, loss of balance, or both. Tinnitus or vertigo may or may not be experienced. Cochlear damage is usually manifested initially by small changes in audiometric test results at the high frequencies and may not be associated with subjective hearing loss. Vestibular dysfunction is usually manifested by nystagmus, vertigo, nausea, vomiting, or acute Meniere’s syndrome.
Nephrotoxicity
Albuminuria, presence of red and white cells, and granular casts; azotemia and oliguria have been reported. Renal function changes are usually reversible when the drug is discontinued. Renal impairment may be characterized by a rise in serum creatinine and may be accompanied by oliguria, presence of casts, cells, and protein in the urine, by rising levels of BUN or by decrease in creatinine clearance.
Neuromuscular Blockade
Acute muscular paralysis and apnea can occur following treatment with aminoglycoside antibiotics. Neurotoxicity can occur after intrapleural and interperitoneal instillation of large doses of an aminoglycoside; however, the reaction has followed intravenous, intramuscular, and even the oral administration of these agents.
Other
Some local irritation or pain may follow the intramuscular injection of kanamycin. Other adverse reactions of the drug reported on rare occasions are skin rash, drug fever, headache, paresthesia, nausea, vomiting, and diarrhea. The “malabsorption syndrome” characterized by an increase in fecal fat, decrease in serum carotene, and fall in xylose absorption, reportedly has occurred with prolonged therapy.
OVERDOSAGE:
In the event of overdosage or toxic reaction, hemodialysis or peritoneal dialysis will aid in the removal of kanamycin from the blood. In the newborn infant, exchange transfusion may also be considered.
DOSAGE AND ADMINISTRATION:
Kanamycin injection may be given intramuscularly or intravenously. The patient’s pretreatment body weight should be obtained for calculation of the correct dosage. The dosage of an aminoglycoside in obese patients should be based on an estimate of the lean body mass. The status of renal function should be determined by measurement of serum creatinine concentration or calculation of the endogenous creatinine clearance rate. The blood urea nitrogen (BUN) level is much less reliable for this purpose. Renal function should be reassessed frequently during therapy.
It is desirable to measure both peak and trough serum concentrations intermittently during therapy since both concentrations are used to determine the adequacy and safety of the dose and to adjust the dosage during treatment. Peak serum concentrations (30 to 90 minutes after injection) above 35 mcg per mL and trough concentrations (just prior to the next dose) above 10 mcg per mL should be avoided.
Intramuscular Route
Inject deeply into the upper outer quadrant of the gluteal muscle. The recommended dose for adults or children is 15 mg/kg/day in two equally divided dosages administered at equally divided intervals; i.e., 7.5 mg/kg q12h. If continuously high blood levels are desired, the daily dose of 15 mg/kg may be given in equally divided doses every 6 or 8 hours. Treatment of patients in the heavier weight classes, i.e., 100 kg, should not exceed 1.5 g/day.
In patients with impaired renal function, it is desirable to follow therapy by appropriate serum assays. If this is not feasible, a suggested method is to reduce the frequency of administration in patients with renal dysfunction. The interval between doses may be calculated with the following formula:
Serum creatinine (mg/100 mL) x 9 = Dosage Interval (in hours); e.g., if the serum creatinine is 2 mg, the recommended dose (7.5 mg/kg) should be administered every 18 hours. Changes in creatinine concentration during therapy would, of course, necessitate changes in the dosage frequency.
It is desirable to limit the duration of treatment with kanamycin to short-term. The usual duration of treatment is 7 to 10 days. Total daily dose by all routes of administration should not exceed 1.5 g/day. If longer therapy is required, measurement of kanamycin peak and trough serum concentrations is particularly important as a basis for determining the adequacy and safety of the dose. These patients should be carefully monitored for changes in renal, auditory, and vestibular function. Dosage should be adjusted as needed. The risks of toxicity multiply as the length of treatment increases.
At the recommended dosage level, uncomplicated infections due to kanamycin-susceptible organisms should respond to therapy in 24 to 48 hours. If definite clinical response does not occur within 3 to 5 days, therapy should be stopped and the antibiotic susceptibility pattern of the invading organism should be rechecked. Failure of the infection to respond may be due to resistance of the organism or to the presence of septic foci requiring surgical drainage.
