PARICALCITOL- paricalcitol injection, solution
Hospira, Inc.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use PARICALCITOL INJECTION safely and effectively. See full prescribing information for PARICALCITOL INJECTION.
PARICALCITOL injection, for intravenous use Initial U.S. Approval: 1998 RECENT MAJOR CHANGESINDICATIONS AND USAGEParicalcitol Injection is a vitamin D2 analog indicated for the prevention and treatment of secondary hyperparathyroidism in patients 5 years of age and older with chronic kidney disease on dialysis (1). DOSAGE AND ADMINISTRATION
Adult Dose:
Pediatric Dose:
DOSAGE FORMS AND STRENGTHSParicalcitol Injection is available as follows (3):
CONTRAINDICATIONSWARNINGS AND PRECAUTIONS
ADVERSE REACTIONSThe most common adverse reactions (> 5% and more frequent than placebo) are nausea, vomiting, and edema (6.1). To report SUSPECTED ADVERSE REACTIONS, contact Hospira, Inc. at 1-800-441-4100, or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. DRUG INTERACTIONSSee 17 for PATIENT COUNSELING INFORMATION. Revised: 1/2020 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
Paricalcitol Injection is indicated for the prevention and treatment of secondary hyperparathyroidism in patients 5 years of age and older with chronic kidney disease (CKD) on dialysis.
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Information
- Ensure serum calcium is not above the upper limit of normal before initiating treatment [see Warnings and Precautions (5.1)].
- Administer Paricalcitol Injection intravenously through a hemodialysis vascular access port at any time during dialysis.
- Do not inject Paricalcitol Injection directly into a vein.
- Inspect Paricalcitol Injection visually prior to administration; the solution should appear clear and colorless. Do not use if the solution is not clear or particles are present.
2.2 Starting Dose and Dose Titration in Adults
- Initiate Paricalcitol Injection as an intravenous bolus dose of 0.04 mcg/kg to 0.1 mcg/kg (2.8 mcg to 7 mcg) no more frequently than every other day at any time during dialysis.
- Target the maintenance dose of Paricalcitol Injection to intact parathyroid hormone (PTH) levels within the desired therapeutic range and serum calcium within normal limits.
- Monitor serum calcium frequently (e.g., twice weekly) and phosphorus and intact PTH levels every 2 to 4 weeks after initiation of therapy or dose adjustment.
- Titrate the dose of Paricalcitol Injection based on intact PTH (see Table 1). Prior to raising the dose, ensure serum calcium is within normal limits. The maximum daily adult dose is 0.24 mcg/kg.
- Suspend or decrease the dose if intact PTH is persistently and abnormally low to reduce the risk of adynamic bone disease [see Warnings and Precautions (5.3)] or if serum calcium is consistently above the normal range to reduce the risk of hypercalcemia [see Warnings and Precautions (5.1)]. If dose suspension is necessary, restart at a reduced dose after laboratory values have normalized.
| Intact PTH Level at Follow-up Visit | Dosage Adjustment |
|---|---|
|
|
| Above target and intact PTH increased | Increase* by 2 mcg to 4 mcg every 2 to 4 weeks |
| Above target and intact PTH decreased by less than 30% | Increase* by 2 mcg to 4 mcg every 2 to 4 weeks |
| Above target and intact PTH decreased by 30% to 60% | No change |
| Above target and intact PTH decreased by more than 60% | Decrease per clinical judgement |
| At target and intact PTH stable | No change |
2.3 Starting Dose and Dose Titration for Pediatric Patients 5 Years of Age and Above
-
Initiate Paricalcitol Injection as an intravenous bolus dose of:
- 0.04 mcg/kg if baseline intact PTH is less than 500 pg/mL, or
- 0.08 mcg/kg if baseline intact PTH is 500 pg/mL or greater
- Administer Paricalcitol Injection three times per week, no more frequently than every other day, at any time during dialysis.
- Target the maintenance dose of Paricalcitol Injection to intact PTH levels within the desired therapeutic range and serum calcium within normal limits.
- Monitor serum calcium frequently (e.g., twice weekly) and phosphorus and intact PTH levels every 2 to 4 weeks after initiation of therapy or dose adjustment.
- Titrate the dose of Paricalcitol Injection based on intact PTH (see Table 2). Prior to raising the dose, ensure serum calcium is within normal limits.
