Label: XHANCE- fluticasone propionate spray, metered
- NDC Code(s): 71143-375-01, 71143-375-98, 71143-375-99
- Packager: OptiNose US, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated March 15, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use XHANCE® safely and effectively. See full prescribing information for XHANCE®.
XHANCE® (fluticasone propionate) nasal spray
Initial U.S. Approval: 1994RECENT MAJOR CHANGES
Indications and Usage (1.2) 03/2024
INDICATIONS AND USAGE
DOSAGE AND ADMINISTRATION
- •
- Recommended Dosage: 186 mcg (1 spray per nostril) or 372 mcg (2 sprays per nostril) twice daily. (2.1)
- •
- For nasal use only. Shake before use. Prime before initial use. (2.2)
- •
- XHANCE is delivered into the nose by actuating the pump spray into one nostril while simultaneously blowing into the mouthpiece of the device. (2.2)
DOSAGE FORMS AND STRENGTHS
Nasal spray: 93 mcg of fluticasone propionate in each spray. (3)
CONTRAINDICATIONS
Hypersensitivity to any ingredient in XHANCE. (4)
WARNINGS AND PRECAUTIONS
- •
- Local Nasal Adverse Reactions: epistaxis, erosion, ulceration, septal perforation, Candida albicans infection, and impaired wound healing. Monitor patients periodically for signs of adverse effects on the nasal mucosa. Avoid use in patients with recent nasal ulcerations, nasal surgery, or nasal trauma. (5.1)
- •
- Glaucoma and Cataracts may occur with long-term use. Consider referral to an ophthalmologist in patients who develop ocular symptoms or use XHANCE long-term. (5.2)
- •
- Hypersensitivity reactions (e.g., anaphylaxis, angioedema, urticaria, contact dermatitis, rash, hypotension, and bronchospasm) have been reported after administration of fluticasone propionate. Discontinue XHANCE if such reactions occur. (5.3)
- •
- Immunosuppression and Risk of Infection: potential increased susceptibility to or worsening of infections (e.g., existing tuberculosis; fungal, bacterial, viral, or parasitic infection; ocular herpes simplex). Use with caution in patients with these infections. More serious or even fatal course of chickenpox or measles can occur in susceptible patients. (5.4)
- •
- Hypercorticism and adrenal suppression may occur with very high dosages or at the regular dosage in susceptible individuals. If such changes occur, discontinue XHANCE slowly. (5.5)
- •
- Assess for decrease in bone mineral density initially and periodically thereafter. (5.7)
ADVERSE REACTIONS
- •
- CRSwNP: The most common adverse reactions (incidence ≥ 3%) are epistaxis, nasal septal ulceration, nasopharyngitis, nasal mucosal erythema, nasal mucosal ulcerations, nasal congestion, acute sinusitis, nasal septal erythema, headache, and pharyngitis. (6.1)
- •
-
CRSsNP: The most common adverse reactions (incidence ≥ 3%) are epistaxis, headache, and nasopharyngitis (6.1).
To report SUSPECTED ADVERSE REACTIONS, contact OptiNose US, Inc. at 1-833-678-6673 and safety@optinose.com or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
Strong cytochrome P450 3A4 inhibitors (e.g., ritonavir, ketoconazole): Use not recommended. May increase risk of systemic corticosteroid effects. (7.1)
USE IN SPECIFIC POPULATIONS
Hepatic impairment: Monitor patients for signs of increased drug exposure. (8.6)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 3/2024
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Chronic Rhinosinusitis with Nasal Polyps
1.2 Chronic Rhinosinusitis without Nasal Polyps
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
2.2 Administration Information
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Local Nasal Adverse Reactions
5.2 Glaucoma and Cataracts
5.3 Hypersensitivity Reactions Including Anaphylaxis
5.4 Immunosuppression and Risk of Infection
5.5 Hypercorticism and Adrenal Suppression
5.6 Drug Interactions with Strong Cytochrome P450 3A4 Inhibitors
5.7 Reduction in Bone Mineral Density
5.8 Effect on Growth
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Inhibitors of Cytochrome P450 3A4
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Hepatic Impairment
8.7 Renal Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Chronic Rhinosinusitis with Nasal Polyps
14.2 Chronic Rhinosinusitis without Nasal Polyps
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
The recommended dosage of XHANCE is 186 mcg (1 spray per nostril) or 372 mcg (2 sprays per nostril) twice daily (total daily dose of 372 mcg or 744 mcg). The maximum total daily dosage should not exceed 2 sprays in each nostril twice daily (total daily dose of 744 mcg).
Patients should use XHANCE at regular intervals since its effectiveness depends on regular use. Individual patients will experience a variable time to onset and different degrees of symptom relief.
The safety and efficacy of XHANCE when administered in excess of recommended doses have not been established.
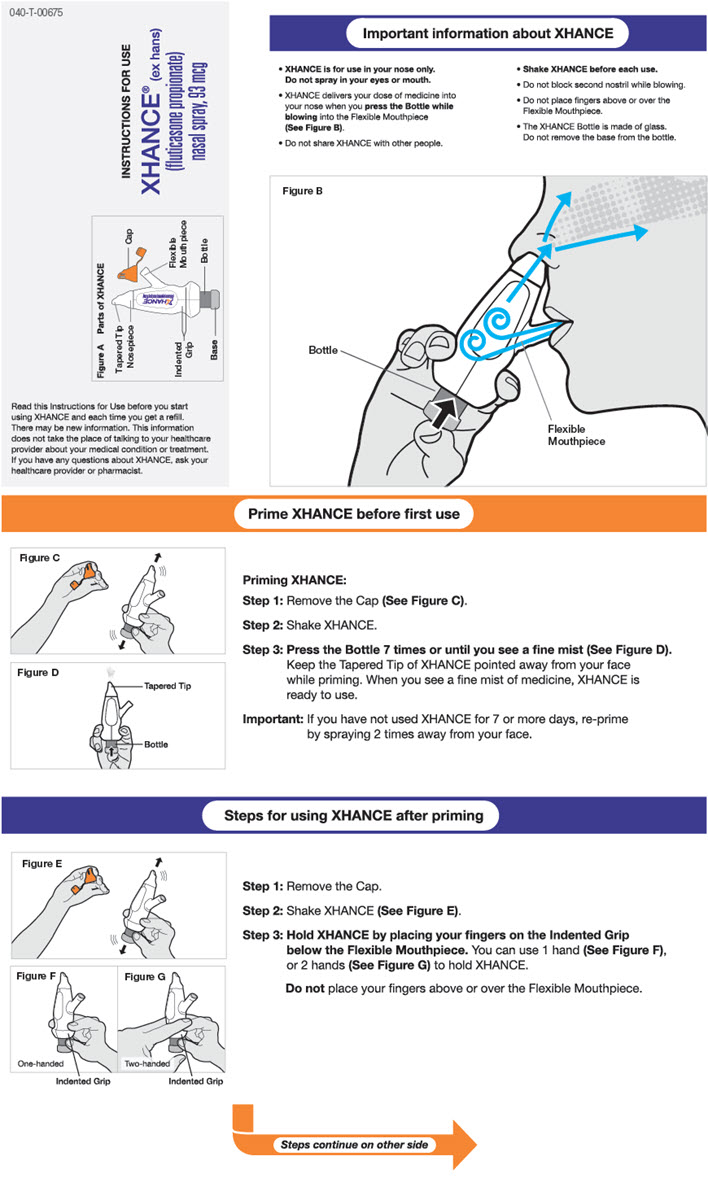
2.2 Administration Information
- •
- Shake XHANCE before each use.
- •
- Administer XHANCE by the nasal route only. Avoid spraying directly on the nasal septum.
Priming
Before initial use, prime XHANCE by first gently shaking and then pressing the bottle 7 times or until a fine mist appears. Direct the spray into the air, away from the face. When XHANCE has not been used for ≥ 7 days, prime the pump again by shaking and releasing 2 sprays into the air, away from the face.