Intravenous Administration
The dose should not exceed 15 mg/kg per day and must be administered slowly. The solution for intravenous use is prepared by adding the contents of a 500 mg vial to 100 to 200 mL of sterile diluent such as Normal Saline or 5% Dextrose in Water, or the contents of a 1 g vial to 200 to 400 mL of sterile diluent. The appropriate dose is administered over a 30- to 60-minute period. The total daily dose should be divided into 2 or 3 equally divided doses.
Kanamycin Injection, USP should not be physically mixed with other antibacterial agents but each should be administered separately in accordance with its recommended route of administration and dosage schedule.
Intraperitoneal Use
(Following exploration for established peritonitis or after peritoneal contamination due to fecal spill during surgery.)
Adults
500 mg diluted in 20 mL sterile distilled water may be instilled through a polyethylene catheter sutured into the wound at closure. If possible, instillation should be postponed until the patient has fully recovered from the effects of anesthesia and muscle-relaxing drugs (see duration of treatment statement above and BOXED WARNING). Serum levels should be carefully monitored during treatment.
NOTE: The pediatric dosage form “75 mg/2 mL” should be used for pediatric patients.
Aerosol Treatment
250 mg two to four times a day. Withdraw 250 mg (1 mL) from a 500 mg vial and dilute it with 3 mL Physiological Saline and nebulize. Serum levels should be carefully monitored during treatment.
Other Routes of Administration
Kanamycin injection in concentrations of 0.25 percent (2.5 mg/mL) has been used as an irrigating solution in abscess cavities, pleural space, peritoneal and ventricular cavities. Possible absorption of kanamycin by such routes must be taken into account and dosage adjustments should be arranged so that a maximum total dose of 1.5 g/day by all routes of administration is not exceeded. Serum levels should be carefully monitored during treatment.
STABILITY:
Occasionally, some vials may darken during the shelf life of the product, but this does not indicate a loss of potency.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever container and solution permit.
HOW SUPPLIED:
| Product
No. | NDC
No. | Strength
| Vial Size
|
| 305903
| 63323-359-03
| 1 gram per
3 mL (333 mg per mL) | 3 mL fill in a 5 mL vial.
|
Kanamycin Injection, USP, is supplied in packages of 10 vials.
The container closure is not made with natural rubber latex.
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
REFERENCES:
- Clinical and Laboratory Standards Institute (CLSI). Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically; Approved Standard - Tenth Edition. CLSI document M07-A10, Clinical and Laboratory Standards Institute, 950 West Valley Road, Suite 2500, Wayne, Pennsylvania 19087, USA, 2015.
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Disk Diffusion Susceptibility Tests; Approved Standard - Twelfth Edition. CLSI document M02- A12, Clinical and Laboratory Standards Institute, 950 West Valley Road, Suite 2500, Wayne, Pennsylvania 19087, USA, 2015.
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Susceptibility Testing; Twenty-fifth Informational Supplement.. CLSI document M100-S25, Clinical and Laboratory Standards Institute, 950 West Valley Road, Suite 2500, Wayne, Pennsylvania 19087, USA, 2015.
PACKAGE LABEL - PRINCIPAL DISPLAY - Kanamycin 3 mL Vial Label
NDC 63323-359-03
305903
Kanamycin Injection, USP
*1 g per 3 mL (333 mg per mL)
For Intramuscular or Intravenous Use
3 mL Vial
Rx only

PACKAGE LABEL - PRINCIPAL DISPLAY - Kanamycin 3 mL Vial Tray Label
NDC 63323-359-03
305903
Kanamycin Injection, USP
*1 g per 3 mL (333 mg per mL)
For Intramuscular or Intravenous Use
3 mL Vial
Rx only
10 Vials

| KANAMYCIN
kanamycin injection, solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Fresenius Kabi USA, LLC (608775388) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Fresenius Kabi USA, LLC | 840771732 | manufacture(63323-359) | |
