MILRINONE LACTATE- milrinone lactate injection, solution
Hospira, Inc.
----------
Milrinone
Lactate Injection
DESCRIPTION
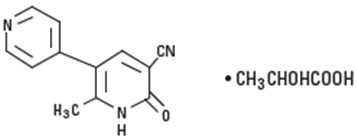
Milrinone lactate injection is a member of a new class of bipyridine inotropic/vasodilator agents with phosphodiesterase inhibitor activity, distinct from digitalis glycosides or catecholamines. Milrinone lactate is designated chemically as 1,6-dihydro-2-methyl-6-oxo-[3,4'-bipyridine]-5-carbonitrile lactate and has the following structure:
Milrinone is an off-white to tan crystalline compound with a molecular weight of 211.2 and an empirical formula of C12H9N3O. It is slightly soluble in methanol, and very slightly soluble in chloroform and in water. As the lactate salt, it is stable and colorless to pale yellow in solution. Milrinone lactate is available as sterile aqueous solutions of the lactate salt of milrinone for injection or infusion intravenously.
The Flexible Containers provide two ready-to-use dilutions of milrinone in volumes of 100 mL and 200 mL of 5% Dextrose Injection. Each mL contains milrinone lactate equivalent to 200 mcg milrinone. The nominal concentration of lactic acid is 0.282 mg/mL. Each mL also contains 49.4 mg Dextrose, Anhydrous, USP. The pH is adjusted to between 3.2 and 4.0 with lactic acid or sodium hydroxide.
The flexible plastic container is fabricated from a specially formulated polyvinylchloride. Water can permeate from inside the container into the overwrap but not in amounts sufficient to affect the solution significantly. Solutions in contact with the plastic container may leach out certain chemical components from the plastic in very small amounts; however, biological testing was supportive of the safety of the plastic container materials. Exposure to temperatures above 25°C/77°F during transport and storage will lead to minor losses in moisture content. Higher temperatures lead to greater losses. It is unlikely that these minor losses will lead to clinically significant changes within the expiration period.
CLINICAL PHARMACOLOGY
Milrinone is a positive inotrope and vasodilator, with little chronotropic activity different in structure and mode of action from either the digitalis glycosides or catecholamines.
Milrinone, at relevant inotropic and vasorelaxant concentrations, is a selective inhibitor of peak III cAMP phosphodiesterase isozyme in cardiac and vascular muscle. This inhibitory action is consistent with cAMP mediated increases in intracellular ionized calcium and contractile force in cardiac muscle, as well as with cAMP dependent contractile protein phosphorylation and relaxation in vascular muscle. Additional experimental evidence also indicates that milrinone is not a beta-adrenergic agonist nor does it inhibit sodium-potassium adenosine triphosphatase activity as do the digitalis glycosides.
Clinical studies in patients with congestive heart failure have shown that milrinone produces dose-related and plasma drug concentration-related increases in the maximum rate of increase of left ventricular pressure. Studies in normal subjects have shown that milrinone produces increases in the slope of the left ventricular pressure-dimension relationship, indicating a direct inotropic effect of the drug. Milrinone also produces dose-related and plasma concentration-related increases in forearm blood flow in patients with congestive heart failure, indicating a direct arterial vasodilator activity of the drug.
Both the inotropic and vasodilatory effects have been observed over the therapeutic range of plasma milrinone concentrations of 100 ng/mL to 300 ng/mL.
In addition to increasing myocardial contractility, milrinone improves diastolic function as evidenced by improvements in left ventricular diastolic relaxation.
The acute administration of intravenous milrinone has also been evaluated in clinical trials in excess of 1600 patients, with chronic heart failure, heart failure associated with cardiac surgery, and heart failure associated with myocardial infarction. The total number of deaths, either on therapy or shortly thereafter (24 hours) was 15, less than 0.9%, few of which were thought to be drug-related.
Pharmacokinetics
Following intravenous injections of 12.5 mcg/kg to 125 mcg/kg to congestive heart failure patients, milrinone had a volume of distribution of 0.38 liters/kg, a mean terminal elimination half-life of 2.3 hours, and a clearance of 0.13 liters/kg/hr. Following intravenous infusions of 0.20 mcg/kg/min to 0.70 mcg/kg/min to congestive heart failure patients, the drug had a volume of distribution of about 0.45 liters/kg, a mean terminal elimination half-life of 2.4 hours, and a clearance of 0.14 liters/kg/hr. These pharmacokinetic parameters were not dose-dependent, and the area under the plasma concentration versus time curve following injections was significantly dose-dependent.