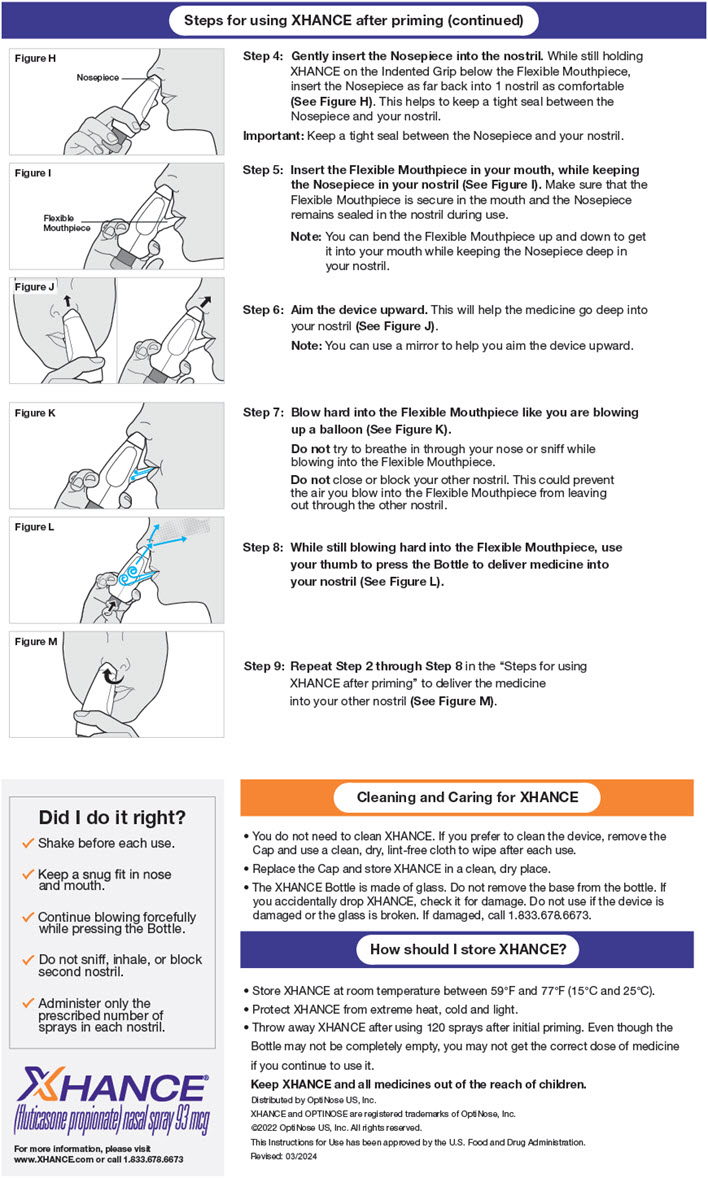
Administration Instructions
XHANCE is delivered into the nose by actuating the pump spray into one nostril while simultaneously blowing (exhaling) into the mouthpiece of the device. To administer XHANCE, insert the tapered tip of the cone-shaped nosepiece deep into one nostril and form a tight seal between the nosepiece and the nostril. Next, place the flexible mouthpiece into the mouth, bending it as necessary to maintain a tight seal with the nostril. Blow into the mouthpiece, and while continuing to blow, push the bottle up to actuate the spray pump. Continuing to blow through the mouth, but not inhaling or exhaling through the nose, at the time of actuation is important to achieve intended drug deposition. Repeat the process in the other nostril for a full dose [see Instructions for Use].
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Local Nasal Adverse Reactions
Epistaxis, Nasal Erosions and Ulcerations
In placebo-controlled clinical trials of 16 weeks duration, epistaxis, nasal erosions, and nasal ulcerations were reported more frequently in patients treated with XHANCE than those who received placebo [see Adverse Reactions (6.1)].
Nasal Septal Perforation
Nasal septal perforations have been reported in patients following the nasal application of XHANCE. In placebo-controlled clinical trials of 16 weeks duration, nasal septal perforation was reported in 1 (0.3%) patient treated with XHANCE compared with none treated with placebo. The patient had a prior history of nasal/sinus surgery. Three (0.3%) patients treated with XHANCE in uncontrolled, open-label trials of 3 to 12 months duration developed nasal septal perforations.
As with any long term topical treatment of the nasal cavity, patients using XHANCE over several months or longer should be examined periodically for possible changes in the nasal mucosa. If a septal perforation is noted, discontinue XHANCE. Avoid spraying XHANCE directly on the septum.
Candida Infection
In clinical trials with XHANCE, localized infections with Candida albicans have been observed. Eight (0.9%) patients in uncontrolled, open-label trials of 3 to 12 months duration developed Candida albicans infections (nasal, pharyngeal, esophageal or intestinal). If such an infection develops, it may require treatment with appropriate local therapy and discontinuation of XHANCE. Patients using XHANCE should be examined periodically for evidence of Candida infection in the nasal and oropharyngeal mucosa.
Impaired Wound Healing
Because of the inhibitory effect of corticosteroids on wound healing, patients who have experienced recent nasal ulcerations, nasal surgery, or nasal trauma should avoid using XHANCE until healing has occurred.
5.2 Glaucoma and Cataracts
Nasal and inhaled corticosteroids, including fluticasone propionate, may result in the development of glaucoma and/or cataracts. In placebo-controlled clinical trials of 16 weeks duration, cataracts were reported in 4 (1.2%) patients treated with XHANCE, compared with 3 (1.9%) patients treated with placebo. Among these patients, 2 patients treated with XHANCE reported subcapsular cataracts compared with none treated with placebo. Eleven patients (1.2%) in uncontrolled, open-label trials of 3 to 12 months duration developed new or worsening cataracts, of which none were subcapsular. Therefore, close monitoring is warranted in patients with a change in vision or with a history of increased intraocular pressure (IOP), glaucoma, and/or cataracts. Consider referral to an ophthalmologist in patients who develop ocular symptoms or use XHANCE long-term.
5.3 Hypersensitivity Reactions Including Anaphylaxis
Hypersensitivity reactions (e.g., anaphylaxis, angioedema, urticaria, contact dermatitis, rash, hypotension, and bronchospasm) have been reported after administration of fluticasone propionate. XHANCE is contraindicated in patients with known hypersensitivity to fluticasone propionate or any of the ingredients of XHANCE. Discontinue XHANCE if such reactions (e.g., anaphylaxis, angioedema, urticaria, contact dermatitis, rash, hypotension, and bronchospasm) occur [see Contraindications (4) and Adverse Reactions (6.1)].
5.4 Immunosuppression and Risk of Infection
Persons who are using drugs that suppress the immune system, such as corticosteroids, including XHANCE, are more susceptible to infections than healthy individuals and may experience a worsening of existing infections. Chickenpox and measles, for example, can have a more serious or even fatal course in susceptible adults using corticosteroids. In such adults who have not had these diseases or been properly immunized, particular care should be taken to avoid exposure. How the dose, route, and duration of corticosteroid administration affect the risk of developing a disseminated infection is not known. The contribution of the underlying disease and/or prior corticosteroid treatment to the risk is also not known. If a patient is exposed to chickenpox, prophylaxis with varicella zoster immune globulin (VZIG) may be indicated. If a patient is exposed to measles, prophylaxis with pooled intramuscular immunoglobulin (IG) may be indicated. (See the respective Prescribing Information for VZIG and IG.) If chickenpox develops, treatment with antiviral agents may be considered.
Corticosteroids should be used with caution, if at all, in patients with active or quiescent tuberculosis infections of the respiratory tract; systemic fungal, bacterial, viral, or parasitic infections; or ocular herpes simplex [see Adverse Reactions (6.1)].
5.5 Hypercorticism and Adrenal Suppression
Hypercorticism and adrenal suppression may occur when nasal corticosteroids, such as XHANCE, are used at higher than recommended dosages or in susceptible individuals at recommended dosages. Since fluticasone propionate is absorbed into the circulation and can be systemically active at higher doses, recommended dosages of XHANCE should not be exceeded to avoid hypothalamic-pituitary-adrenal (HPA) dysfunction. A relationship between plasma levels of fluticasone propionate and inhibitory effects on stimulated cortisol production has been shown after 4 weeks of pulmonary treatment with fluticasone propionate inhalation aerosol. Since individual sensitivity to effects on cortisol production exists, physicians should consider this information when prescribing XHANCE.
Patients treated with XHANCE should be observed carefully for any evidence of systemic corticosteroid effects such as hypercorticism and adrenal suppression (including adrenal crisis). If such effects occur, the dosage of XHANCE should be reduced slowly, consistent with accepted procedures for reducing systemic corticosteroids, and other treatments for management of nasal symptoms should be considered. Particular care should be taken in observing patients postoperatively or during periods of stress for evidence of inadequate adrenal response.
The replacement of a systemic corticosteroid with a topical corticosteroid can be accompanied by signs of adrenal insufficiency. In addition, some patients may experience symptoms of corticosteroid withdrawal (e.g., joint and/or muscular pain, lassitude, depression). After withdrawal from systemic corticosteroids, a number of months are required for recovery of HPA function. Patients previously treated for prolonged periods with systemic corticosteroids and transferred to topical corticosteroids should be carefully monitored for acute adrenal insufficiency in response to stress such as trauma, surgery, infection (particularly gastroenteritis), or other conditions associated with severe electrolyte loss. In patients who have asthma or other clinical conditions requiring long-term systemic corticosteroid treatment, rapid decreases in systemic corticosteroid dosages may cause a severe exacerbation of their symptoms [see Adverse Reactions (6.1) and Clinical Pharmacology (12.2)].
5.6 Drug Interactions with Strong Cytochrome P450 3A4 Inhibitors
The use of strong cytochrome P450 3A4 (CYP3A4) inhibitors (e.g., ritonavir, atazanavir, clarithromycin, indinavir, itraconazole, nefazodone, nelfinavir, saquinavir, ketoconazole, telithromycin, conivaptan, lopinavir, voriconazole) with XHANCE is not recommended because increased systemic corticosteroid adverse effects may occur [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)].
5.7 Reduction in Bone Mineral Density
Decreases in bone mineral density (BMD) have been observed with long-term oral inhalation of products containing corticosteroids into the lungs. The clinical significance of small changes in BMD with regard to long-term consequences such as fracture is unknown. Patients with major risk factors for decreased bone mineral content, such as prolonged immobilization, family history of osteoporosis, postmenopausal status, tobacco use, advanced age, poor nutrition, or chronic use of drugs that can reduce bone mass (e.g., anticonvulsants, oral corticosteroids), should be monitored and treated with established standards of care.