- Suspend or decrease the dose if intact PTH is persistently and abnormally low to reduce the risk of adynamic bone disease [see Warnings and Precautions (5.3)] or if serum calcium is consistently above the normal range to reduce the risk of hypercalcemia [see Warnings and Precautions (5.1)]. If dose suspension is necessary, restart at a reduced dose after laboratory values have normalized.
| Intact PTH Level at Follow-up Visit | Dosage Adjustment |
|---|---|
| Above target and intact PTH decreased by less than 30% | Increase by 0.04 mcg/kg every 2 to 4 weeks |
| Intact PTH 150 pg/mL or greater and decreased by 30% to 60% | No change |
| Intact PTH less than 150 pg/mL or decreased by more than 60% | Decrease by 0.04 mcg/kg weekly, or by 50% if decreased dose equals zero |
2.4 Drug Interactions that May Require Dosage Adjustments of Paricalcitol Injection
- Increased monitoring of serum calcium and dose adjustment of Paricalcitol Injection may be necessary when given concomitantly with drugs that may increase the risk of hypercalcemia [see Drug Interactions (7)].
- Increased monitoring of both serum calcium and intact PTH as well as dose adjustment of Paricalcitol Injection may be necessary when given concomitantly with strong CYP3A inhibitors [see Drug Interactions (7)].
3 DOSAGE FORMS AND STRENGTHS
Paricalcitol Injection is a clear, colorless solution available in multiple-dose vials as follows:
- 2 mcg/mL
- 5 mcg/mL
- 10 mcg/2 mL (5 mcg/mL)
4 CONTRAINDICATIONS
Paricalcitol Injection is contraindicated in patients with:
- Hypercalcemia [see Warnings and Precautions (5.1)]
- Vitamin D toxicity [see Warnings and Precautions (5.1)] or
- Known hypersensitivity to paricalcitol or any inactive ingredient in Paricalcitol Injection. Hypersensitivity adverse reactions have been reported [e.g., angioedema (including laryngeal edema) and urticaria] [see Adverse Reactions (6.2)]
5 WARNINGS AND PRECAUTIONS
5.1 Hypercalcemia
Hypercalcemia may occur during Paricalcitol Injection treatment. Acute hypercalcemia may increase the risk of cardiac arrhythmias and seizures and may potentiate the effect of digitalis on the heart [see Warnings and Precautions (5.2)]. Chronic hypercalcemia can lead to generalized vascular calcification and other soft-tissue calcification. Severe hypercalcemia may require emergency attention.
Hypercalcemia may be exacerbated by concomitant administration of high doses of calcium-containing preparations, thiazide diuretics, or other vitamin D compounds [see Drug Interactions (7)]. In addition, high intake of calcium and phosphate concomitantly with vitamin D compounds may lead to hypercalciuria and hyperphosphatemia. Patients with a history of hypercalcemia prior to initiating therapy may be at increased risk for development of hypercalcemia with Paricalcitol Injection. In these circumstances, frequent serum calcium monitoring and Paricalcitol Injection dose adjustments may be required.
When initiating Paricalcitol Injection or adjusting Paricalcitol Injection dose, measure serum calcium frequently (e.g., twice weekly). Once a maintenance dose has been established, measure serum calcium at least monthly. If hypercalcemia occurs, reduce the dose or discontinue Paricalcitol Injection until serum calcium is normal [see Dosage and Administration (2.2, 2.3)].
Inform patients about the symptoms of elevated calcium (feeling tired, difficulty thinking clearly, loss of appetite, nausea, vomiting, constipation, increased thirst, increased urination, and weight loss) and instruct them to report new or worsening symptoms when they occur.
5.2 Digitalis Toxicity
Paricalcitol Injection can cause hypercalcemia [see Warnings and Precautions (5.1)] which increases the risk of digitalis toxicity. In patients using Paricalcitol Injection concomitantly with digitalis compounds, monitor serum calcium and patients for signs and symptoms of digitalis toxicity. Increase the frequency of monitoring when initiating or adjusting the dose of Paricalcitol Injection [see Drug Interactions (7)].
5.3 Adynamic Bone Disease
Adynamic bone disease with subsequent increased risk of fractures may develop if intact PTH levels are suppressed by Paricalcitol Injection to abnormally low levels. Monitor intact PTH levels to avoid over suppression and adjust Paricalcitol Injection dose, if needed [see Dosage and Administration (2.2, 2.3)].