Milrinone has been shown (by equilibrium dialysis) to be approximately 70% bound to human plasma protein.
The primary route of excretion of milrinone in man is via the urine. The major urinary excretions of orally administered milrinone in man are milrinone (83%) and its 0-glucuronide metabolite (12%). Elimination in normal subjects via the urine is rapid, with approximately 60% recovered within the first two hours following dosing and approximately 90% recovered within the first eight hours following dosing. The mean renal clearance of milrinone is approximately 0.3 liters/min, indicative of active secretion.
Pharmacodynamics
In patients with heart failure due to depressed myocardial function, milrinone produced a prompt dose and plasma concentration related increase in cardiac output and decreases in pulmonary capillary wedge pressure and vascular resistance, which were accompanied by mild-to-moderate increases in heart rate. Additionally, there is no increased effect on myocardial oxygen consumption. In uncontrolled studies, hemodynamic improvement during intravenous therapy with milrinone was accompanied by clinical symptomatic improvement, but the ability of milrinone to relieve symptoms has not been evaluated in controlled clinical trials. The great majority of patients experience improvements in hemodynamic function within 5 to 15 minutes of the initiation of therapy.
In studies in congestive heart failure patients, milrinone when administered as a loading injection followed by a maintenance infusion produced significant mean initial increases in cardiac index of 25 percent, 38 percent, and 42 percent at dose regimens of 37.5 mcg/kg/0.375 mcg/kg/min, 50 mcg/kg/0.50 mcg/kg/min, and 75 mcg/kg/0.75 mcg/kg/min, respectively. Over the same range of loading injections and maintenance infusions, pulmonary capillary wedge pressure significantly decreased by 20 percent, 23 percent, and 36 percent, respectively, while systemic vascular resistance significantly decreased by 17 percent, 21 percent, and 37 percent. Mean arterial pressure fell by up to 5 percent at the two lower dose regimens, but by 17 percent at the highest dose. Patients evaluated for 48 hours maintained improvements in hemodynamic function, with no evidence of diminished response (tachyphylaxis). A smaller number of patients have received infusions of milrinone for periods up to 72 hours without evidence of tachyphylaxis.
The duration of therapy should depend upon patient responsiveness.
Milrinone has a favorable inotropic effect in fully digitalized patients without causing signs of glycoside toxicity. Theoretically, in cases of atrial flutter/fibrillation, it is possible that milrinone may increase ventricular response rate because of its slight enhancement of AV node conduction. In these cases, digitalis should be considered prior to the institution of therapy with milrinone.
Improvement in left ventricular function in patients with ischemic heart disease has been observed. The improvement has occurred without inducing symptoms or electrocardiographic signs of myocardial ischemia.
The steady-state plasma milrinone concentrations after approximately 6 to 12 hours of unchanging maintenance infusion of 0.50 mcg/kg/min are approximately 200 ng/mL. Near maximum favorable effects of milrinone on cardiac output and pulmonary capillary wedge pressure are seen at plasma milrinone concentrations in the 150 ng/mL to 250 ng/mL range.
INDICATIONS AND USAGE
Milrinone lactate injection is indicated for the short-term intravenous treatment of patients with acute decompensated heart failure. Patients receiving milrinone should be observed closely with appropriate electrocardiographic equipment. The facility for immediate treatment of potential cardiac events, which may include life threatening ventricular arrhythmias, must be available. The majority of experience with milrinone has been in patients receiving digoxin and diuretics. There is no experience in controlled trials with infusions of milrinone for periods exceeding 48 hours.
WARNINGS
Whether given orally or by continuous or intermittent intravenous infusion, milrinone has not been shown to be safe or effective in the longer (greater than 48 hours) treatment of patients with heart failure. In a multicenter trial of 1088 patients with Class III and IV heart failure, long-term oral treatment with milrinone was associated with no improvement in symptoms and an increased risk of hospitalization and death. In this study, patients with Class IV symptoms appeared to be at particular risk of life-threatening cardiovascular reactions. There is no evidence that milrinone given by long-term continuous or intermittent infusion does not carry a similar risk.
