CUTIVATE- fluticasone propionate ointment
PharmaDerm a division of Fougera Pharmaceuticals Inc.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use CUTIVATE® Ointment safely and effectively. See full prescribing information for CUTIVATE® Ointment.
CUTIVATE (fluticasone propionate) ointment, for topical use Initial U.S. Approval: 1990 RECENT MAJOR CHANGES
Warnings and Precautions, Ophthalmic Adverse Reactions (5.5) 03/2021 INDICATIONS AND USAGE
DOSAGE AND ADMINISTRATIONDOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
ADVERSE REACTIONS
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling. Revised: 3/2021 |
FULL PRESCRIBING INFORMATION
2 DOSAGE AND ADMINISTRATION
- Apply a thin film of CUTIVATE® Ointment to the affected skin areas twice daily. Rub in gently.
- Avoid use with occlusive dressing.
CUTIVATE® Ointment is for topical use only; it is not for ophthalmic, oral or intravaginal use.
3 DOSAGE FORMS AND STRENGTHS
Ointment, 0.005%. Each gram of CUTIVATE® Ointment contains 0.05 mg fluticasone propionate in a white to off-white translucent ointment base.
4 CONTRAINDICATIONS
- CUTIVATE® Ointment is contraindicated in patients with a history of hypersensitivity to any of the components in the preparation.
5 WARNINGS AND PRECAUTIONS
5.1 Hypothalamic-Pituitary-Adrenal (HPA) Axis Suppression and Other Adverse Endocrine Effects
- Topical corticosteroids, including CUTIVATE® Ointment, can produce reversible HPA axis suppression with the potential for clinical glucocorticoid insufficiency. Factors that predispose to HPA axis suppression include large treatment surface areas, prolonged use, use under occlusion, altered skin barrier, liver failure, and young age. Cushing’s syndrome, hyperglycemia, and unmasking of latent diabetes mellitus can also result from systemic absorption of topical corticosteroids.
- HPA axis suppression may occur during or after withdrawal of treatment. If HPA axis suppression is suspected, gradually withdraw the drug, reduce the frequency of application, or substitute with a less potent corticosteroid. Evaluation of HPA axis suppression may be done by using the cosyntropin stimulation test.
- Pediatric patients may be at greater risk of HPA axis suppression due to their higher skin surface area to body mass ratios [see Use in Specific Populations (8.4)].
5.2 Local Adverse Reactions
- The following local adverse reactions have been reported with topical corticosteroids. They may occur more frequently with the use of occlusive dressings and higher potency corticosteroids. These reactions are listed in an approximate decreasing order of occurrence: irritation, folliculitis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, secondary infection, skin atrophy, striae, hypertrichosis, and miliaria.
5.3 Allergic Contact Dermatitis
- Allergic contact dermatitis with topical corticosteroids is usually diagnosed by observing failure to heal rather than noting a clinical exacerbation. Such an observation can be corroborated with appropriate diagnostic patch testing. Discontinue CUTIVATE® Ointment if appropriate.
5.4 Skin Infections
- If concomitant skin infections are present or develop, use an appropriate antimicrobial. If a favorable response does not occur promptly, discontinue use of CUTIVATE® Ointment until the infection has been adequately controlled.
5.5 Ophthalmic Adverse Reactions
Use of topical corticosteroids may increase the risk of posterior subcapsular cataracts and glaucoma. Cataracts and glaucoma have been reported postmarketing with the use of topical corticosteroids.
Avoid contact of CUTIVATE® Ointment with eyes. Advise patients to report any visual symptoms and consider referral to an ophthalmologist for evaluation.
6 ADVERSE REACTIONS
The following adverse reactions are discussed in greater detail in other sections of the labeling:
- •
- HPA Axis Suppression and Other Adverse Endocrine Effects [see Warnings and Precautions (5.1)]
- •
- Local Adverse Reactions [see Warnings and Precautions (5.2)]
- •
- Concomitant Skin Infections [see Warnings and Precautions (5.4)]
6.1 Clinical Trials Experience
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
- In controlled clinical trials, the total incidence of adverse reactions associated with the use of CUTIVATE® Ointment was approximately 4%. These adverse reactions were usually mild, self-limiting, and consisted primarily of pruritus, burning, hypertrichosis, increased erythema, urticaria, irritation, and lightheadedness. Each of these events occurred individually in less than 1% of subjects.