6 ADVERSE REACTIONS
The following serious adverse reactions are described below and elsewhere in the labeling:
- Hypercalcemia [see Warnings and Precautions (5.1)]
- Adynamic Bone Disease [see Warnings and Precautions (5.3)]
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in practice.
The safety of Paricalcitol Injection has been established from adequate and well-controlled studies of another paricalcitol injection product [see Clinical Studies (14)]. Below is a display of the adverse reactions of paricalcitol injection in these adequate and well-controlled studies.
Four placebo-controlled, double-blind, multicenter studies were conducted in 113 patients (51% male, 10% Caucasian, 81% African-American and 9% Hispanic, ranging in age from 18 to 90 years). Sixty-two patients were exposed to paricalcitol injection and the average dose at the end of treatment was 0.12 mcg/kg/dose with a mean number of 55 days of dosing across the studies. Discontinuation of therapy due to any adverse reaction occurred in 6.5% of patients treated with paricalcitol injection and 2.0% of patients treated with placebo. Adverse reactions occurring with greater frequency in the group treated with paricalcitol injection and at a frequency of 2% or greater are presented in Table 3.
| Adverse Reaction | Placebo (n = 51) % | Paricalcitol Injection (n = 62) % |
|---|---|---|
| Nausea | 8 | 13 |
| Vomiting | 6 | 8 |
| Edema | 0 | 7 |
| Gastrointestinal Hemorrhage | 2 | 5 |
| Chills | 2 | 5 |
| Pyrexia | 2 | 5 |
| Pneumonia | 0 | 5 |
| Sepsis | 2 | 5 |
| Influenza | 4 | 5 |
| Arthralgia | 4 | 5 |
| Palpitations | 0 | 3 |
| Dry Mouth | 2 | 3 |
| Malaise | 0 | 3 |
Other Adverse Reactions
The following adverse reactions occurred in less than 2% of the patients treated with paricalcitol injection in the above-mentioned studies and in additional double-blind, active-controlled and open-label studies:
Blood and Lymphatic System Disorders: Anemia, lymphadenopathy
Cardiac Disorders: Arrhythmia, atrial flutter, irregular heart rate, cardiac arrest, chest discomfort, chest pain, edema peripheral
Ear and Labyrinth Disorders: Ear discomfort
Endocrine Disorders: Hypoparathyroidism
Eye Disorders: Conjunctivitis, glaucoma, ocular hyperemia
Gastrointestinal Disorders: Abdominal discomfort, constipation, diarrhea, dysphagia, gastritis, intestinal ischemia, rectal hemorrhage
General Disorders: Asthenia, condition aggravated, fatigue, feeling abnormal, pain, swelling
Infections: Nasopharyngitis, upper respiratory tract infection, vaginal infection
Injection Site Reactions: Injection site extravasation, injection site pain
Laboratory Abnormalities: Hypercalcemia, hyperkalemia, hyperphosphatemia, hypocalcemia, increased aspartate aminotransferase, prolonged bleeding time
Metabolism and Nutrition Disorders: Decreased appetite, thirst, decreased weight
Musculoskeletal and Connective Tissue Disorders: Joint stiffness, muscle twitching, myalgia
Neoplasms Benign, Malignant and Unspecified: Breast cancer
Nervous System Disorders: Cerebrovascular accident, dizziness, dysgeusia, headache, hypoesthesia, myoclonus, paresthesia, syncope, unresponsive to stimuli, gait disturbance
Psychiatric Disorders: Agitation, confusional state, delirium, insomnia, nervousness, restlessness
Reproductive System and Breast Disorders: Breast pain, erectile dysfunction
Respiratory, Thoracic and Mediastinal Disorders: Cough, dyspnea, orthopnea, pulmonary edema, wheezing
Skin and Subcutaneous Tissue Disorders: Alopecia, blister, hirsutism, night sweats, rash pruritic, pruritus, skin burning sensation
Vascular Disorders: Hypertension, hypotension
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of paricalcitol injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Allergic reactions including rash, urticaria, and angioedema (including laryngeal edema) have been reported.