The use of milrinone both intravenously and orally has been associated with increased frequency of ventricular arrhythmias, including nonsustained ventricular tachycardia. Long-term oral use has been associated with an increased risk of sudden death. Hence, patients receiving milrinone should be observed closely with the use of continuous electrocardiographic monitoring to allow the prompt detection and management of ventricular arrhythmias.
PRECAUTIONS
General
Milrinone should not be used in patients with severe obstructive aortic or pulmonic valvular disease in lieu of surgical relief of the obstruction. Like other inotropic agents, it may aggravate outflow tract obstruction in hypertrophic subaortic stenosis.
Supraventricular and ventricular arrhythmias have been observed in the high-risk population treated. In some patients, injections of milrinone and oral milrinone have been shown to increase ventricular ectopy, including nonsustained ventricular tachycardia. The potential for arrhythmia, present in congestive heart failure itself, may be increased by many drugs or combinations of drugs. Patients receiving milrinone should be closely monitored during infusion.
Milrinone produces a slight shortening of AV node conduction time, indicating a potential for an increased ventricular response rate in patients with atrial flutter/fibrillation which is not controlled with digitalis therapy.
During therapy with milrinone, blood pressure and heart rate should be monitored and the rate of infusion slowed or stopped in patients showing excessive decreases in blood pressure.
If prior vigorous diuretic therapy is suspected to have caused significant decreases in cardiac filling pressure, milrinone should be cautiously administered with monitoring of blood pressure, heart rate, and clinical symptomatology.
There is no experience in controlled trials with infusions of milrinone for periods exceeding 48 hours. Cases of infusion site reaction have been reported with intravenous milrinone therapy (see ADVERSE REACTIONS). Consequently, careful monitoring of the infusion site should be maintained to avoid possible extravasation.
Use in acute myocardial infarction
No clinical studies have been conducted in patients in the acute phase of post myocardial infarction. Until further clinical experience with this class of drugs is gained, milrinone is not recommended in these patients.
Laboratory Tests
Fluid and Electrolytes
Fluid and electrolyte changes and renal function should be carefully monitored during therapy with milrinone. Improvement in cardiac output with resultant diuresis may necessitate a reduction in the dose of diuretic. Potassium loss due to excessive diuresis may predispose digitalized patients to arrhythmias. Therefore, hypokalemia should be corrected by potassium supplementation in advance of or during use of milrinone.
Drug Interactions
No untoward clinical manifestations have been observed in limited experience with patients in whom milrinone was used concurrently with the following drugs: digitalis glycosides; lidocaine, quinidine; hydralazine, prazosin; isosorbide dinitrate, nitroglycerin; chlorthalidone, furosemide, hydrochlorothiazide, spironolactone; captopril; heparin, warfarin, diazepam, insulin; and potassium supplements.
Chemical Interactions
There is an immediate chemical interaction which is evidenced by the formation of a precipitate when furosemide is injected into an intravenous line of an infusion of milrinone. Therefore, furosemide should not be administered in intravenous lines containing milrinone.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Twenty-four months of oral administration of milrinone to mice at doses up to 40 mg/kg/day (about 50 times the human oral therapeutic dose in a 50 kg patient) was unassociated with evidence of carcinogenic potential. Neither was there evidence of carcinogenic potential when milrinone was orally administered to rats at doses up to 5 mg/kg/day (about 6 times the human oral therapeutic dose) for twenty-four months or at 25 mg/kg/day (about 30 times the human oral therapeutic dose) for up to 18 months in males and 20 months in females. Whereas the Chinese Hamster Ovary Chromosome Aberration Assay was positive in the presence of a metabolic activation system, results from the Ames Test, the Mouse Lymphoma Assay, the Micronucleus Test, and the in vivo Rat Bone Marrow Metaphase Analysis indicated an absence of mutagenic potential. In reproductive performance studies in rats, milrinone had no effect on male or female fertility at oral doses up to 32 mg/kg/day.
Animal Toxicity
Oral and intravenous administration of toxic dosages of milrinone to rats and dogs resulted in myocardial degeneration/fibrosis and endocardial hemorrhage, principally affecting the left ventricular papillary muscles. Coronary vascular lesions characterized by periarterial edema and inflammation have been observed in dogs only. The myocardial/endocardial changes are similar to those produced by beta-adrenergic receptor agonists such as isoproterenol, while the vascular changes are similar to those produced by minoxidil and hydralazine. Doses within the recommended clinical dose range (up to 1.13 mg/kg/day) for congestive heart failure patients have not produced significant adverse effects in animals.
