NYSTATIN- nystatin cream
Actavis Pharma, Inc.
----------
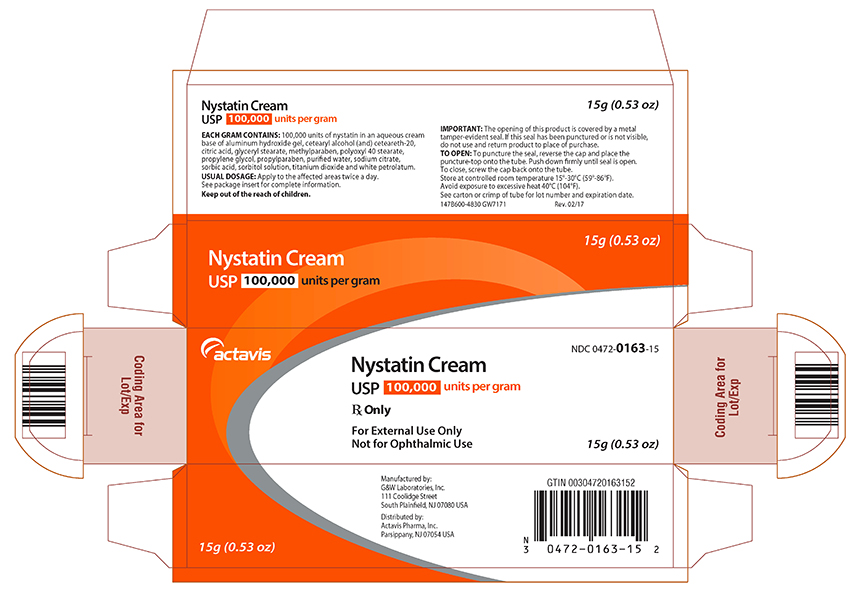
NYSTATIN CREAM USP
DESCRIPTION
EACH GRAM OF NYSTATIN CREAM USP CONTAINS: 100,000 units in an aqueous cream base of aluminum hydroxide gel, cetearyl alcohol (and) ceteareth-20, citric acid, glyceryl stearate, methylparaben, polyoxyl 40 stearate, propylene glycol, propylparaben, purified water, sodium citrate, sorbic acid, sorbitol solution, titanium dioxide and white petrolatum.
CLINICAL PHARMACOLOGY
Nystatin is an antifungal antibiotic which is both fungistatic and fungicidal in vitro against a wide variety of yeasts and yeast-like fungi. It probably acts by binding to sterols in the cell membrane of the fungus with a resultant change in membrane permeability allowing leakage of intracellular components. Nystatin is a polyene antibiotic of undetermined structural formula that is obtained from Streptomyces noursei, and is the first well tolerated antifungal antibiotic of dependable efficacy for the treatment of cutaneous, oral and intestinal infections caused by Candida (Monilia) albicans and other Candida species. It exhibits no appreciable activity against bacteria.
Nystatin provides specific therapy for all localized forms of candidiasis. Symptomatic relief is rapid, often occurring within 24 to 72 hours after the initiation of treatment. Cure is effected both clinically and mycologically in most cases of localized candidiasis.
INDICATIONS AND USAGE
Nystatin Cream USP is indicated in the treatment of cutaneous or mucocutaneous mycotic infections caused by Candida (Monilia) albicans and other Candida species.
CONTRAINDICATIONS
Nystatin Cream USP is contraindicated in patients with a history of hypersensitivity to any of its components.
PRECAUTIONS
Should a reaction of hypersensitivity occur the drug should be immediately withdrawn and appropriate measures taken.
This preparation is not for ophthalmic use.
ADVERSE REACTIONS
Nystatin is virtually nontoxic and nonsensitizing and is well tolerated by all age groups including debilitated infants, even on prolonged administration.
If irritation on topical application should occur, discontinue medication.
DOSAGE AND ADMINISTRATION
Nystatin Cream USP should be applied liberally to the affected areas twice a day or as indicated until healing is complete. Nystatin cream is usually preferred to nystatin ointment in candidiasis involving intertriginous areas; very moist lesions however are best treated with nystatin topical powder.
The cream does not stain the skin or mucous membranes and it provides a simple, convenient means of treatment.
HOW SUPPLIED
Nystatin Cream USP 100,000 units/g
15 g tube (0.53 oz)
30 g tube (1.1 oz)
Store at controlled room temperature 15°-30°C (59°-86°F). Avoid exposure to excessive heat, 40°C (104°F).
Manufactured by:
G&W Laboratories, Inc.
111 Coolidge Street
South Plainfield, NJ 07080 USA
Distributed by:
Actavis Pharma, Inc.
Parsippany, NJ 07054 USA
Revised – April 2017
I600-4830/4835A GW7211
| NYSTATIN
nystatin cream |
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
| Labeler - Actavis Pharma, Inc. (119723554) |