7 DRUG INTERACTIONS
Table 4 includes clinically significant drug interactions with Paricalcitol Injection.
| Drugs that May Increase the Risk of Hypercalcemia | |
| Clinical Impact | Concomitant administration of high doses of calcium-containing preparations or other vitamin D compounds may increase the risk of hypercalcemia. Thiazide diuretics are known to induce hypercalcemia by reducing excretion of calcium in the urine. |
| Examples | Calcium-containing products, other vitamin D compounds or thiazide diuretics |
| Intervention | Monitor calcium more frequently and adjust Paricalcitol Injection dose as needed [see Warnings and Precautions (5.1)]. |
| Digitalis Compounds | |
| Clinical Impact | Paricalcitol Injection can cause hypercalcemia which can potentiate the risk of digitalis toxicity. |
| Intervention | Monitor patients for signs and symptoms of digitalis toxicity and increase frequency of serum calcium monitoring when initiating or adjusting the dose of Paricalcitol Injection in patients receiving digitalis compounds [see Warnings and Precautions (5.2)]. |
| Strong CYP3A Inhibitors | |
| Clinical Impact | Paricalcitol Injection is partially metabolized by CYP3A. Exposure of Paricalcitol Injection will increase upon coadministration with strong CYP3A inhibitors [see Clinical Pharmacology (12.3)]. |
| Examples | Clarithromycin, conivaptan, grapefruit juice, indinavir, itraconazole, ketoconazole, lopinavir/ritonavir, mibefradil, nefazodone, nelfinavir, posaconazole, ritonavir, saquinavir, and voriconazole |
| Intervention | If a patient initiates or discontinues therapy with a strong CYP3A4 inhibitor, dose adjustment of Paricalcitol Injection may be necessary. Monitor intact PTH and serum calcium concentrations closely. |
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Limited data with Paricalcitol Injection in pregnant women are insufficient to inform a drug-associated risk for major birth defects and miscarriage. There are risks to the mother and fetus associated with chronic kidney disease in pregnancy (see Clinical Considerations).
In animal reproduction studies, slightly increased embryofetal loss was observed in pregnant rats and rabbits administered another paricalcitol product intravenously during the period of organogenesis at doses 2 and 0.5 times, respectively, a human dose of 14 mcg (equivalent to 0.24 mcg/kg), based on body surface area (mg/m2). Adverse reproductive outcomes were observed at doses that caused maternal toxicity (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
Pregnant rats and rabbits were treated with another paricalcitol product by once-daily intravenous injection during the period of organogenesis (in rats, from gestation day [GD] 6 to 17; in rabbits, from GD 6 to 18). Rats were dosed at 0, 0.3, 1, or 3 mcg/kg/day and rabbits at 0, 0.03, 0.1, or 0.3 mcg/kg/day, representing up to 2 or 0.5 times, respectively, a human dose of 0.24 mcg/kg, based on body surface area (mg/m2). Slightly decreased fetal viability was observed in both studies at the highest doses representing 2 and 0.5 times, respectively, a human dose of 0.24 mcg/kg, in the presence of maternal toxicity (decreased body weight and food consumption). Pregnant rats were administered another paricalcitol product by intravenous injection 3 times per week at doses of 0, 0.3, 3, or 20 mcg/kg/day throughout gestation, parturition, and lactation (GD 6 to lactation day [LD] 20) representing exposures up to 13 times a human dose of 0.24 mcg/kg. A small increase in stillbirths and pup deaths from parturition to LD 4 were observed at the high dose when compared to the control group (9.2% versus 3.3% in controls) at 13 times a human dose of 0.24 mcg/kg, which occurred at a maternally toxic dose known to cause hypercalcemia in rats. Surviving pups were not adversely affected; body weight gains, developmental landmarks, reflex ontogeny, learning indices, and locomotor activity were all within normal parameters. F1 reproductive capacity was unaffected.
8.2 Lactation
Risk Summary
There is no information available on the presence of paricalcitol in human milk, the effects of the drug on the breastfed infant or the effects of the drug on milk production. Studies in rats have shown that paricalcitol and/or its metabolites are present in the milk of lactating rats. When a drug is present in animal milk, it is likely that the drug will be present in human milk (see Data). Infants exposed to Paricalcitol Injection through breast milk should be monitored for signs and symptoms of hypercalcemia (see Clinical Considerations). The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for Paricalcitol Injection and any potential adverse effects on the breastfed child from Paricalcitol Injection or from the underlying maternal condition.
Clinical Considerations
Infants exposed to Paricalcitol Injection through breast milk should be monitored for signs and symptoms of hypercalcemia, including seizures, vomiting, constipation, and weight loss. Monitoring of serum calcium in the infant should be considered.