Pregnancy
Oral administration of milrinone to pregnant rats and rabbits during organogenesis produced no evidence of teratogenicity at dose levels up to 40 mg/kg/day and 12 mg/kg/day, respectively. Milrinone did not appear to be teratogenic when administered intravenously to pregnant rats at doses up to 3 mg/kg/day (about 2.5 times the maximum recommended clinical intravenous dose) or pregnant rabbits at doses up to 12 mg/kg/day, although an increased resorption rate was apparent at both 8 mg/kg/day and 12 mg/kg/day (intravenous) in the latter species. There are no adequate and well-controlled studies in pregnant women. Milrinone should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nursing Mothers
Caution should be exercised when milrinone is administered to nursing women, since it is not known whether it is excreted in human milk.
Use in Elderly Patients
There are no special dosage recommendations for the elderly patient. Ninety percent of all patients administered milrinone in clinical studies were within the age range of 45 to 70 years, with a mean age of 61 years. Patients in all age groups demonstrated clinically and statistically significant responses. No age-related effects on the incidence of adverse reactions have been observed. Controlled pharmacokinetic studies have not disclosed any age-related effects on the distribution and elimination of milrinone.
ADVERSE REACTIONS
Cardiovascular Effects
In patients receiving milrinone in Phase II and III clinical trials, ventricular arrhythmias were reported in 12.1%: Ventricular ectopic activity, 8.5%; nonsustained ventricular tachycardia, 2.8%; sustained ventricular tachycardia, 1% and ventricular fibrillation, 0.2% (2 patients experienced more than one type of arrhythmia). Holter recordings demonstrated that in some patients injection of milrinone increased ventricular ectopy, including nonsustained ventricular tachycardia. Life-threatening arrhythmias were infrequent and when present have been associated with certain underlying factors such as preexisting arrhythmias, metabolic abnormalities (e.g. hypokalemia), abnormal digoxin levels and catheter insertion. Milrinone was not shown to be arrhythmogenic in an electrophysiology study. Supraventricular arrhythmias were reported in 3.8% of the patients receiving milrinone. The incidence of both supraventricular and ventricular arrhythmias has not been related to the dose or plasma milrinone concentration.
Other cardiovascular adverse reactions include hypotension, 2.9% and angina/chest pain, 1.2%.
In the post marketing experience, there have been rare cases of "torsades de pointes" reported.
CNS Effects
Headaches, usually mild to moderate in severity, have been reported in 2.9% of patients receiving milrinone.
Other Effects
Other adverse reactions reported, but not definitely related to the administration of milrinone include hypokalemia, 0.6%; tremor, 0.4%; and thrombocytopenia, 0.4%.
Isolated spontaneous reports of bronchospasm and anaphylactic shock have been received; and in the post-marketing experience, liver function test abnormalities and skin reactions have been reported.
Post-Marketing Adverse Event Reports
In addition to adverse events reported from clinical trials, the following events have been reported from worldwide post-marketing experience with milrinone:
Isolated spontaneous reports of bronchospasm and anaphylactic shock.
Liver function test abnormalities and skin reactions such as rash.
Administration site conditions: Infusion site reaction
OVERDOSAGE
Doses of milrinone may produce hypotension because of its vasodilator effect. If this occurs, administration of milrinone should be reduced or temporarily discontinued until the patient's condition stabilizes. No specific antidote is known, but general measures for circulatory support should be taken.
DOSAGE AND ADMINISTRATION
Milrinone should be administered with a loading dose followed by a continuous infusion (maintenance dose) according to the following guidelines:
LOADING DOSE
50 mcg/kg: Administer slowly over 10 minutes
The table below shows the loading dose in milliliters (mL) of milrinone (1mg/mL) by patient body weight (kg).
| Patient Body Weight (kg) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
kg |
30 |
40 |
50 |
60 |
70 |
80 |
90 |
100 |
110 |
120 |
|
mL |
1.5 |
2 |
2.5 |
3 |
3.5 |
4 |
4.5 |
5 |
5.5 |
6 |
The loading dose may be given undiluted, but diluting to a rounded total volume of 10 or 20 mL (see Maintenance Dose for diluents) may simplify the visualization of the injection rate.