A 2-year trial in 160 patients (females aged 18 to 40 years, males aged 18 to 50 years) with asthma receiving chlorofluorocarbon (CFC)-propelled fluticasone propionate inhalation aerosol 88 or 440 mcg twice daily demonstrated no statistically significant changes in BMD at any time point (24, 52, 76, and 104 weeks of double-blind treatment) as assessed by dual-energy x-ray absorptiometry at lumbar regions L1 through L4.
5.8 Effect on Growth
Nasal corticosteroids, including XHANCE, may cause a reduction in growth velocity when administered to pediatric patients. The safety and effectiveness of XHANCE has not been established in pediatric patients [see Use in Specific Populations (8.4)].
-
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
- •
- Local Nasal Adverse Reactions: [see Warnings and Precautions (5.1)]
- •
- Glaucoma and Cataracts [see Warnings and Precautions (5.2)]
- •
- Hypersensitivity Reactions including Anaphylaxis [see Contraindications (4) and Warnings and Precautions (5.3)]
- •
- Immunosuppression and Risk of Infection [see Warnings and Precautions (5.4)]
- •
- Hypercorticism and Adrenal Suppression [see Warnings and Precautions (5.5)]
- •
- Reduction in Bone Mineral Density [see Warnings and Precautions (5.7)]
- •
- Effect on Growth [see Warnings and Precautions (5.8)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Chronic Rhinosinusitis with Nasal Polyps
The safety data described below are based on two placebo-controlled clinical trials evaluating doses of a fluticasone propionate nasal spray with an exhalation delivery system from 93 mcg twice daily to 372 mcg twice daily. Both trials were 16-weeks in duration with an additional 8-week open-label extension. The trials included a total of 643 adult patients with bilateral nasal polyps and associated moderate or severe nasal congestion of which 161 received 93 mcg twice daily, 160 received 186 mcg twice daily, 161 received 372 mcg twice daily and 161 received placebo. The overall pooled safety data included 296 (46.0%) Female, 347 (54.0%) Male, 584 (90.8%) White, 39 (6.1%) Black, 9 (1.4%) Asian, and 11 (1.7%) patients classified as Other. Of these patients, 45 (7%) were 65 years of age or older.
Table 1 displays adverse reactions with an incidence of ≥ 3% in the XHANCE 186 mcg and 372 mcg twice daily patients, and more common than placebo.
Table 1. Summary of Adverse Reactions with XHANCE Reported in ≥ 3% of Patients with CRSwNP and More Common Than Placebo in Placebo-Controlled Studies XHANCE Adverse Reaction Placebo
(N = 161)
n (%)186 mcg bid*
(N = 160)
n (%)372 mcg bid^
(N = 161)
n (%)*186 mcg bid = 1 spray per nostril twice daily
^372 mcg bid = 2 sprays per nostril twice daily
1 Includes spontaneous adverse reaction reports
2 Include ulcerations and erosionsEpistaxis1
4 (2.5)
19 (11.9)
16 (9.9)
Nasopharyngitis
8 (5.0)
3 (1.9)
12 (7.5)
Nasal septal ulceration2
3 (1.9)
11 (6.9)
12 (7.5)
Nasal congestion
6 (3.7)
7 (4.4)
9 (5.6)
Acute sinusitis
6 (3.7)
7 (4.4)
8 (5.0)
Headache
5 (3.1)
8 (5.0)
6 (3.7)
Pharyngitis
2 (1.2)
2 (1.3)
5 (3.1)
Nasal mucosal ulceration2
2 (1.3)
6 (3.8)
4 (2.5)
Nasal mucosal erythema
6 (3.7)
9 (5.6)
8 (5.0)
Nasal septal erythema
3 (1.9)
6 (3.8)
7 (4.3)
Other adverse reactions with XHANCE observed with an incidence < 3% but ≥ 1% and more common than placebo included: nasal dryness, sinusitis, oropharyngeal pain, toothache, intraocular pressure increase, dizziness, abdominal discomfort, and weight increase.
5.0% of patients treated with XHANCE 186 mcg twice daily and 1.2% of patients treated with 372 mcg twice daily discontinued from the clinical trials prior to the open-label extension because of adverse reactions compared to 4.3% of patients treated with placebo.
There were no clinically relevant differences in the incidence of adverse reactions based on gender. Clinical trials did not include sufficient numbers of non-Caucasian patients or patients aged 65 years and older to determine whether they respond differently from Caucasian or younger patients, respectively.
Chronic Rhinosinusitis without Nasal Polyps
The safety of XHANCE was based on a pooled safety population that reflect the exposure of XHANCE in 367 adults with chronic rhinosinusitis with (CRSwNP) or without nasal polyps (CRSsNP) exposed for 24 weeks duration. XHANCE was studied in two randomized, placebo-controlled, multicenter trials (Trial 3 and Trial 4). Patients received either XHANCE 186 mcg (1 spray per nostril) twice daily, XHANCE 372 mcg (2 sprays per nostril) twice daily, or placebo delivered with an exhalation delivery system. The pooled safety population had a mean age of 49 years [age range: 18 to 87], and were 55% male, 94% White, 96% non-Hispanic or Latino, 4% Black or African American, and 2% Asian.
In these trials, 1.6% of patients treated with XHANCE 186 mcg (1 spray per nostril) twice daily and 1.6% of patients treated with 372 mcg (2 sprays per nostril) twice daily discontinued treatment due to adverse reactions compared to 2.7% of patients treated with placebo.
The most common adverse reactions that occurred at a rate of ≥ 3% in patients treated with XHANCE and at a higher rate in at least one treatment group than placebo from the pooled safety population (Trials 3 and 4) are shown in Table 2.
Table 2. Summary of Adverse Reactions with XHANCE Reported in ≥ 3% of Patients and More Common Than Placebo from Pooled Safety Population (Trials 3 and 4)1 XHANCE Adverse Reaction Placebo
(N = 187)
n (%)186 mcg bid*
(N = 184)
n (%)372 mcg bid^
(N = 183)
n (%)*186 mcg bid = 1 spray per nostril twice daily
^372 mcg bid = 2 sprays per nostril twice daily
1 Patients in Trial 3 included adults with CRSsNP and Trial 4 included adults with CRSsNP and CRSwNPEpistaxis
1 (0.5%)
9 (4.9%)
20 (10.9%)
Headache
7 (3.7%)
4 (2.2%)
10 (5.5%)
Nasopharyngitis
8 (4.3%)
9 (4.9%)
7 (3.8%)
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of XHANCE. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Eye disorders: central serous chorioretinopathy
Respiratory, thoracic, and mediastinal disorders: dysphonia, nasal discomfort, and nasal dryness
Skin and subcutaneous tissue disorders: pruritus, rash
-
7 DRUG INTERACTIONS
7.1 Inhibitors of Cytochrome P450 3A4
Fluticasone propionate is a substrate of CYP3A4. The use of strong CYP3A4 inhibitors (e.g., ritonavir, atazanavir, clarithromycin, indinavir, itraconazole, nefazodone, nelfinavir, saquinavir, ketoconazole, telithromycin, conivaptan, lopinavir, voriconazole) with XHANCE is not recommended because increased systemic corticosteroid adverse effects may occur.
Ritonavir
A drug interaction trial with fluticasone propionate aqueous nasal spray in healthy subjects has shown that ritonavir (a strong CYP3A4 inhibitor) can significantly increase plasma fluticasone propionate exposure, resulting in significantly reduced serum cortisol concentrations [see Clinical Pharmacology (12.3)]. During postmarketing use, there have been reports of clinically significant drug interactions in patients receiving fluticasone propionate products with ritonavir, resulting in systemic corticosteroid effects including Cushing’s syndrome and adrenal suppression.
Ketoconazole
Coadministration of orally inhaled fluticasone propionate (1000 mcg) and ketoconazole (200 mg once daily) resulted in a 1.9-fold increase in plasma fluticasone propionate exposure and a 45% decrease in plasma cortisol area under the curve (AUC), but had no effect on urinary excretion of cortisol.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Available data from published literature on the use of inhaled or nasal fluticasone propionate in pregnant women have not reported a clear association with adverse developmental outcomes. In animals, teratogenicity characteristic of corticosteroids, decreased fetal body weight, and/or skeletal variations in rats, mice, and rabbits were observed with subcutaneously administered maternal toxic doses of fluticasone propionate less than the maximum recommended human daily inhaled dose (MRHDID) on a mcg/m2 basis. However, fluticasone propionate administered via inhalation to rats decreased fetal body weight, but did not induce teratogenicity at a maternal toxic dose less than the MRHDID on a mcg/m2 basis (see Data). Experience with oral corticosteroids suggests that rodents are more prone to teratogenic effects from corticosteroids than humans.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated risk of major birth defects and miscarriages in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
In embryofetal development studies with pregnant rats and mice dosed by the subcutaneous route throughout the period of organogenesis, fluticasone propionate was teratogenic in both species. Omphalocele, decreased body weight, and skeletal variations were observed in rat fetuses, in the presence of maternal toxicity, at a dose approximately equivalent to the MRHDID (on a mcg/m2 basis with a maternal subcutaneous dose of 100 mcg/kg/day). The rat no observed adverse effect level (NOAEL) was observed at approximately 0.4 times the MRHDID (on a mcg/m2 basis with a maternal subcutaneous dose of 30 mcg/kg/day). Cleft palate and fetal skeletal variations were observed in mouse fetuses at a dose approximately 0.3 times the MRHDID (on a mcg/m2 basis with a maternal subcutaneous dose of 45 mcg/kg/day). The mouse NOAEL was observed with a dose approximately 0.1 times the MRHDID (on a mcg/m2 basis with a maternal subcutaneous dose of 15 mcg/kg/day).