Data
Following a single oral administration of 20 mcg/kg of radioactive [3H] paricalcitol to lactating rats, the concentrations of total radioactivity was determined. Lower levels of total radioactivity were present in the milk compared to that in the plasma of the dams indicating that low levels of [3H] paricalcitol and/or its metabolites are secreted into milk. Exposure of the pups to [3H] paricalcitol through milk was confirmed by the presence of radioactive material in the pups' stomachs.
8.4 Pediatric Use
The safety and efficacy of Paricalcitol Injection for the prevention and treatment of secondary hyperparathyroidism associated with CKD have been established in pediatric patients 5 years of age and older with CKD on dialysis. Use of Paricalcitol Injection in pediatric patients 5 years of age and older is supported by evidence from an adequate and well-controlled study with another paricalcitol product in 29 patients, 5 to 19 years of age, with CKD on hemodialysis [see Clinical Studies (14)].
The safety and efficacy of Paricalcitol Injection have not been established in pediatric patients less than 5 years old.
8.5 Geriatric Use
Clinical studies of paricalcitol injection did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
8.6 Hepatic Impairment
The pharmacokinetics of paricalcitol were studied in patients with mild and moderate hepatic impairment and were similar to that of patients with normal hepatic function. No dose adjustment is required in patients with mild or moderate hepatic function.
Paricalcitol Injection has not been studied in patients with severe hepatic impairment.
10 OVERDOSAGE
Overdosage of Paricalcitol Injection may lead to hypercalcemia, hypercalciuria, and hyperphosphatemia. [see Warnings and Precautions (5.1)].
The treatment of acute overdosage should consist of supportive measures and discontinuation of drug administration. Serum calcium levels should be measured until normal.
Paricalcitol is not significantly removed by dialysis.
11 DESCRIPTION
Paricalcitol, USP, the active ingredient in Paricalcitol Injection, is a synthetically manufactured active vitamin D2 analog. It is a white powder chemically designated as 19-nor-1α,3β,25-trihydroxy-9,10-secoergosta-5(Z),7(E),22(E)-triene and has the following structural formula:
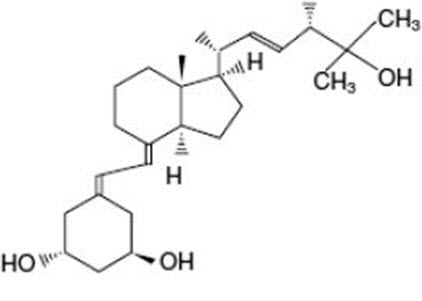 |
| Molecular formula is C27H44O3. Molecular weight is 416.64. |
Paricalcitol Injection is available as a sterile, clear, colorless, aqueous solution for intravenous use. Each mL contains paricalcitol, 2 mcg or 5 mcg and the following inactive ingredients: alcohol 40% (v/v) and propylene glycol 10% (v/v) in water for injection.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Paricalcitol is a synthetic, biologically active vitamin D2 analog. Preclinical and in vitro studies have demonstrated that paricalcitol's biological actions are mediated through binding of the vitamin D receptor (VDR), which results in the selective activation of vitamin D responsive pathways. Vitamin D and paricalcitol have been shown to reduce parathyroid hormone levels by inhibiting PTH synthesis and secretion.
12.3 Pharmacokinetics
Within two hours after administering paricalcitol intravenous doses ranging from 0.04 to 0.24 mcg/kg, concentrations of paricalcitol decreased rapidly; thereafter, concentrations of paricalcitol declined log-linearly. No accumulation of paricalcitol was observed with three times a week dosing.
Distribution
Paricalcitol is extensively bound to plasma proteins (≥ 99.8%). In healthy subjects, the steady state volume of distribution is approximately 23.8 L. The mean apparent volume of distribution following a 0.24 mcg/kg dose of paricalcitol in CKD patients requiring hemodialysis (HD) and peritoneal dialysis (PD) is between 31 and 35 L.
Elimination
Metabolism
After intravenous administration of a 0.48 mcg/kg dose of 3H-paricalcitol, parent drug was extensively metabolized, with only about 2% of the dose eliminated unchanged in the feces and no parent drug found in the urine. Several metabolites were detected in both the urine and feces. Most of the systemic exposure was from the parent drug. Two minor metabolites, relative to paricalcitol, were detected in human plasma. One metabolite was identified as 24(R)-hydroxy paricalcitol, while the other metabolite was unidentified. The 24(R)-hydroxy paricalcitol is less active than paricalcitol in an in vivo rat model of PTH suppression.