| Infusion Rate | Total Daily Dose
(24 Hours) | ||
|---|---|---|---|
|
Minimum |
0.375 mcg/kg/min |
0.59 mg/kg |
Administer as a continuous intravenous infusion. |
|
Standard |
0.50 mcg/kg/min |
0.77 mg/kg |
|
|
Maximum |
0.75 mcg/kg/min |
1.13 mg/kg |
The diluents that may be used are 0.45% Sodium Chloride Injection USP, 0.9% Sodium Chloride Injection USP, or 5% Dextrose Injection USP. The table below shows the volume of diluent in milliliters (mL) that must be used to achieve 200 mcg/mL concentration for infusion, and the resultant total volumes.
| Desired Infusion Concentration
mcg/mL | Milrinone
1 mg/mL (mL) | Diluent
(mL) | Total Volume
(mL) |
|---|---|---|---|
|
200 |
10 |
40 |
50 |
|
200 |
20 |
80 |
100 |
The infusion rate should be adjusted according to hemodynamic and clinical response. Patients should be closely monitored. In controlled clinical studies, most patients showed an improvement in hemodynamic status as evidenced by increases in cardiac output and reductions in pulmonary capillary wedge pressure.
Note: See "Dosage Adjustment in Renally Impaired Patients." Dosage may be titrated to the maximum hemodynamic effect and should not exceed 1.13 mg/kg/day. Duration of therapy should depend upon patient responsiveness.
The maintenance dose in mL/hr by patient body weight (kg) may be determined by reference to the following table.
Note: Milrinone supplied in 100 mL and 200 mL Flexible Containers (200 mcg/mL in 5% Dextrose Injection) need not be diluted prior to use.
| Maintenance Dose
(mcg/kg/min) | Patient Body Weight (kg) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 30 | 40 | 50 | 60 | 70 | 80 | 90 | 100 | 110 | 120 | |
|
0.375 |
3.4 |
4.5 |
5.6 |
6.8 |
7.9 |
9 |
10.1 |
11.3 |
12.4 |
13.5 |
|
0.400 |
3.6 |
4.8 |
6 |
7.2 |
8.4 |
9.6 |
10.8 |
12 |
13.2 |
14.4 |
|
0.500 |
4.5 |
6 |
7.5 |
9 |
10.5 |
12 |
13.5 |
15 |
16.5 |
18 |
|
0.600 |
5.4 |
7.2 |
9 |
10.8 |
12.6 |
14.4 |
16.2 |
18 |
19.8 |
21.6 |
|
0.700 |
6.3 |
8.4 |
10.5 |
12.6 |
14.7 |
16.8 |
18.9 |
21 |
23.1 |
25.2 |
|
0.750 |
6.8 |
9 |
11.3 |
13.5 |
15.8 |
18 |
20.3 |
22.5 |
24.8 |
27 |
When administering milrinone lactate by continuous infusion, it is advisable to use a calibrated electronic infusion device.
The Flexible Container has a concentration of milrinone equivalent to 200 mcg/mL in 5% Dextrose Injection.
INSTRUCTIONS FOR USE
To Open
Tear outer wrap at notch and remove solution container. Some opacity of the plastic due to moisture absorption during the sterilization process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually.
Preparation for Administration
(Use aseptic technique)
- 1.
- Close flow control clamp of administration set.
- 2.
- Remove cover from outlet port at bottom of container.
- 3.
- Insert piercing pin of administration set into port with a twisting motion until the set is firmly seated.
NOTE: See full directions on administration set carton. - 4.
- Suspend container from hanger.
- 5.
- Squeeze and release drip chamber to establish proper fluid level in chamber.
- 6.
- Open flow control clamp and clear air from set. Close clamp.
- 7.
- Attach set to venipuncture device. If device is not indwelling, prime and make venipuncture.
- 8.
- Regulate rate of administration with flow control clamp.
WARNING: Do not use flexible container in series connections. Such use could result in air embolism due to residual air being drawn from the primary container before administration of the fluid from the secondary container is complete.
Intravenous drug products should be inspected visually and should not be used if particulate matter or discoloration is present.
