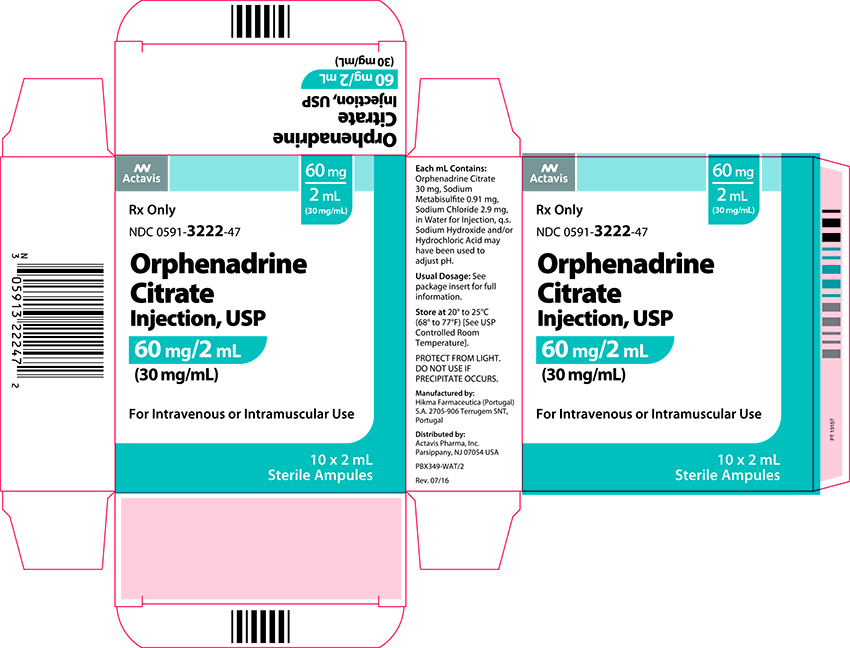
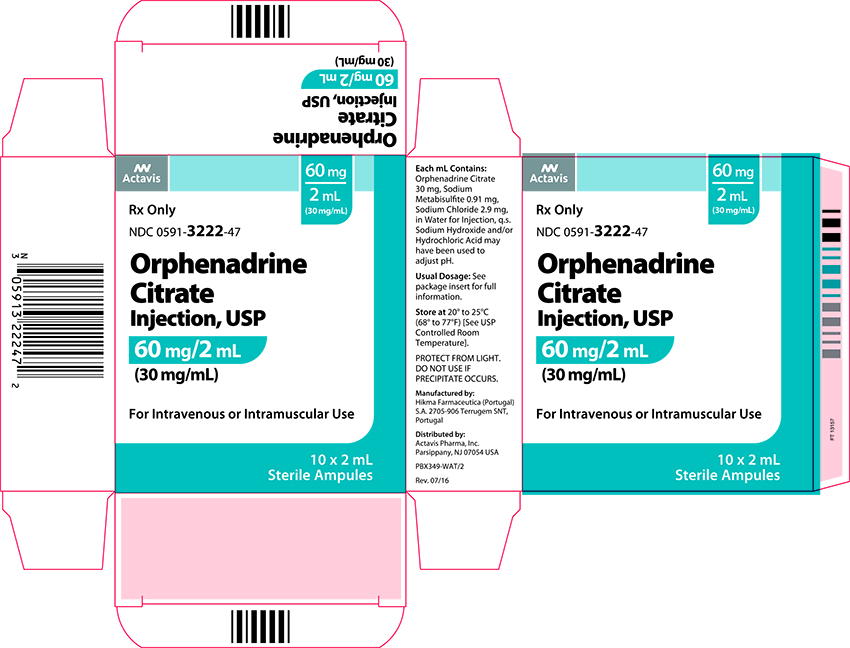
Label: ORPHENADRINE CITRATE injection
- NDC Code(s): 0591-3222-02, 0591-3222-47
- Packager: Actavis Pharma, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated July 20, 2016
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Orphenadrine Citrate Injection is a sterile aqueous solution of orphenadrine citrate. It is intended for intravenous or intramuscular administration.
Orphenadrine citrate is the citrate salt of orphenadrine and occurs as a white, crystalline powder having a bitter taste. It is practically odorless, sparingly soluble in water and slightly soluble in alcohol. It has the following structural formula:
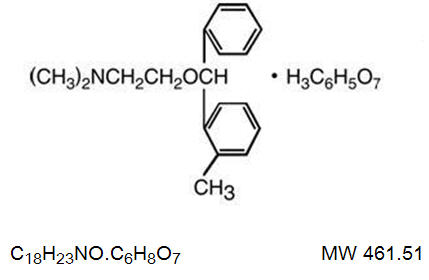
N,N-Dimethyl-2-[(o-methyl-α-phenylbenzyl)oxy]ethylamine Citrate (1:1).