In vitro data suggest that paricalcitol is metabolized by multiple hepatic and non-hepatic enzymes, including mitochondrial CYP24, as well as CYP3A4 and UGT1A4. The identified metabolites include the product of 24(R)-hydroxylation (present at low levels in plasma), as well as 24,26- and 24,28-dihydroxylation and direct glucuronidation.
Excretion
Paricalcitol is excreted primarily by hepatobiliary excretion. Approximately 63% of a radioactive dose was recovered in the feces and 19% was recovered in the urine in healthy subjects. In healthy subjects, the mean elimination half-life of paricalcitol is about five to seven hours over the studied dose range of 0.04 to 0.16 mcg/kg. The pharmacokinetics of paricalcitol has been studied in CKD patients requiring hemodialysis (HD) and peritoneal dialysis (PD). The mean elimination half-life of paricalcitol after administration of 0.24 mcg/kg paricalcitol intravenous bolus dose in CKD HD and PD patients is 13.9 and 15.4 hours, respectively (Table 5).
| CKD - HD n = 14 | CKD - PD n = 8 |
|
|---|---|---|
|
||
| Cmax (ng/mL) | 1.680 ± 0.511 | 1.832 ± 0.315 |
| AUC0–∞ (ng∙h/mL) | 14.51 ± 4.12 | 16.01 ± 5.98 |
| β (1/h) | 0.050 ± 0.023 | 0.045 ± 0.026 |
| t1/2 (h)* | 13.9 ± 7.3 | 15.4 ± 10.5 |
| CL (L/h) | 1.49 ± 0.60 | 1.54 ± 0.95 |
| Vdβ (L) | 30.8 ± 7.5 | 34.9 ± 9.5 |
Specific Populations
The pharmacokinetics of paricalcitol has not been investigated in geriatric and pediatric patients.
Patients with Hepatic Impairment
The disposition of intravenous paricalcitol (0.24 mcg/kg) was compared in patients with mild (n=5) and moderate (n=5) hepatic impairment (as indicated by the Child-Pugh method) and subjects with normal hepatic function (n=10). The pharmacokinetics of unbound paricalcitol were similar across the range of hepatic function evaluated in this study. The influence of severe hepatic impairment on the pharmacokinetics of paricalcitol has not been evaluated.
Patients with Renal Impairment
The pharmacokinetics of paricalcitol have been studied in CKD patients requiring hemodialysis (HD) and peritoneal dialysis (PD). Hemodialysis procedure has essentially no effect on paricalcitol elimination. However, compared to healthy subjects, CKD patients on dialysis showed a decreased CL and increased half-life.
Drug Interaction Studies
An in vitro study indicates that paricalcitol is not an inhibitor of CYP1A2, CYP2A6, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, CYP2E1, or CYP3A at concentrations up to 50 nM (21 ng/mL) (approximately 20-fold greater than that obtained after highest tested dose). In fresh primary cultured hepatocytes, the induction observed at paricalcitol concentrations up to 50 nM was less than two-fold for CYP2B6, CYP2C9 or CYP3A, where the positive controls rendered a six- to nineteen-fold induction. Hence, paricalcitol is not expected to inhibit or induce the clearance of drugs metabolized by these enzymes.
Drug interactions with Paricalcitol Injection have not been studied. The following studies have been performed with oral paricalcitol capsules.
Omeprazole
The pharmacokinetic interaction between paricalcitol capsule (16 mcg) and omeprazole (40 mg; oral), a strong inhibitor of CYP2C19, was investigated in a single dose, crossover study in healthy subjects. The pharmacokinetics of paricalcitol were unaffected when omeprazole was administrated approximately 2 hours prior to the paricalcitol dose.
Strong CYP3A Inhibitors
Ketoconazole
The effect of multiple doses of ketoconazole, a strong inhibitor of CYP3A, administered as 200 mg BID for 5 days on the pharmacokinetics of paricalcitol capsule has been studied in healthy subjects. The Cmax of paricalcitol was minimally affected, but AUC0–∞ approximately doubled in the presence of ketoconazole. The mean half-life of paricalcitol was 17.0 hours in the presence of ketoconazole as compared to 9.8 hours, when paricalcitol was administered alone [see Drug Interactions (7)].