Dosage Adjustment in Renally Impaired Patients
Data obtained from patients with severe renal impairment (creatinine clearance = 0 to 30 mL/min) but without congestive heart failure have demonstrated that the presence of renal impairment significantly increases the terminal elimination half-life of milrinone. Reductions in infusion rate may be necessary in patients with renal impairment. For patients with clinical evidence of renal impairment, the recommended infusion rate can be obtained from the following table:
| Creatinine Clearance
(mL/min/1.73 m2) | Infusion Rate
(mcg/kg/min) |
|---|---|
|
5 |
0.20 |
|
10 |
0.23 |
|
20 |
0.28 |
|
30 |
0.33 |
|
40 |
0.38 |
|
50 |
0.43 |
HOW SUPPLIED
Milrinone Lactate Injection in 5% Dextrose is supplied as follows:
| Unit of Sale | Concentration |
|---|---|
|
NDC 0409-2776-23 | |
|
10 single-dose 100 mL flexible plastic containers in a case |
20 mg/100 mL |
|
NDC 0409-2776-02 | |
|
10 single-dose 200 mL flexible plastic containers in a case |
40 mg/200 mL |
PRINCIPAL DISPLAY PANEL - 100 mL Bag Label
100 mL
NDC 0409-2776-43
Milrinone Lactate in
5% Dextrose Injection
20 mg/100 mL (200 mcg (0.2 mg) per mL)
FOR INTRAVENOUS USE.
SINGLE-DOSE FLEXIBLE CONTAINER
EACH mL CONTAINS MILRINONE LACTATE
EQUIVALENT TO 0.2 mg MILRINONE, 0.282 mg
LACTIC ACID, 49.4 mg DEXTROSE, ANHYDROUS,
USP, IN WATER FOR INJECTION, USP. THE pH IS
ADJUSTED TO BETWEEN 3.2 AND 4.0 WITH LACTIC
ACID OR SODIUM HYDROXIDE. NO PRESERVATIVES
ADDED. USE ONLY IF SOLUTION IS CLEAR,
COLORLESS TO PALE YELLOW AND CONTAINER IS
UNDAMAGED. DISCARD UNUSED PORTION. EACH
FLEXIBLE CONTAINER IS FOR ONE INFUSION ONLY.
USUAL DOSAGE: SEE INSERT. MUST NOT BE USED
IN SERIES CONNECTIONS.
RX ONLY
IM-5204
3
V
CONTAINS DEHP
Hospira
DIST. BY HOSPIRA, INC., LAKE FOREST, IL 60045 USA
PRINCIPAL DISPLAY PANEL - 100 mL Bag Overwrap Label
TO OPEN – TEAR AT NOTCH
100 mL
NDC 0409-2776-43
Milrinone Lactate in
5% Dextrose Injection
20 mg/100 mL (200 mcg (0.2 mg) per mL)
For Intravenous Use.
Each mL contains milrinone lactate equivalent to 0.2 mg milrinone, 0.282 mg
lactic acid, 49.4 mg dextrose, anhydrous, USP, in water for injection, USP. The
pH is adjusted to between 3.2 and 4.0 with lactic acid or sodium hydroxide. No
preservatives added.
Use only if solution is clear, colorless to pale yellow, and container is
undamaged. Single-dose container. Usual dosage: See insert. Discard unused
portion. Each flexible container is for one infusion only. MUST NOT BE USED IN
SERIES CONNECTIONS.
The overwrap is a moisture barrier. Do not remove unit from overwrap until
ready for use. After removing the overwrap, check for minute leaks by squeezing
container firmly. If leaks are found, discard unit as sterility may be impaired. Use
unit promptly when pouch is opened. Store at 20°C to 25°C (68°F to 77°F). [See
USP Controlled Room Temperature.] Protect from freezing. Do not use if
overwrap has been previously opened or damaged.