In an embryofetal development study with pregnant rats dosed by the inhalation route throughout the period of organogenesis, fluticasone propionate produced decreased fetal body weights and skeletal variations, in the presence of maternal toxicity, at a dose approximately 0.34 times the MRHDID (on a mcg/m2 basis with a maternal inhalation dose of 25.7 mcg/kg/day); however, there was no evidence of teratogenicity. The NOAEL was observed with a dose approximately 0.1 times the MRHDID (on a mcg/m2 basis with a maternal inhalation dose of 5.5 mcg/kg/day).
In an embryofetal development study in pregnant rabbits that were dosed by the subcutaneous route throughout organogenesis, fluticasone propionate produced reductions of fetal body weights, in the presence of maternal toxicity, at doses approximately 0.02 times the MRHDID and higher (on a mcg/m2 basis with a maternal subcutaneous dose of 0.57 mcg/kg/day). Teratogenicity was evident based upon a finding of cleft palate for 1 fetus at a dose approximately 0.1 times the MRHDID (on a mcg/m2 basis with a maternal subcutaneous dose of 4 mcg/kg/day). The NOAEL was observed in rabbit fetuses with a dose approximately 0.002 times the MRHDID (on a mcg/m2 basis with a maternal subcutaneous dose of 0.08 mcg/kg/day).
Fluticasone propionate crossed the placenta following subcutaneous administration to mice and rats and oral administration to rabbits.
In a pre- and post-natal development study in pregnant rats dosed from late gestation through delivery and lactation (Gestation Day 17 to Postpartum Day 22), fluticasone propionate was not associated with decreases in pup body weight, and had no effects on developmental landmarks, learning, memory, reflexes, or fertility at doses up to 0.7 times the MRHDID (on a mcg/m2 basis with maternal subcutaneous doses up to 50 mcg/kg/day).
8.2 Lactation
Risk Summary
There are no available data on the presence of fluticasone propionate in human milk, the effects on the breastfed child, or the effects on milk production. Fluticasone propionate is present in rat milk (see Data). Other corticosteroids have been detected in human milk. However, fluticasone propionate concentrations in plasma after orally inhaled therapeutic doses are low, and therefore, concentrations in human breast milk are likely to be correspondingly low [see Clinical Pharmacology (12.3)]. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for XHANCE and any potential adverse effects on the breastfed child from XHANCE or from the underlying maternal condition.
8.4 Pediatric Use
The safety and effectiveness of XHANCE in pediatric patients have not been established.
Effects on Growth
Controlled clinical trials have shown that nasal corticosteroids may cause a reduction in growth velocity when administered to pediatric patients. This effect was observed in the absence of laboratory evidence of HPA axis suppression, suggesting that growth velocity is a more sensitive indicator of systemic corticosteroid exposure in pediatric patients than some commonly used tests of HPA axis function. The long-term effects of this reduction in growth velocity associated with nasal corticosteroids, including the impact on final adult height, are unknown. The potential for “catch-up” growth following discontinuation of treatment with nasal corticosteroids has not been adequately studied. The growth of pediatric patients receiving nasal corticosteroids should be monitored routinely (e.g., via stadiometry). The potential growth effects of prolonged treatment should be weighed against the clinical benefits obtained and the risks associated with alternative therapies.
Controlled clinical trials have shown that corticosteroids orally inhaled into the lungs may cause a reduction in growth in pediatric patients. In these trials, the mean reduction in growth velocity was approximately 1 cm/year (range: 0.3 to 1.8 cm/year) and appeared to depend upon dose and duration of exposure. The effects on growth velocity of treatment with corticosteroids orally inhaled into the lungs for over 1 year, including the impact on final adult height, are unknown. The growth of children and adolescents receiving corticosteroids should be monitored routinely (e.g., via stadiometry) [see Warnings and Precautions (5.8)].
8.5 Geriatric Use
Clinical trials of XHANCE for Chronic Rhinosinusitis with Nasal Polyps did not include sufficient numbers of patients aged 65 years and older to determine whether they responded differently than younger patients. Other reported clinical experience with fluticasone administered nasally or orally inhaled has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
There were 82 patients 65 years of age and older in clinical trials for Chronic Rhinosinusitis without Nasal Polyps [see Clinical Studies (14)]. Of the total number of XHANCE-treated patients in these studies, 51 (14%) were 65 years of age and older. No overall differences in safety and effectiveness of XHANCE have been observed between patients 65 years of age and older and younger adult patients.
8.6 Hepatic Impairment
Formal pharmacokinetic trials using XHANCE have not been conducted in subjects with hepatic impairment. Since fluticasone propionate is predominantly cleared by hepatic metabolism, impairment of liver function may lead to accumulation of fluticasone propionate in plasma. Therefore, patients with hepatic disease should be closely monitored.
- 10 OVERDOSAGE
-
11 DESCRIPTION
The active component of XHANCE is fluticasone propionate, a corticosteroid, having the chemical name S-(fluoromethyl) 6α,9-difluoro-11β,17-dihydroxy-16α-methyl-3oxoandrosta-1,4-diene-17β-carbothioate, 17‑propionate and the following chemical structure:
Fluticasone propionate is a white powder with a molecular weight of 500.57, and the empirical formula is C25H31F3O5S. It is practically insoluble in water, freely soluble in dimethylformamide, sparingly soluble in acetone and dichloromethane, and slightly soluble in 96% ethanol.
XHANCE (fluticasone propionate) nasal spray, 93 mcg, for nasal administration, with an Optinose exhalation delivery system that delivers an aqueous suspension of microfine fluticasone propionate having a particle size distribution in the range of 0 to 5 microns for topical nasal administration by means of a metering, atomizing spray pump and exhaled breath. XHANCE also contains microcrystalline cellulose and carboxymethylcellulose sodium, dextrose, benzalkonium chloride, polysorbate 80, edetate disodium dihydrate, sodium hydroxide and hydrochloric acid (to adjust pH), and purified water, and has a pH between 5 and 7.
Before initial use, prime XHANCE by gently shaking and then pressing the amber glass bottle 7 times or until a fine mist appears. Once primed, XHANCE contains 120 metered sprays. When XHANCE has not been used for ≥ 7 days, prime again by releasing 2 sprays into the air, away from the face [see Dosage and Administration (2.2) and Instructions for Use].
After priming, each spray delivers 93 mcg of fluticasone propionate in an aqueous suspension through the cone-shaped nosepiece. The system also has a flexible mouthpiece. Within the device is a non‑removable amber glass bottle with a metering spray pump, an applicator, and a valve that prevents release of breath until the bottle is pressed. A base covers the bottom of the bottle, and a removable orange cap covers both the nosepiece and mouthpiece.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Fluticasone propionate is a synthetic trifluorinated corticosteroid with anti-inflammatory activity. Fluticasone propionate has been shown in vitro to exhibit a binding affinity for the human glucocorticoid receptor that is 18 times that of dexamethasone, almost twice that of beclomethasone-17-monopropionate (BMP), the active metabolite of beclomethasone dipropionate, and over 3 times that of budesonide. Data from the McKenzie vasoconstrictor assay in man are consistent with these results. The clinical significance of these findings is unknown.
The precise mechanism through which fluticasone propionate affects CRSsNP and CRSwNP and associated inflammatory symptoms is not known. Corticosteroids have been shown to have a wide range of effects on multiple cell types (e.g., mast cells, eosinophils, neutrophils, macrophages, lymphocytes) and mediators (e.g., histamine, eicosanoids, leukotrienes, cytokines) involved in inflammation. The anti-inflammatory action of corticosteroids contributes to their efficacy. In 7 trials in adults, fluticasone propionate nasal spray decreased nasal mucosal eosinophils in 66% of patients (35% for placebo) and basophils in 39% of patients (28% for placebo). In addition, studies suggest that carbon dioxide, which is present in the exhaled breath delivered into the nose through the device, may influence inflammatory mediator activity and neuropeptide activity, possibly through mechanisms of action that also include removal of nitric oxide, change in pH, or positive pressure. The direct relationship of these findings to long-term symptom relief is not known.
12.2 Pharmacodynamics
HPA Axis Effect
The potential systemic effects of XHANCE on the HPA axis have not been evaluated.
Serum cortisol concentrations, urinary excretion of cortisol, and urine 6-β- hydroxycortisol excretion collected over 24 hours in 24 healthy subjects following 8 oral inhalations of fluticasone propionate 44, 110, and 220 mcg decreased with increasing dose. However, in patients with asthma treated with 2 oral inhalations of fluticasone propionate 44, 110, and 220 mcg twice daily for at least 4 weeks, differences in serum cortisol AUC0-12h (n = 65) and 24-hour urinary excretion of cortisol (n = 47) compared with placebo were not related to dose and generally not significant.