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 104-week carcinogenicity study in CD-1 mice conducted with another paricalcitol product, an increased incidence of uterine leiomyoma and leiomyosarcoma was observed at subcutaneous doses of 1, 3, 10 mcg/kg administered 3 times per week (2 to 15 times the AUC at a human dose of 14 mcg, equivalent to 0.24 mcg/kg based on AUC). The incidence rate of uterine leiomyoma was significantly different than the control group at the highest dose of 10 mcg/kg.
In a 104-week carcinogenicity study in rats conducted with another paricalcitol product, there was an increased incidence of benign adrenal pheochromocytoma at subcutaneous doses of 0.15, 0.5, 1.5 mcg/kg administered 3 times per week (at less than clinical exposure to 7 times the exposure following a human dose of 14 mcg, equivalent to 0.24 mcg/kg based on AUC). The increased incidence of pheochromocytomas in rats may be related to the alteration of calcium homeostasis by paricalcitol.
Paricalcitol did not exhibit genetic toxicity in vitro with or without metabolic activation in the microbial mutagenesis assay (Ames Assay), mouse lymphoma mutagenesis assay (L5178Y), or a human lymphocyte cell chromosomal aberration assay. There was also no evidence of genetic toxicity in an in vivo mouse micronucleus assay.
Paricalcitol had no effect on fertility (male or female) in rats at intravenous doses up to 20 mcg/kg/dose (13 times a human dose of 14 mcg, equivalent to 0.24 mcg/kg based on body surface area, mg/m2).
14 CLINICAL STUDIES
The safety and efficacy of Paricalcitol Injection have been established based on adult studies of another paricalcitol injection product in patients with CKD on hemodialysis. Below is a display of the results of these studies of paricalcitol injection.
Adult Studies in CKD on Dialysis
Three 12-week, placebo-controlled studies were conducted with paricalcitol injection in 78 patients with chronic kidney disease on dialysis. In these studies, patients ranged in age from 22 to 90 years, 51% were males, 13% were Caucasian, 79% were African-American, and 8% were Hispanic. The most common causes of renal failure were hypertension and diabetes. The dose of paricalcitol injection was started at 0.04 mcg/kg 3 times per week intravenously. The dose was increased by 0.04 mcg/kg every 2 weeks until intact PTH levels were decreased at least 30% from baseline or a fifth escalation brought the dose to 0.24 mcg/kg, or intact PTH fell to less than 100 pg/mL, or the Ca × P product was greater than 75 within any 2 week period, or serum calcium became greater than 11.5 mg/dL at any time.
Patients treated with paricalcitol injection achieved a mean intact PTH reduction of 30% within 6 weeks. The results from these studies are as follows:
| Group (No. of Pts.) | Baseline Mean (Range) | Mean (SE) Change From Baseline to Final Evaluation | |
|---|---|---|---|
| Intact PTH (pg/mL) | Paricalcitol injection (n=40) | 783 (291 – 2076) | -379 (43.7) |
| Placebo (n=38) | 745 (320 – 1671) | -69.6 (44.8) | |
| Alkaline Phosphatase (U/L) | Paricalcitol injection (n=31) | 150 (40 – 600) | -41.5 (10.6) |
| Placebo (n=34) | 169 (56 – 911) | +2.6 (10.1) | |
| Phosphorus (mg/dL) | Paricalcitol injection (n=40) | 5.8 (3.7 – 10.2) | +0.47 (0.3) |
| Placebo (n=38) | 6.0 (2.8 – 8.8) | -0.47 (0.3) | |
| Calcium × Phosphorus Product | Paricalcitol injection (n=40) | 54 (32 – 106) | +7.9 (2.2) |
| Placebo (n=38) | 54 (26 – 77) | -3.9 (2.3) | |
Pediatric Study in CKD on Dialysis
Paricalcitol injection was evaluated in a 12-week randomized, double-blind, placebo-controlled study of 29 pediatric patients, aged 5 to 19 years, with CKD on hemodialysis; nearly all had received some form of vitamin D prior to the study. Of the 29 patients, 76% were male, 52% were Caucasian and 45% were African-American. The initial dose of paricalcitol injection was 0.04 mcg/kg 3 times per week, based on baseline intact PTH level of less than 500 pg/mL, or 0.08 mcg/kg 3 times per week, based on baseline intact PTH level of 500 pg/mL or greater. The dose of paricalcitol injection was adjusted in 0.04 mcg/kg increments based on the levels of serum intact PTH, calcium and Ca × P. The mean baseline levels of intact PTH were 841 pg/mL for the 15 patients treated with paricalcitol injection and 740 pg/mL for the 14 placebo-treated patients. The mean dose of paricalcitol injection administered was 4.6 mcg (range: 0.8 mcg to 9.6 mcg). Sixty-seven percent of the patients treated with paricalcitol injection and 14% of the placebo-treated patients completed the trial. Seventy-one percent of the placebo-treated patients discontinued due to excessive elevations in intact PTH levels, as defined by 2 consecutive intact PTH levels greater than 700 pg/mL and greater than baseline after 4 weeks of treatment.