Rx only
F WR-1554
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
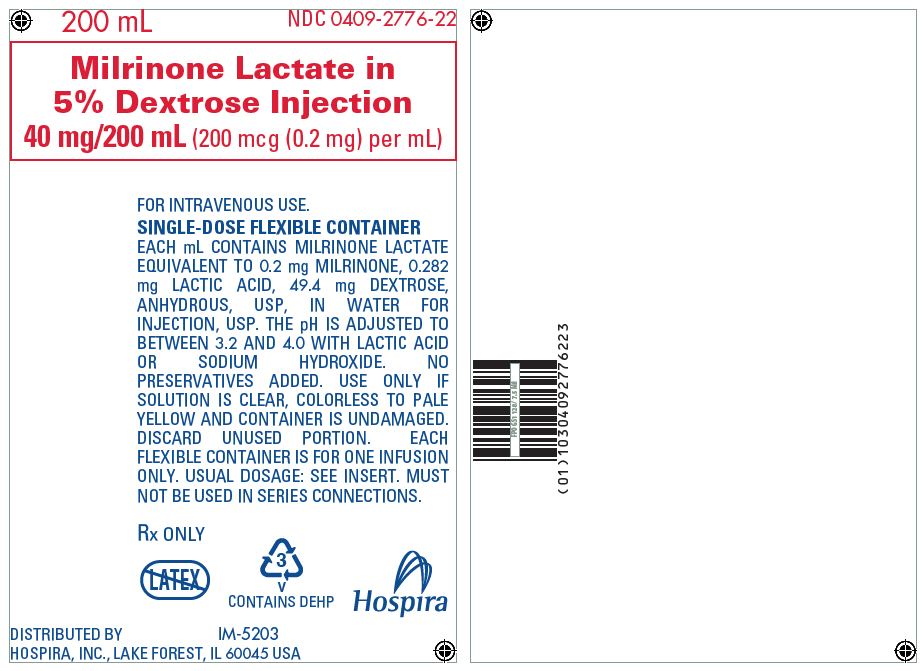
PRINCIPAL DISPLAY PANEL - 200 mL Bag Label
200 mL
NDC 0409-2776-22
Milrinone Lactate in
5% Dextrose Injection
40 mg/200 mL (200 mcg (0.2 mg) per mL)
FOR INTRAVENOUS USE.
SINGLE-DOSE FLEXIBLE CONTAINER
EACH mL CONTAINS MILRINONE LACTATE
EQUIVALENT TO 0.2 mg MILRINONE, 0.282
mg LACTIC ACID, 49.4 mg DEXTROSE,
ANHYDROUS, USP, IN WATER FOR
INJECTION, USP. THE pH IS ADJUSTED TO
BETWEEN 3.2 AND 4.0 WITH LACTIC ACID
OR SODIUM HYDROXIDE. NO
PRESERVATIVES ADDED. USE ONLY IF
SOLUTION IS CLEAR, COLORLESS TO PALE
YELLOW AND CONTAINER IS UNDAMAGED.
DISCARD UNUSED PORTION. EACH
FLEXIBLE CONTAINER IS FOR ONE INFUSION
ONLY. USUAL DOSAGE: SEE INSERT. MUST
NOT BE USED IN SERIES CONNECTIONS.
Rx ONLY
3
V
CONTAINS DEHP
Hospira
DISTRIBUTED BY
HOSPIRA, INC., LAKE FOREST, IL 60045 USA
IM-5203
PRINCIPAL DISPLAY PANEL - 200 mL Bag Overwrap Label
TO OPEN — TEAR AT NOTCH
200 mL
NDC 0409-2776-22
Milrinone Lactate in
5% Dextrose Injection
40 mg/200 mL (200 mcg (0.2 mg) per mL)
For Intravenous Use.
Each mL contains milrinone lactate equivalent to 0.2 mg
milrinone, 0.282 mg lactic acid, 49.4 mg dextrose, anhydrous,
USP, in water for injection, USP. The pH is adjusted to between
3.2 and 4.0 with lactic acid or sodium hydroxide. No preservatives
added.
Use only if solution is clear, colorless to pale yellow, and
container is undamaged. Single-dose container. Usual dosage:
See insert. Discard unused portion. Each flexible container is for
one infusion only. MUST NOT BE USED IN SERIES
CONNECTIONS.
The overwrap is a moisture barrier. Do not remove unit from
overwrap until ready for use. After removing the overwrap,
check for minute leaks by squeezing container firmly. If leaks are
found, discard unit as sterility may be impaired. Use unit promptly
when pouch is opened. Store at 20°C to 25°C (68°F to 77°F).
[See USP Controlled Room Temperature.] Protect from freezing.
Do not use if overwrap has been previously opened or damaged.
Rx only
F WR-1553
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
| MILRINONE LACTATE
milrinone lactate injection, solution |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Hospira, Inc. (141588017) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Hospira, Inc. | 093132819 | ANALYSIS(0409-2776) , MANUFACTURE(0409-2776) , PACK(0409-2776) , LABEL(0409-2776) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Hospira, Inc. | 827731089 | ANALYSIS(0409-2776) | |