The potential systemic effects of orally inhaled fluticasone propionate on the HPA axis were also studied in subjects with asthma [see Warnings and Precautions (5.5) and Adverse Reactions (6)]. Fluticasone propionate given by oral inhalation aerosol at dosages of 440 or 880 mcg twice daily was compared with placebo in oral corticosteroid‑dependent subjects with asthma (range of mean dose of prednisone at baseline: 13 to 14 mg/day) in a 16-week trial. Consistent with maintenance treatment with oral corticosteroids, abnormal plasma cortisol responses to short cosyntropin stimulation (peak plasma cortisol less than 18 mcg/dL) were present at baseline in the majority of subjects participating in this trial (69% of subjects later randomized to placebo and 72% to 78% of subjects later randomized to fluticasone propionate HFA). At week 16, 8 subjects (73%) on placebo compared with 14 (54%) and 13 (68%) subjects receiving fluticasone propionate HFA (440 and 880 mcg twice daily, respectively) had poststimulation cortisol levels of less than 18 mcg/dL.
Cardiac Electrophysiology
A study specifically designed to evaluate the effect of XHANCE on the QT interval has not been conducted.
12.3 Pharmacokinetics
The activity of XHANCE is due to the parent drug, fluticasone propionate. Due to the low bioavailability by the nasal route, the majority of the pharmacokinetic data were obtained via other routes of administration.
Absorption
The mean (SD) peak exposure (Cmax) and total exposure (AUC0-∞) following administration of a dose of 186 mcg of XHANCE during exhalation were 17.2 ± 7.40 pg/mL and 111.7 ± 49.75 pg·h/mL, respectively, and were 25.3 ± 10.34 pg/mL and 171.7 ± 85.55 pg·h/mL, respectively, following a dose of 372 mcg of XHANCE in healthy subjects. The Cmax and AUC0-∞ following a dose of 372 mcg of XHANCE in patients with mild to moderate asthma were 28.7±18.72 pg/mL and 222.6±84.60 pg·h/mL, respectively.
Distribution
Following intravenous administration, the initial disposition phase for fluticasone propionate was rapid and consistent with its high lipid solubility and tissue binding. The volume of distribution averaged 4.2 L/kg.
The percentage of fluticasone propionate bound to human plasma proteins averaged 99%. Fluticasone propionate is weakly and reversibly bound to erythrocytes and is not significantly bound to human transcortin.
Local exposure within the nasal cavity with XHANCE will differ when used without exhalation through the device.
Elimination
Following intravenous dosing, fluticasone propionate showed polyexponential kinetics and had a terminal elimination half-life of approximately 7.8 hours. The total blood clearance of fluticasone propionate is high (average: 1093 mL/min), with renal clearance accounting for less than 0.02% of the total.
Metabolism: The only circulating metabolite detected in man is the 17β-carboxylic acid derivative of fluticasone propionate, which is formed through the CYP3A4 pathway. This metabolite had less affinity (approximately 1/2000) than the parent drug for the glucocorticoid receptor of human lung cytosol in vitro and negligible pharmacological activity in animal studies. Other metabolites detected in vitro using cultured human hepatoma cells have not been detected in man.
Excretion: Less than 5% of a radiolabeled oral dose was excreted in the urine as metabolites, with the remainder excreted in the feces as parent drug and metabolites.
Specific Populations
XHANCE was not studied in any special populations, and no gender-specific pharmacokinetic data have been obtained.
Pediatric Patients: XHANCE was not studied in pediatric patients, and no pediatric-specific pharmacokinetic data have been obtained with the product.
Patients with Hepatic and Renal Impairment: Formal pharmacokinetic studies using XHANCE have not been conducted in patients with hepatic or renal impairment. However, since fluticasone propionate is predominantly cleared by hepatic metabolism, impairment of liver function may lead to accumulation of fluticasone propionate in plasma. Therefore, patients with hepatic disease should be closely monitored.
Racial or Ethnic Groups: No significant difference in clearance (CL/F) of fluticasone propionate in Caucasian, African-American, Asian, or Hispanic populations has been observed.
Drug Interaction Studies
Inhibitors of Cytochrome P450 3A4
Ritonavir: Fluticasone propionate is a substrate of CYP3A4. Coadministration of fluticasone propionate and the strong CYP3A4 inhibitor, ritonavir, is not recommended based upon a multiple-dose, crossover drug interaction trial in 18 healthy subjects. Fluticasone propionate aqueous nasal spray (200 mcg once daily) was coadministered for 7 days with ritonavir (100 mg twice daily). Plasma fluticasone propionate concentrations following fluticasone propionate aqueous nasal spray alone were undetectable (< 10 pg/mL) in most subjects, and when concentrations were detectable, peak levels (Cmax) averaged 11.9 pg/mL (range: 10.8 to 14.1 pg/mL) and AUC0-τ averaged 8.43 pg·h/mL (range: 4.2 to 18.8 pg·h/mL). Fluticasone propionate Cmax and AUC0-τ increased to 318 pg/mL (range: 110 to 648 pg/mL) and 3102.6 pg·h/mL (range: 1207.1 to 5662.0 pg·h/mL), respectively, after coadministration of ritonavir with fluticasone propionate aqueous nasal spray. This significant increase in plasma fluticasone propionate exposure resulted in a significant decrease (86%) in serum cortisol AUC.
Ketoconazole: Coadministration of fluticasone propionate orally inhaled into the lungs (1000 mcg) and ketoconazole (200 mg once daily) resulted in a 1.9-fold increase in plasma fluticasone propionate exposure and a 45% decrease in plasma cortisol AUC, but had no effect on urinary excretion of cortisol.
Following orally-inhaled fluticasone propionate alone, AUC2-last averaged 1559 pg·h/mL (range: 555 to 2906 pg·h/mL) and AUC2-∞ averaged 2269 pg·h/mL (range: 836 to 3707 pg·h/mL). Fluticasone propionate AUC2-last and AUC2-∞ increased to 2781 pg·h/mL (range: 2489 to 8486 pg·h/mL) and 4317 pg·h/mL (range: 3256 to 9408 pg·h/mL), respectively, after coadministration of ketoconazole with orally-inhaled fluticasone propionate. This increase in plasma fluticasone propionate concentration resulted in a decrease (45%) in serum cortisol AUC.
Erythromycin: In a multiple-dose drug interaction study, coadministration of orally inhaled fluticasone propionate (500 mcg twice daily) and erythromycin (333 mg 3 times daily) did not affect fluticasone propionate pharmacokinetics.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Fluticasone propionate demonstrated no tumorigenic potential in mice at oral doses up to 1000 mcg/kg (approximately 7 times the MRHDID for adults on a mcg/m2 basis) for 78 weeks or in rats at inhalation doses up to 57 mcg/kg (approximately equivalent to the MRHDID for adults on a mcg/m2 basis) for 104 weeks.
Fluticasone propionate did not induce gene mutation in prokaryotic or eukaryotic cells in vitro. No significant clastogenic effect was seen in cultured human peripheral lymphocytes in vitro or in the in vivo mouse micronucleus test.
Fertility and reproductive performance were unaffected in male and female rats at subcutaneous doses up to 50 mcg/kg (approximately 0.7 times the MRHDID for adults on a mcg/m2 basis).
-
14 CLINICAL STUDIES
14.1 Chronic Rhinosinusitis with Nasal Polyps
The efficacy of XHANCE was evaluated in two randomized, double-blind, parallel‑group, multicenter, placebo-controlled, dose-ranging trials in adults 18 years and older with Chronic Rhinosinusitis with Nasal Polyps (CRSwNP) and associated moderate to severe nasal congestion (Trial 1 [NCT01622569], Trial 2 [NCT01624662]). The two trials included a total of 646 patients [348 (53.9%) males and 298 (46.1%) females] with a mean age of 45.5 years. Patients were randomized 1:1:1:1 to receive 93 mcg, 186 mcg, or 372 mcg twice daily or placebo for a period of 16 weeks. At baseline 35.7%, 79.0%, and 18.3% had polyps graded as mild, moderate, or severe, respectively. In addition, 90.6% of patients reported previous use of a topical steroid nasal spray for the treatment of CRSwNP and 53.6% reported previous sinus surgery or polypectomy.
The co-primary efficacy endpoints were 1) change from baseline to Week 4 in nasal congestion / obstruction averaged over the preceding 7 days of treatment and 2) change from baseline to Week 16 in bilateral polyp grade. Nasal congestion was rated by the patient on a 0 to 3 categorical severity scale (0 = none, 1 = mild, 2 = moderate, 3 = severe) at the time immediately prior to the next dose (instantaneous). Polyp grade was determined by the clinician using nasal endoscopy. Polyps on each side of the nose were graded on a categorical scale (0 = No polyps; 1 = Mild – polyps not reaching below the inferior border of the middle turbinate; 2 = Moderate – polyps reaching below the inferior border of the middle concha, but not the inferior border of the inferior turbinate; 3 = Severe – large polyps reaching below the lower inferior border of the inferior turbinate).
Efficacy was demonstrated for both XHANCE 186-mcg twice daily and XHANCE 372-mcg twice daily (Table 3).