The primary efficacy analysis demonstrated that 60% of patients treated with paricalcitol injection and 21% of placebo-treated patients achieved two consecutive greater than or equal to 30% reductions from baseline in intact PTH.
16 HOW SUPPLIED/STORAGE AND HANDLING
Paricalcitol Injection is a clear, colorless solution available in trays of 25 vials as follows:
| NDC No. | Total Strength per Total Volume | Total Vial Volume and Vial Type |
|---|---|---|
| NDC 0409-1007-01 | 2 mcg/mL | 1 mL multiple-dose vials |
| NDC 0409-1008-01 | 5 mcg/mL | 1 mL multiple-dose vials |
| NDC 0409-1008-02 | 10 mcg/2 mL (5 mcg/mL) | 2 mL multiple-dose vials |
17 PATIENT COUNSELING INFORMATION
Hypercalcemia
Advise patients to contact a health care provider if they develop symptoms of elevated calcium (e.g. feeling tired, difficulty thinking clearly, loss of appetite, nausea, vomiting, constipation, increased thirst, increased urination, and weight loss) [see Warnings and Precautions (5.1)].
Monitoring
Inform patients that they will need routine monitoring of laboratory parameters such as calcium, phosphorus, and intact PTH while receiving Paricalcitol Injection. Inform patients that more frequent monitoring is necessary during the initiation of therapy, following dose changes or when potentially interacting medications are started or discontinued [see Dosage and Administration (2), Drug Interactions (7)].
Drug Interactions
Advise patients to inform their physician of all medications, including prescription and nonprescription drugs, and supplements they are taking. Advise patients to also inform their physician that they are receiving Paricalcitol Injection if a new medication is prescribed [see Drug Interactions (7)].
Diet
Advise patients to adhere to instructions regarding diet and phosphorus restriction [see Warnings and Precautions (5.1)].
PRINCIPAL DISPLAY PANEL - 5 mcg/mL Vial Label
1 mL
Multi-dose Vial
NDC 0409-1008-11
Rx only
Paricalcitol Injection
5 mcg/mL
For intravenous use through
hemodialysis vascular access
port only
RL-4685
Hospira
PRINCIPAL DISPLAY PANEL - 5 mcg/mL Vial Tray
1 mL Multi-dose Vial
NDC 0409-1008-01
Rx only
Paricalcitol Injection
5 mcg/mL
For intravenous use through hemodialysis
vascular access port only
25 Units
Hospira

PRINCIPAL DISPLAY PANEL - 10 mcg/2 mL Vial Label
2 mL
Multi-dose Vial
NDC 0409-1008-12
Rx only
Paricalcitol Injection
10 mcg/2 mL (5 mcg/mL)
For intravenous use through
hemodialysis vascular access
port only
RL-4686
Hospira
PRINCIPAL DISPLAY PANEL - 10 mcg/2 mL Vial Tray
2 mL Multi-dose Vial
NDC 0409-1008-02
Rx only
Paricalcitol Injection
10 mcg/2 mL (5 mcg/mL)
For intravenous use through hemodialysis
vascular access port only
25 Units
Hospira

| PARICALCITOL
paricalcitol injection, solution |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| PARICALCITOL
paricalcitol injection, solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Hospira, Inc. (141588017) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Hospira, Inc. | 093132819 | ANALYSIS(0409-1007, 0409-1008) , MANUFACTURE(0409-1007, 0409-1008) , PACK(0409-1007, 0409-1008) , LABEL(0409-1007, 0409-1008) | |