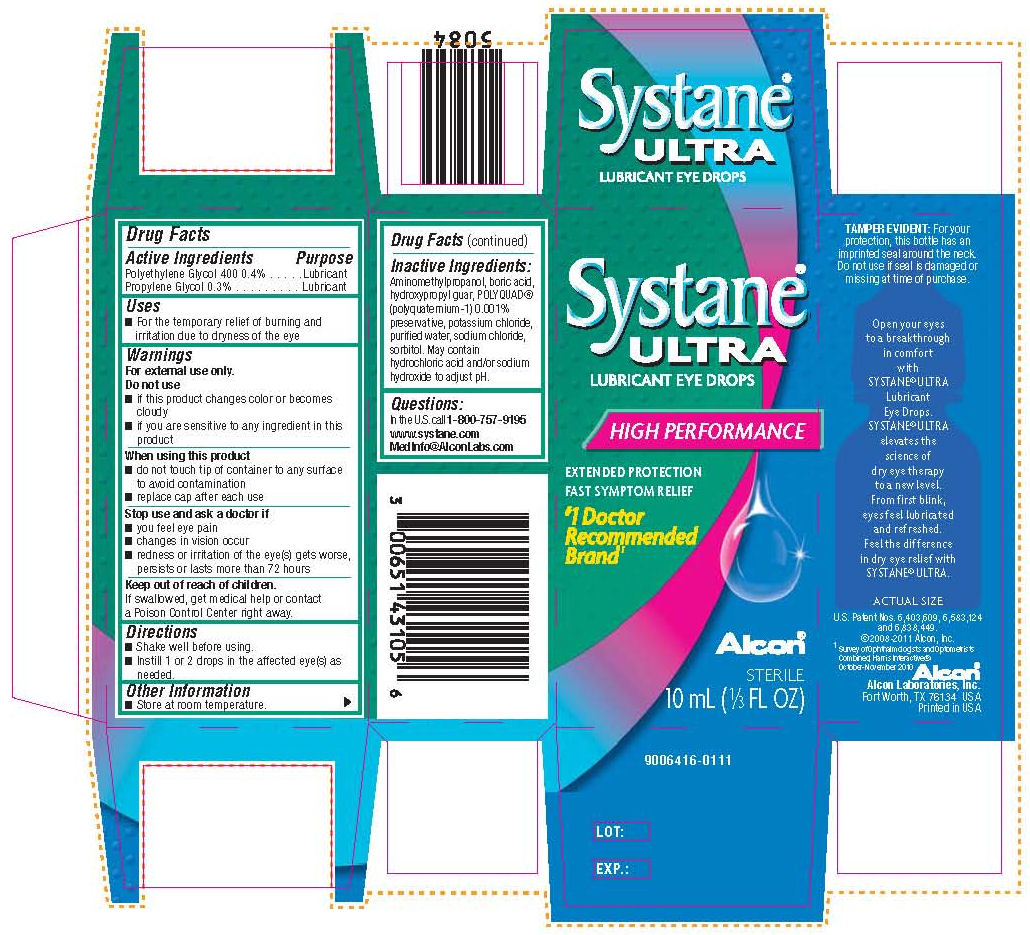
SYSTANE ULTRA - polyethylene glycol 400 and propylene glycol solution/ drops
Alcon Laboratories, Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Drug Facts
INDICATIONS & USAGE SECTION
- For the temporary relief of burning and irritation due to dryness of the eye
OTC - DO NOT USE SECTION
Do not use
- if this product changes color or becomes cloudy
- if you are sensitive to any ingredient in this product
OTC - WHEN USING SECTION
When using this product
- do not touch tip of container to any surface to avoid contamination
- replace cap after each use
OTC - STOP USE SECTION
Stop use and ask a doctor if
- you feel eye pain
- changes in vision occur
- redness or irritation of the eye(s) gets worse, persists or lasts more than 72 hours
OTC - KEEP OUT OF REACH OF CHILDREN SECTION
If swallowed, get medical help or contact a Poison Control Center right away.
DOSAGE & ADMINISTRATION SECTION
- Shake well before using.
- Instill 1 or 2 drops in the affected eye(s) as needed.
| SYSTANE ULTRA
polyethylene glycol 400 and propylene glycol solution/ drops |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| Labeler - Alcon Laboratories, Inc. (008018525) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Alcon Laboratories, Inc. | 008018525 | MANUFACTURE(0065-1431) | |
Revised: 6/2011
Document Id: 04158e63-bdbc-071f-1300-302b3255e943
Set id: 91379a76-fb7f-8357-7d75-7da54fd6d08e
Version: 1
Effective Time: 20110621
Alcon Laboratories, Inc.