Table 3: Effect of XHANCE nasal spray in two randomized, placebo-controlled trials in patients with CRSwNP. bid= twice daily Placebo
XHANCE
186-mcg
bidXHANCE
372-mcg
bidDiff. (95% CI)
XHANCE
186-mcg bid
vs placeboDiff. (95% CI)
XHANCE
372-mcg bid
vs placeboTrial 1 (N)
82
80
79
Baseline nasal congestion
2.31
2.24
2.29
LS mean change from baseline in nasal congestion at week 4
-0.24
-0.54
-0.62
-0.30
(-0.48, -0.11)-0.38
(-0.57, -0.19)Baseline total bilateral polyp grade
3.8
3.9
3.7
LS mean change from baseline in total bilateral polyp grade at week 16
-0.45
-1.03
-1.06
-0.59
(-0.93, -0.24)-0.62
(-0.96, -0.27)Trial 2 (N)
79
80
82
Baseline nasal congestion
2.29
2.20
2.25
LS mean change from baseline in nasal congestion at week 4
-0.24
-0.68
-0.62
-0.45
(-0.64, -0.25)-0.38
(-0.58, -0.18)Baseline total bilateral polyp grade
3.8
3.9
3.9
LS mean change from baseline in total bilateral polyp grade at week 16
-0.61
-1.22
-1.41
-0.60
(-0.89, -0.31)-0.80
(-1.08, -0.51)There were no clinically relevant differences in effectiveness of XHANCE across subgroups of patients defined by gender, age, or race.
Onset of action, evaluated by determining the starting period that the treatment effect of XHANCE on daily instantaneous AM congestion score started to achieve statistical significance in comparison to placebo and roughly maintained thereafter, was generally observed within 2 weeks for both XHANCE doses.
14.2 Chronic Rhinosinusitis without Nasal Polyps
The efficacy of XHANCE for Chronic Rhinosinusitis without Nasal Polyps (CRSsNP) was evaluated in two 24-week randomized, double-blind, parallel-group, multicenter, placebo-controlled trials in 555 adults 18 years and older (Trial 3 [NCT03960580] and Trial 4 [NCT03781804]). Trial 3 included 223 patients with CRSsNP and Trial 4 included 332 patients with either CRSsNP (N=124) or CRSwNP (N=208). While Trial 4 included CRSwNP patients, efficacy results from Trials 3 and 4 are presented for CRSsNP patients only.
In both trials, patients were randomized 1:1:1 to receive XHANCE 186 mcg twice daily, XHANCE 372 mcg twice daily, or placebo, all administered nasally for 24 weeks. All patients enrolled in Trial 3 and Trial 4 had at least 2 active nasal symptoms (congestion/obstruction, discharge, facial pain or pressure, reduction or loss of smell) with a minimum nasal congestion score ≥1.5 out of 3 and a baseline CT scan showing ≥25% opacification of both ethmoid and at least 1 maxillary sinus. Refer to Table 4 for the demographic and baseline characteristics of all randomized CRSsNP patients in Trials 3 and 4.
Table 4: Demographics and Baseline Characteristics of Trials 3 & 4 1SD = standard deviation Trial 3
(N=223)Trial 4
(N=124)Mean age (years) (SD)
48 (13)
50 (14)
Male, n (%)
112 (50)
64 (52)
Race, n (%)
White
220 (99)
114 (92)
Asian
0
6 (5)
Black or African American
2 (1)
4 (3)
Other
1 (<1)
0
Number (%) of patients with a history of environmental allergies
69 (31)
58 (47)
Number (%) of patients using standard-delivery nasal steroids at study entry
57 (26)
64 (52)
Mean number (SD) of acute sinusitis exacerbations where subject received an antibiotic or oral steroids in last year
1.1 (1.4)
1.8 (1.6)
In both trials, the coprimary efficacy endpoints were 1) change from baseline at Week 4 in the composite symptom score (CSS) as determined by patients using a daily diary and 2) change from baseline at Week 24 in percent opacified sinus volume. CSS was the sum of the individual nasal symptom scores for congestion/obstruction, facial pain/pressure, and nasal discharge, each rated by the patient on a 0 to 3 categorical severity scale (0 = none, 1 = mild, 2 = moderate, 3 = severe) in the morning immediately prior to the next dose. Sinus opacification was measured by CT scan and scored using a 3-dimensional computer-assisted volumetric assessment using software to quantify the average percent of opacified volume in the ethmoid and maxillary sinuses.
Efficacy was demonstrated for both coprimary endpoints (CSS and percent opacified sinus volume) for XHANCE 186 mcg twice daily and XHANCE 372 mcg twice daily. Refer to Table 5 for coprimary endpoint results. Improvements demonstrated in CSS at Week 4 with either dose of XHANCE remained at Weeks 8 and 12.
Table 5: LS Mean Change in Composite Symptom Scores (CSS)a at Week 4 and Percent Opacified Sinus Volume at Week 24 in Patients with CRSsNP in Trials 3 and 4 Results were based on all patients who received at least one dose of study drug and had baseline measurements for coprimary endpoints, and at least one post-baseline CSS on or before Week 4.
aCSS range was 0 – 9. Scores were averaged over 7 days before a visit.
bData presented for Trial 4 patients with CRSsNP only
LS= Least Square; BID= twice daily; CI = Confidence IntervalPlacebo
XHANCE
186-mcg
BIDXHANCE
372-mcg
BIDDiff. (95% CI)
XHANCE
186-mcg BID
vs placeboDiff. (95% CI)
XHANCE
372-mcg BID
vs placeboTrial 3 (N)
75
72
73
Baseline Mean CSSa
6.2
5.9
6.0
LS mean change from baseline in CSS at week 4
-0.8
-1.5
-1.7
-0.7
(-1.3, -0.2)-0.9
(-1.5, -0.4)Baseline Percent of Opacified Sinus Volume
64.1
60.5
61.5
LS mean change from baseline in Percent Opacified Sinus Volume at week 24
0.4
-7.0
-5.5
-7.5
(-12.1, -2.8)-5.9
(-10.6, -1.3)Trial 4 (N)b
41
41
40
Baseline Mean CSS
5.7
5.5
5.8
LS mean change from baseline in CSS at week 4
-0.7
-1.6
-1.6
-0.9
(-1.6, -0.2)-0.9
(-1.6, -0.2)Baseline Percent of Opacified Sinus Volume
61.9
63.0
60.7
LS mean change from baseline in Percent Opacified Sinus Volume at week 24
-5.3
-5.7
-8.4
-0.5
(-6.8, 5.9)-3.2
(-9.5, 3.2)Secondary efficacy endpoints included change from baseline in individual symptoms of the CSS (nasal congestion, nasal pain/pressure, and nasal discharge) at Week 4. Refer to Table 6 for individual symptom score results.
Table 6: LS Mean Change in Individual Symptom Scoresa at Week 4 in Patients with CRSsNP in Trials 3 and 4 Individual symptom scores were not prespecified to adjust for multiplicity.
aScore range was 0-3.
bData presented for Trial 4 patients with CRSsNP only
LS= Least Square; BID= twice daily; CI = Confidence IntervalPlacebo
XHANCE
186-mcg
BIDXHANCE
372-mcg
BIDDiff. (95% CI)
XHANCE
186-mcg BID
vs placeboDiff. (95% CI)
XHANCE
372-mcg BID
vs placeboMean Baseline
LS Mean Change
Mean Baseline
LS Mean Change
Mean Baseline
LS Mean Change
Trial 3 (N)
75
72
73
Nasal Congestion/
Obstruction2.3
-0.3
2.1
-0.6
2.2
-0.7
-0.3
(-0.5, -0.1)-0.4
(-0.7, -0.2)Facial Pain/Pressure
1.8
-0.3
1.7
-0.4
1.7
-0.5
-0.2
(-0.4, 0.1)-0.2
(-0.5, -0.0)Nasal Discharge
2.1
-0.3
2.1
-0.5
2.0
-0.6
-0.2
(-0.4, -0.0)-0.3
(-0.5, -0.1)Trial 4 (N)b
41
41
40
Nasal Congestion/
Obstruction2.2
-0.3
2.1
-0.6
2.2
-0.6
-0.3
(-0.6, -0.1)-0.3
(-0.6, -0.0)Facial Pain/Pressure
1.7
-0.2
1.6
-0.4
1.7
-0.4
-0.2
(-0.5, 0.0)-0.2
(-0.5, 0.0)Nasal Discharge
1.9
-0.2
1.8
-0.6
1.9
-0.6
-0.4
(-0.6, -0.1)-0.4
(-0.6, -0.1)Acute Exacerbations
Acute exacerbations of chronic rhinosinusitis (AECRS), defined as a worsening of symptoms that required escalation of treatment (e.g., antibiotics, oral steroids, acute care visits), was assessed in the CRSsNP patients from pooled data (Trials 3 and 4) through Week 24. The rate of AECRS was reduced by 53% among patients in each XHANCE treatment group versus placebo. This was derived from incidence rate ratios of 0.47 (95% CI: 0.21, 1.08) and 0.47 (95% CI: 0.20, 1.09) among patients using XHANCE 186 mcg and 372 mcg twice daily versus placebo, respectively; which were not statistically significant.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
XHANCE (fluticasone propionate) nasal spray is supplied as a non-removable amber glass bottle fitted with a metered-dose manual spray pump unit inside the white XHANCE device with a nasal applicator, valve mechanism, asymmetrical cone-shaped nosepiece, flexible mouthpiece, base, and orange cap in a box of 1 (NDC 71143-375-01) with FDA‑approved Patient Labeling [see Instructions for Use].