Each mL contains: Orphenadrine Citrate 30 mg, Sodium Metabisulfite 0.91 mg, Sodium Chloride 2.9 mg, in Water for Injection q.s. Sodium Hydroxide and/or Hydrochloric Acid may have been used to adjust pH.
- CLINICAL PHARMACOLOGY
-
INDICATIONS AND USAGE
Orphenadrine citrate is indicated as an adjunct to rest, physical therapy, and other measures for the relief of discomfort associated with acute, painful musculo-skeletal conditions. The mode of action of this drug has not been clearly identified, but may be related to its analgesic properties. Orphenadrine citrate does not directly relax tense skeletal muscles in man.
-
CONTRAINDICATIONS
Orphenadrine citrate is contraindicated in patients with glaucoma, pyloric or duodenal obstruction, stenosing peptic ulcers, prostatic hypertrophy or obstruction at the bladder neck, cardiospasm (megaesophagus) and myasthenia gravis.
Orphenadrine citrate is contraindicated in patients who have demonstrated a previous hypersensitivity to the drug.
-
WARNINGS
Contains Sodium Metabisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in nonasthmatic people.
Some patients may experience transient episodes of lightheadedness, dizziness or syncope.
Orphenadrine citrate may impair the ability of the patient to engage in potentially hazardous activities such as operating machinery or driving a motor vehicle; ambulatory patients should therefore be cautioned accordingly.
Usage in Pregnancy:
Safe use of orphenadrine citrate has not been established with respect to adverse effects upon fetal development. Therefore, orphenadrine citrate should be used in women of childbearing potential and particularly during early pregnancy only when in the judgment of the physician the potential benefits outweigh the possible hazards.
-
PRECAUTIONS
Confusion, anxiety and tremors have been reported in few patients receiving propoxyphene and orphenadrine concomitantly. As these symptoms may be simply due to an additive effect, reduction of dosage and/or discontinuation of one or both agents is recommended in such cases.
Orphenadrine citrate should be used with caution in patients with cardiac decompensation, coronary insufficiency, cardiac arrhythmias, and tachycardia.
Safety of continuous long term therapy with orphenadrine citrate has not been established. Therefore if orphenadrine citrate is prescribed for prolonged use, periodic monitoring of blood, urine, and liver function values is recommended.
-
ADVERSE REACTIONS
Adverse effects of orphenadrine citrate are mainly due to the mild anticholinergic action, and are usually associated with higher dosage. Dryness of the mouth is usually the first adverse effect to appear. When the daily dose is increased, possible adverse effects include: tachycardia, palpitation, urinary hesitancy or retention, blurred vision, dilatation of pupils, increased ocular tension, weakness, nausea, vomiting, headache, dizziness, constipation, drowsiness, hypersensitivity reactions, pruritus, hallucinations, agitation, tremor, gastric irritation, and rarely urticaria and other dermatoses. Infrequently, an elderly patient may experience some degree of mental confusion. These adverse reactions can usually be eliminated by reduction in dosage. Very rare cases of aplastic anemia associated with the use of orphenadrine tablets have been reported. No causal relationship has been established.
Rare instances of anaphylactic reaction have been reported associated with the intramuscular injection of orphenadrine citrate.
-
DOSAGE AND ADMINISTRATION
Adults: 2 mL (60 mg) intravenously or intramuscularly - May be repeated every 12 hours. Relief may be maintained by one (100 mg) orphenadrine citrate tablet twice daily.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever the solution and container permit.
-
HOW SUPPLIED
Orphenadrine Citrate Injection USP, 30 mg/mL is available in 2 mL ampules in cartons of 10.
Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature.] PROTECT FROM LIGHT. DO NOT USE IF PRECIPITATE OCCURS.
Literature revised: July 2016
Product No.: 1108-82
Mfd. by: Hikma Farmaceutica
(Portugal) S.A. 2705-906 Terrugem SNT, PortugalDist. by: Actavis Pharma, Inc.
Parsippany, NJ 07054 USA - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ORPHENADRINE CITRATE
orphenadrine citrate injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0591-3222 Route of Administration INTRAMUSCULAR, INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ORPHENADRINE CITRATE (UNII: X0A40N8I4S) (ORPHENADRINE - UNII:AL805O9OG9) ORPHENADRINE CITRATE 30 mg in 1 mL Inactive Ingredients Ingredient Name Strength SODIUM METABISULFITE (UNII: 4VON5FNS3C) 0.91 mg in 1 mL SODIUM CHLORIDE (UNII: 451W47IQ8X) 2.9 mg in 1 mL WATER (UNII: 059QF0KO0R) SODIUM HYDROXIDE (UNII: 55X04QC32I) HYDROCHLORIC ACID (UNII: QTT17582CB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0591-3222-47 10 in 1 CARTON 07/08/2010 1 NDC:0591-3222-02 2 mL in 1 AMPULE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA084779 07/08/2010 Labeler - Actavis Pharma, Inc. (119723554)