6.2 Postmarketing Experience
Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- The following local adverse reactions have been identified during post-approval use of CUTIVATE® Ointment: acneiform dermatitis, edema, rash, hypoaesthesia, pustular psoriasis, skin atrophy.
- The following systemic adverse reactions have been identified during post-approval use of CUTIVATE® Cream and CUTIVATE® Ointment: immunosuppression/Pneumocystis jirovecii pneumonia/leukopenia/thrombocytopenia; hyperglycemia/glycosuria; Cushing syndrome; generalized body edema/blurred vision; and acute urticarial reaction (edema, urticaria, pruritus, and throat swelling).
The following local adverse reactions have been reported with the use of topical corticosteroids: telangiectasia, striae, dryness, folliculitis, hypopigmentation, perioral dermatitis, allergic contact dermatitis, secondary infection, and miliaria.
- Ophthalmic adverse reactions including cataracts, glaucoma and increased intraocular pressure have been reported with the use of topical corticosteroids.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no available data on CUTIVATE® Ointment use in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. Observational studies suggest an increased risk of low birthweight infants with the use of greater than 300 grams of potent or very potent topical corticosteroids during pregnancy (see Data). Advise pregnant women that CUTIVATE® Ointment may increase the risk of having a low birthweight infant and to use CUTIVATE® Ointment on the smallest area of skin and for the shortest duration possible.
In animal reproduction studies, subcutaneous administration of fluticasone propionate to pregnant mice, rats, and rabbits during organogenesis caused malformations characteristic of corticosteroids in each species (seeData). The available data do not allow the calculation of relevant comparisons between the systemic exposure of fluticasone propionate observed in animal studies to the systemic exposure that would be expected in humans after topical use of CUTIVATE® Ointment.
The background risk of major birth defects and miscarriage for the indicated population(s) is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Data
Human Data
Available observational studies in pregnant women did not identify a drug-associated risk of major birth defects, preterm delivery, or fetal mortality with the use of topical corticosteroids of any potency. However, when the dispensed amount of potent or very potent topical corticosteroids exceeded 300 grams during the entire pregnancy, maternal use was associated with an increased risk of low birth weight infants.
- Animal Data
- In embryo-fetal development studies, pregnant rabbits, rats, and mice received subcutaneous doses of fluticasone propionate during organogenesis at doses up to 4, 100, and 150 µg/kg/day, respectively. A malformation characteristic of corticosteroids (cleft palate) was noted at the high dose in each species. Additional adverse effects were noted in rats and rabbits. Decreased fetal weights and retarded skeletal ossification were noted in rabbits at 4 µg/kg/day and rats at 100 µg/kg/day. Maternal toxicity and omphalocele were also noted in rats at 100 µg/kg/day. No malformations or developmental toxicity was noted in rabbits at 0.57 µg/kg/day, in rats at 10 µg/kg/day, or in mice at 15 µg/kg/day.
- Fluticasone propionate crossed the placenta following administration of a subcutaneous or an oral dose of 100 μg/kg tritiated fluticasone propionate to pregnant rats.
8.2 Lactation
Risk Summary
There are no data on the presence of fluticasone propionate in human milk, its effects on the breastfed infant, or its effects on milk production. It is not known whether topical administration of CUTIVATE® Ointment could result in sufficient systemic absorption to produce detectable quantities in human milk (see Clinical Considerations).
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for
CUTIVATE® Ointment and any potential adverse effects on the breastfed child from CUTIVATE® Ointment or from the
underlying maternal condition.
Clinical Considerations
To minimize potential exposure to the breastfed infant via breast milk, use CUTIVATE® Ointment on the smallest area of skin and for the shortest duration possible while breastfeeding. Advise breastfeeding women not to apply CUTIVATE® Ointment directly to the nipple and areola prior to breastfeeding to avoid direct infant exposure [see Use in Specific Populations (8.4)].
8.4 Pediatric Use
- The safety and effectiveness of CUTIVATE® Ointment have not been established in pediatric patients. Use of CUTIVATE® Ointment in pediatric patients is not recommended.