Each bottle contains a net fill content of 16 mL, and after priming will provide 120 metered sprays. Each metered spray delivers 93 mcg of fluticasone propionate in an aqueous suspension through the cone‑shaped nosepiece. The correct amount of medication in each metered spray cannot be assured after 120 metered sprays even though the bottle is not completely empty. The bottle should be discarded when the labeled number of metered sprays has been used.
Store at room temperature (between 15°C and 25°C; 59°F and 77°F), excursions permitted from 15°C to 30°C (59°F to 86°F). Avoid exposure to extreme heat, cold or light. Shake XHANCE before each use.
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
Local Nasal Adverse Reactions
Inform patients that treatment with XHANCE may lead to adverse reactions, which include epistaxis, nasal erosions, and nasal ulceration. Candida infection may also occur with treatment with XHANCE. In addition, XHANCE has been associated with nasal septal perforation and impaired wound healing. Patients who have experienced recent nasal ulcerations, nasal surgery, or nasal trauma should not use XHANCE until healing has occurred [see Warnings and Precautions (5.1)].
Glaucoma and Cataracts
Inform patients that glaucoma and cataracts are associated with long-term use of nasal and orally inhaled corticosteroids, including fluticasone propionate, and may increase the risk of some eye problems. Consider regular eye exams. Advise patients to notify their healthcare providers if a change in vision is noted while using XHANCE [see Warnings and Precautions (5.2)].
Hypersensitivity Reactions, Including Anaphylaxis
Inform patients that hypersensitivity reactions, including anaphylaxis, angioedema, urticaria, contact dermatitis, rash, bronchospasm, and hypotension, may occur after administration of fluticasone. If such reactions occur during use with XHANCE, patients should discontinue use of the product [see Warnings and Precautions (5.3)].
Immunosuppression and Risk of Infection
Warn patients who are on immunosuppressant doses of corticosteroids to avoid exposure to chickenpox or measles, and if they are exposed to consult their healthcare provider without delay. Inform patients of potential worsening of existing tuberculosis; fungal, bacterial, viral, or parasitic infections; or ocular herpes simplex [see Warnings and Precautions (5.4)].
Hypercorticism and Adrenal Suppression
Advise patients that XHANCE may cause systemic corticosteroid effects of hypercorticism and adrenal suppression. Additionally, inform patients that deaths due to adrenal insufficiency have occurred during and after transfer from systemic corticosteroids. Patients should taper slowly from systemic corticosteroids if transferring to XHANCE [see Warnings and Precautions (5.5)].
Reduction in Bone Mineral Density
Advise patients who are at an increased risk for decreased bone mineral density that the use of corticosteroids may pose an additional risk [see Warnings and Precautions (5.7)].
Reduced Growth Velocity
The safety and effectiveness of XHANCE use in pediatric patients has not been established. Inform patients that corticosteroids administered by oral inhalation into the lungs or nasally may cause a reduction in growth velocity when administered to pediatric patients [see Warnings and Precautions (5.8)].
Use Twice Daily for Best Effect
Inform patients that they should use XHANCE on a regular basis as directed. XHANCE, like other corticosteroids, does not have an immediate effect on CRSsNP or CRSwNP or symptoms. Individual patients will experience a variable time to onset and degree of symptom relief and the full benefit may not be achieved until treatment has been administered for up to 16 weeks or longer. Maximum benefit may not be reached for a period of months. Patients should not increase the prescribed dosage, but should contact their healthcare providers if symptoms do not improve or if the condition worsens.
If a patient missed a dose, the patient should be advised to take the dose as soon as they remember. The patient should not take more than the recommended dose for the day.
Keep Spray Out of Eyes and Mouth
Inform patients to avoid spraying XHANCE in their eyes and mouth.
How to Use XHANCE
It is important for patients to understand how to correctly administer XHANCE nasal spray using the exhalation delivery system. Advise the patient to carefully read the patient Instructions for Use. Any questions regarding use that the patient has should be directed to the physician or pharmacist.
Advise the patient to shake before each use.
The patient should note the difference in appearance of the cone-shaped, non-flexible nosepiece and the longer flexible mouthpiece.
The patient should be instructed to gently insert the tapered tip of the cone-shaped nosepiece deeply into the nose in order to gently expand the nasal passage and to create a tight seal between the nosepiece and the nostril. A seal must be maintained as the patient blows into the mouthpiece and actuates the spray pump.
To actuate the device, patients should be advised to push the bottle up while continuing to blow forcefully into the mouthpiece. Within the device is a valve that prevents the release of breath until the bottle is pushed. Pushing the bottle also actuates the spray pump, releasing a metered dose of aerosolized medication while simultaneously allowing a “burst” of exhaled breath to pass through the device. This helps deliver the medication deep into the patient’s nose.
Patients should be advised not to try to inhale (e.g., “sniff”) when blowing (exhaling) into the mouthpiece.
Patients should be advised not to block the other nostril because the exhaled breath must pass around the back of the nasal septum and out the other side of the nose.
Distributed by:
OptiNose US, Inc.
1020 Stony Hill Rd
Yardley, PA 19067XHANCE and OPTINOSE are registered trademarks of OptiNose, Inc.
©2024, OptiNose US, Inc. All rights reserved.
- INSTRUCTIONS FOR USE - 16 mL
-
PATIENT INFORMATION
PATIENT INFORMATION
XHANCE® (ex hans)
(fluticasone propionate)
nasal spray, 93 mcg
What is XHANCE?
XHANCE is a prescription medicine used to treat:
- •
- chronic rhinosinusitis with nasal polyps in adults.
- •
- chronic rhinosinusitis without nasal polyps in adults.
It is not known if XHANCE is safe and effective in children.
Do not use XHANCE if you are allergic to fluticasone propionate or any of the ingredients in XHANCE. See the end of this Patient Information leaflet for a complete list of ingredients in XHANCE.
Before using XHANCE, tell your healthcare provider about all of your medical conditions, including if you:
- •
- have or have had nasal sores, nasal surgery, or nasal injury.
- •
- have eye problems, such as cataracts or glaucoma.
- •
- have an immune system problem.
- •
- have any type of viral, bacterial, or fungal infection.
- •
- are exposed to chickenpox or measles.
- •
- have weak bones (osteoporosis).
- •
- have liver problems.
- •
- are pregnant or plan to become pregnant. It is not known if XHANCE may harm your unborn baby.
- •
- are breastfeeding or plan to breastfeed. It is not known if XHANCE passes into your breast milk and if it can harm your baby.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
XHANCE and certain other medicines may interact with each other. This may cause serious side effects.
Especially tell your healthcare provider if you take antifungal or anti-HIV medicines.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine.
How should I use XHANCE?
Read the Instructions for Use leaflet for information about the right way to use XHANCE.
- •
- Use XHANCE exactly as your healthcare provider tells you.
- •
- Do not use XHANCE more often than prescribed. Ask your healthcare provider if you have any questions.
- •
- XHANCE is for use in your nose only. Do not spray it in your eyes or mouth.
- •
- XHANCE may take several days of regular use for your symptoms to get better. It may take several months for the medicine to have its greatest effect. If your symptoms do not improve or get worse, call your healthcare provider.
- •
- You will get the best results if you keep using XHANCE regularly twice each day without missing a dose. Do not stop using XHANCE unless your healthcare provider tells you to do so.
- •
- If you miss a dose of XHANCE, take it as soon as you remember the same day. Do not take more than your prescribed dose of XHANCE each day.
What are the possible side effects of XHANCE?
XHANCE may cause serious side effects, including:
- •
-
Nasal problems. Symptoms of nasal problems may include:
- o
- nose bleeds.
- o
- crusting in the nose.
- o
- sores (ulcers) in the nose.
- o
- hole in the cartilage of the nose (nasal septal perforation). A whistling sound when you breathe may be a symptom of nasal septum perforation.
- o
- thrush (candida), a fungal infection in your nose and throat. Tell your healthcare provider if you have any redness or white colored patches in your nose or mouth.
- o
- slow wound healing. You should not use XHANCE until your nose has healed if you have a sore in your nose, have had surgery on your nose, or if your nose has been injured.
- •
- Eye problems including glaucoma and cataracts. You should have regular eye exams while you use XHANCE. Call your healthcare provider if you have vision changes while using XHANCE.
- •
- Serious allergic reactions. Call your healthcare provider or get emergency medical care if you get any of the following signs of a serious allergic reaction:
-
- o
- rash
- o
- hives
- o
- swelling of your face, mouth, and tongue
-
- o
- breathing problems
- o
- low blood pressure
- •
- Weakened immune system and increased chance of getting infections (immunosuppression). Taking medicines that weaken your immune system makes you more likely to get infections and can make certain infections worse. These infections may include tuberculosis (TB), herpes simplex infections of the eyes (ocular herpes simplex infections), and infections caused by fungi, bacteria, viruses, and parasites. Avoid contact with people who have a contagious disease such as chickenpox or measles while using XHANCE. If you come in contact with someone who has chickenpox or measles, call your healthcare provider right away. Symptoms of an infection may include:
-
- o
- fever
- o
- pain
- o
- aches
- o
- chills
-
- o
- feeling tired
- o
- nausea
- o
- vomiting
- •
- Reduced adrenal function (adrenal insufficiency). Reduced adrenal function happens when your adrenal glands do not make enough steroid hormones. This can happen when you stop taking oral corticosteroid medicines (such as prednisone) and start taking medicine containing a steroid inhaled into the lungs or for use in the nose. Adrenal insufficiency can also happen when nasal corticosteroids, such as XHANCE, are used at a dose higher than the usual dose or in people who are likely to have adrenal insufficiency at the usual dose. This may be more likely to happen after surgery or during periods of stress. Symptoms of reduced adrenal function may include:
-
- o
- tiredness
- o
- weakness
-
- o
- nausea and vomiting
- o
- low blood pressure
- •
- Weak bones (osteoporosis).