- Because of a higher ratio of skin surface area to body mass, pediatric patients are at a greater risk than adults of systemic effects when treated with topical drugs. They are, therefore, also at greater risk of HPA axis suppression and adrenal insufficiency upon the use of topical corticosteroids [see Warnings and Precautions (5.1)].
- In a trial of 35 pediatric subjects treated with fluticasone propionate ointment, 0.005% for atopic dermatitis over at least 35% of body surface area, subnormal adrenal function was observed with cosyntropin stimulation testing at the end of 3 to 4 weeks of treatment in 4 subjects who had normal testing prior to treatment. It is not known if these subjects had recovery of adrenal function because follow-up testing was not performed. The decreased responsiveness of cosyntropin testing was not correlated to age of subject, amount of fluticasone propionate ointment used, or serum levels of fluticasone propionate.
- In the above trial, telangiectasia on the face was noted in one subject on the eighth day of a 4-week treatment period. Facial use was discontinued and the telangiectasia resolved.
- HPA axis suppression, Cushing’s syndrome, linear growth retardation, delayed weight gain, and intracranial hypertension have been reported in pediatric patients receiving topical corticosteroids.
8.5 Geriatric Use
- A limited number of patients above 65 years of age (n = 203) have been treated with CUTIVATE® Ointment in US and non-US clinical trials. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients. However, greater sensitivity of some older individuals cannot be ruled out.
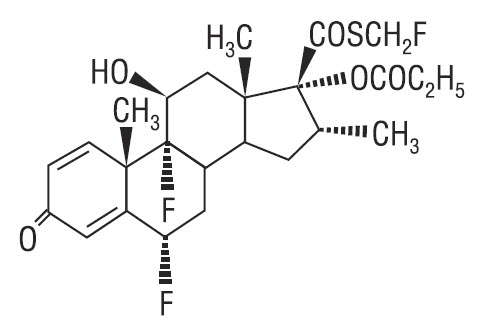
11 DESCRIPTION
CUTIVATE® (fluticasone propionate) Ointment, 0.005% contains fluticasone propionate [S-Fluoromethyl 6α, 9α-difluoro-11β-hydroxy-16α-methyl-3-oxo-17α-propionyloxyandrosta-1,4-diene-17β-carbothioate], a synthetic fluorinated corticosteroid, for topical use.
Chemically, fluticasone propionate is C25H31F3O5S.
It has the following structural formula:
Fluticasone propionate has a molecular weight of 500.6. It is a white to off-white powder and is insoluble in water.
Each gram of CUTIVATE® Ointment contains fluticasone propionate 0.05 mg in a white to off-white translucent ointment base of liquid paraffin, microcrystalline wax, propylene glycol, and sorbitan sesquioleate.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Corticosteroids play a role in cellular signaling, immune function, inflammation, and protein regulation; however, the precise mechanism of action of CUTIVATE® Ointment in corticosteroid-responsive dermatoses is unknown.
12.2 Pharmacodynamics
Vasoconstrictor Assay
- Studies performed with CUTIVATE® Ointment indicate that it is in the medium range of potency as demonstrated in vasoconstrictor trials in healthy subjects when compared with other topical corticosteroids. However, similar blanching scores do not necessarily imply therapeutic equivalence.
12.3 Pharmacokinetics
-
Absorption
The extent of percutaneous absorption of topical corticosteroids is determined by many factors, including the vehicle and the integrity of the epidermal barrier. Occlusive dressing enhances penetration. Topical corticosteroids can be absorbed from normal intact skin. Inflammation and/or other disease processes in the skin increase percutaneous absorption. - In a study of 6 healthy subjects applying 25 g of fluticasone propionate ointment 0.005% twice daily to the trunk and legs for up to 5 days under occlusion, plasma levels of fluticasone ranged from 0.08 to 0.22 ng/mL.
-
Distribution
The percentage of fluticasone propionate bound to human plasma proteins averaged 91%. Fluticasone propionate is weakly and reversibly bound to erythrocytes. Fluticasone propionate is not significantly bound to human transcortin. -
Metabolism
No metabolites of fluticasone propionate were detected in an in vitro study of radiolabeled fluticasone propionate incubated in a human skin homogenate. - Fluticasone propionate is metabolized in the liver by cytochrome P450 3A4-mediated hydrolysis of the 5-fluoromethyl carbothiolate grouping. This transformation occurs in 1 metabolic step to produce the inactive 17β-carboxylic acid metabolite, the only known metabolite detected in man. This metabolite has approximately 2000 times less affinity than the parent drug for the glucocorticoid receptor of human lung cytosol in vitro and negligible pharmacological activity in animal studies. Other metabolites detected in vitro using cultured human hepatoma cells have not been detected in man.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
- In an oral (gavage) mouse carcinogenicity study, doses of 0.1, 0.3, and 1 mg/kg/day fluticasone propionate were administered to mice for 18 months. Fluticasone propionate demonstrated no tumorigenic potential at oral doses up to 1 mg/kg/day in this study.
- In a dermal mouse carcinogenicity study, 0.05% fluticasone propionate ointment (40 μL) was topically administered for 1, 3, or 7 days/week for 80 weeks. Fluticasone propionate demonstrated no tumorigenic potential at dermal doses up to 6.7 μg/kg/day in this study.
- Fluticasone propionate was not mutagenic or clastogenic in five in vitro genotoxicity tests (Ames assay, E. coli fluctuation test, S. cerevisiae gene conversion test, Chinese hamster ovary cell chromosome aberration assay and human lymphocyte chromosome aberration assay) and one in vivo genotoxicity test (mouse micronucleus assay).
- In a fertility study, male and female rats received subcutaneous doses of fluticasone propionate at doses up to 50 μg/kg/day. No impairment of fertility or mating performance was observed.
16 HOW SUPPLIED/STORAGE AND HANDLING
CUTIVATE® (fluticasone propionate) Ointment, 0.005% is a white to off-white translucent ointment supplied as follows: supplied as follows:
30 gram tubes NDC 10337-333-30
60 gram tubes NDC 10337-333-60
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Advise the patient:
- •
- Avoid contact with the eyes.
- •
- Do not bandage the treated skin area, or cover or wrap it to cause occlusion unless directed by the healthcare provider.
- •
- Report any signs of local adverse reaction to their healthcare provider.
- •
- Do not use on the face, underarms, or groin areas unless directed by the healthcare provider.
- •
- Advise a woman to use CUTIVATE® Ointment on the smallest area of skin and for the shortest duration possible while pregnant or breastfeeding. Advise breastfeeding women not to apply CUTIVATE® Ointment directly to the nipple and areola to avoid direct infant exposure.
- Manufactured by:
PharmaDerm®
A division of
Fougera
PHARMACEUTICALS INC.
Melville, New York 11747
www.pharmaderm.com
46156942B
#144
PATIENT INFORMATION
|
PATIENT INFORMATION
CUTIVATE® (CUE' ti vāt) |
|
Important: CUTIVATE® Ointment is for use on skin only (topical).
Read this Patient Information before you start using CUTIVATE® Ointment and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or treatment. |
|
What is CUTIVATE® Ointment?
It is not known if CUTIVATE® Ointment is safe and effective in children. |
|
Before using CUTIVATE® Ointment, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Especially tell your healthcare provider if you take other corticosteroid medicines by mouth or use other products on your skin that contain corticosteroids. |
|
How should I use CUTIVATE® Ointment?
|
|
What are possible side effects with CUTIVATE® Ointment?
The most common side effects of CUTIVATE® Ointment include itching, burning, excessive hair growth, skin redness, hives, and lightheadedness. |
|
How should I store CUTIVATE® Ointment?
Keep CUTIVATE® Ointment and all medicines out of the reach of children. |
|
General information about the safe and effective use of CUTIVATE® Ointment.
|
|
What are the ingredients in CUTIVATE® Ointment?
|
Manufactured by:
PharmaDerm®
A division of
Fougera
PHARMACEUTICAL INC.
Melville, New York 11747
www.pharmaderm.com
This Patient Information has been approved by the U.S. Food and Drug Administration
46156942B
Revised: March 2021
#144
| CUTIVATE
fluticasone propionate ointment |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - PharmaDerm a division of Fougera Pharmaceuticals Inc. (043838424) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| GlaxoSmithKline LLC | 149372109 | MANUFACTURE(10337-333) | |