- •
- Slowed growth in children. A child’s growth should be checked often.
The most common side effects of XHANCE in adults with chronic rhinosinusitis with nasal polyps include:
-
- •
- nose bleeds
- •
- sores (ulcers) in your nose
- •
- pain or swelling of your nose or throat (nasopharyngitis)
-
- •
- redness in your nose
- •
- nasal congestion
- •
- sinus infection
- •
- headache
The most common side effects of XHANCE in adults with chronic rhinosinusitis without nasal polyps include:
-
- •
- nose bleeds
- •
- headache
- •
- pain or swelling of your nose or throat (nasopharyngitis)
These are not all the possible side effects of XHANCE.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store XHANCE?
- •
- Store XHANCE at room temperature between 59°F and 77°F (15°C and 25°C).
- •
- Protect XHANCE from extreme heat, cold and light.
- •
- Throw away XHANCE after using 120 sprays after initial priming. Even though the bottle may not be completely empty, you may not get the correct dose of medicine if you continue to use it.
- •
- The XHANCE Bottle is made of glass. Do not remove the base from the bottle.
Keep XHANCE and all medicines out of the reach of children.
General information about the safe and effective use of XHANCE.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use XHANCE for a condition for which it was not prescribed. Do not give your XHANCE to other people, even if they have the same symptoms that you have. It may harm them.
You can ask your pharmacist or healthcare provider for information about XHANCE that is written for health professionals.
What are the ingredients in XHANCE?
Active ingredient: fluticasone propionate
Inactive ingredients: microcrystalline cellulose, carboxymethylcellulose sodium, dextrose, benzalkonium chloride, polysorbate 80, edetate disodium dihydrate, sodium hydroxide and hydrochloric acid (to adjust pH), and purified water
Distributed by: OptiNose US, Inc.1020 Stony Hill Rd, Yardley, PA 19067
XHANCE and OPTINOSE are registered trademarks of OptiNose, Inc.
©2024, OptiNose US, Inc. All rights reserved.
Patented. See www.xhance.com/patents
For more information, go to www.XHANCE.com or call 1-833-678-6673.
This Patient Information has been approved by the U.S. Food and Drug Administration
Revised: 03/2024
-
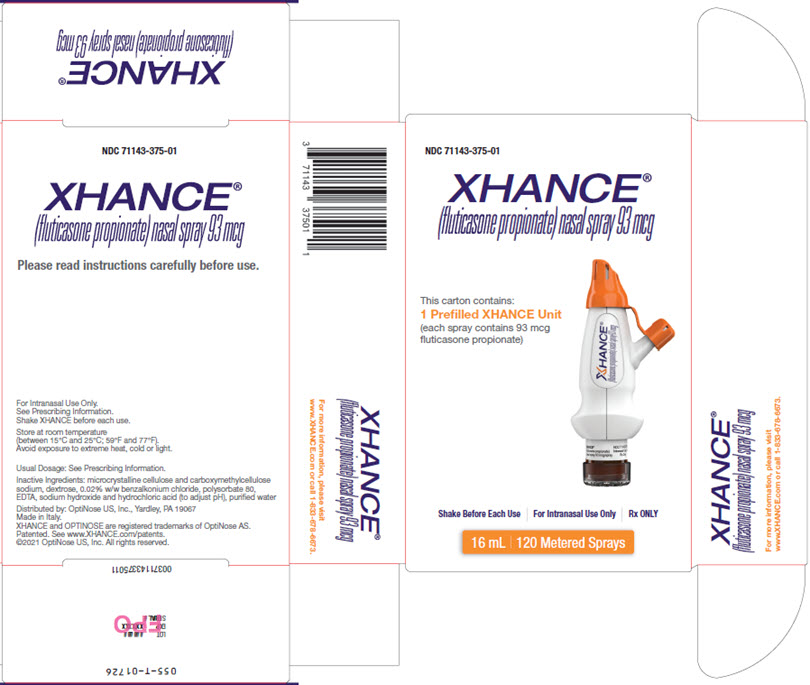
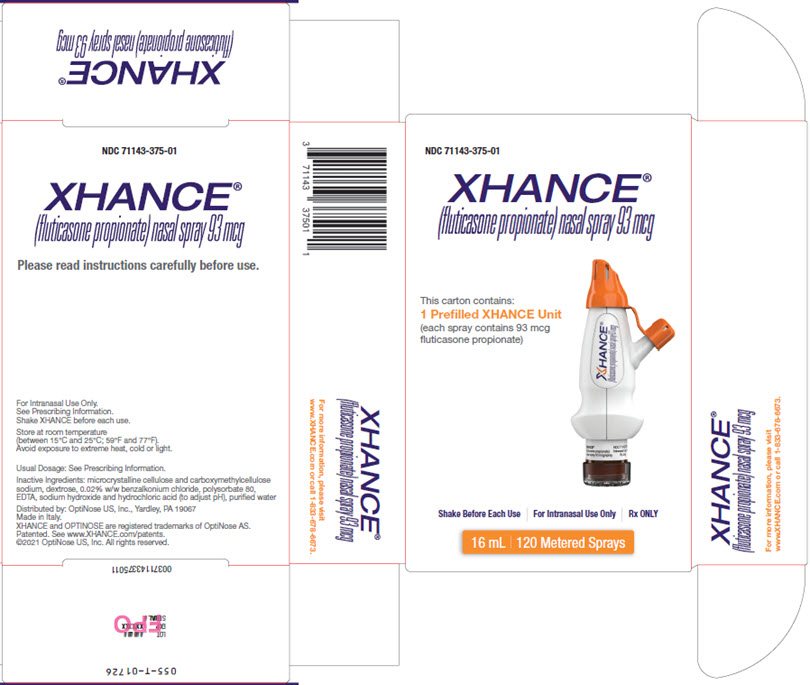
PRINCIPAL DISPLAY PANEL - Carton Label - Outside
NDC 71143-375-01
XHANCE®
(fluticasone propionate) nasal spray 93 mcgThis carton contains:
1 Prefilled XHANCE Unit
(each spray contains 93 mcg
fluticasone propionate)Shake Before Each Use For Intranasal Use Only Rx ONLY
16 mL 120 Metered Sprays
NDC 71143-375-01
XHANCE®
(fluticasone propionate) nasal spray 93 mcgPlease read instructions carefully before use.
For Intranasal Use Only.
See Prescribing Information.
Shake XHANCE before each use.Store at room temperature
(between 15°C and 25°C; 59°F and 77°F).
Avoid exposure to extreme heat, cold or light.Usual Dosage: See Prescribing Information.
Inactive Ingredients: microcrystalline cellulose and carboxymethylcellulose
sodium, dextrose, 0.02% w/w benzalkonium chloride, polysorbate 80,
EDTA, sodium hydroxide and hydrochloric acid (to adjust pH), purified waterDistributed by: OptiNose US, Inc., Yardley, PA 19067
Made in Italy.
XHANCE and OPTINOSE are registered trademarks of OptiNose AS.
Patented. See www.XHANCE.com/patents.
©2021 OptiNose US, Inc. All rights reserved.XHANCE®
(fluticasone propionate) nasal spray 93 mcgFor more information, please visit
www.XHANCE.com or call 1.833.678.6673 - PRINCIPAL DISPLAY PANEL - Carton Label - Inside
-
INGREDIENTS AND APPEARANCE
XHANCE
fluticasone propionate spray, meteredProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:71143-375 Route of Administration NASAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FLUTICASONE PROPIONATE (UNII: O2GMZ0LF5W) (FLUTICASONE - UNII:CUT2W21N7U) FLUTICASONE PROPIONATE 93 ug Inactive Ingredients Ingredient Name Strength MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED FORM (UNII: K679OBS311) DEXTROSE, UNSPECIFIED FORM (UNII: IY9XDZ35W2) BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) POLYSORBATE 80 (UNII: 6OZP39ZG8H) EDETATE DISODIUM (UNII: 7FLD91C86K) WATER (UNII: 059QF0KO0R) SODIUM HYDROXIDE (UNII: 55X04QC32I) HYDROCHLORIC ACID (UNII: QTT17582CB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71143-375-01 1 in 1 CARTON 10/03/2017 1 120 in 1 BOTTLE; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) 2 NDC:71143-375-99 1 in 1 CARTON 10/03/2017 01/31/2023 2 120 in 1 BOTTLE; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) 3 NDC:71143-375-98 1 in 1 CARTON 03/01/2019 3 28 in 1 BOTTLE; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA209022 10/03/2017 Labeler - OptiNose US, Inc. (963350926)