ONDANSETRON
-
ondansetron hydrochloride injection, solution
Baxter Healthcare Corporation
----------
|
|||||||||||||||||||||
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
Prevention of nausea and vomiting associated with initial and repeat courses of emetogenic cancer chemotherapy in adult patients, including high-dose cisplatin. Efficacy of the 32 mg single dose beyond 24 hours in these patients has not been established.
2 DOSAGE AND ADMINISTRATION
2.1 Adult Population
Ondansetron Injection, USP is used in the prevention of chemotherapy-induced nausea and vomiting. The recommended intravenous dosage of Ondansetron Injection, USP is a single 32 mg dose infused over 15 minutes beginning 30 minutes before the start of emetogenic chemotherapy. The recommended infusion rate should not be exceeded [see Overdosage (10)].
Ondansetron Injection, USP should not be mixed with solutions for which physical and chemical compatibility have not been established. In particular, this applies to alkaline solutions as a precipitate may form.
Ondansetron Injection, USP REQUIRES NO DILUTION.
2.2 Pediatric Use
The ondansetron premixed formulation is not for use in children. The ondansetron premixed formulation is a fixed dosage form that has not been studied in the pediatric population, and is not appropriate for weight based dosing in pediatric patients [see Use in Specific Populations (8.4) ].
2.3 Dosage Adjustment for Patients with Impaired Hepatic Function
In patients with severe hepatic impairment (Child-Pugh1 Class C), a single maximal daily dose of 8 mg to be infused over 15 minutes, beginning 30 minutes before the start of the emetogenic chemotherapy is recommended. There is no experience beyond first-day administration of ondansetron [see Use in Specific Populations (8.7)].
2.4 Administration
Ondansetron Injection, USP is intended for intravenous infusion only.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
Do not use if the solution is cloudy or precipitated or if the seal is not intact.
Consult with pharmacist for additive compatibility. If additives are introduced, use aseptic technique, mix thoroughly and use immediately.
Ondansetron Injection, USP should not be mixed with solutions for which physical and chemical compatibility have not been established. In particular, this applies to alkaline solutions as a precipitate may form.
Directions for Use:
Do not remove unit from overwrap until ready for use. The overwrap is a moisture and light barrier. The inner bag maintains the sterility of the product.
Caution:
Do not use plastic containers in series connections. Such use could result in air embolism due to residual air being drawn from the primary container before administration of the fluid from the secondary container is completed.
To Open:
Tear overwrap down side at slit and remove solution container. Some opacity of the plastic due to moisture absorption during the sterilization process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually. After removing overwrap, check for minute leaks by squeezing inner bag firmly. If leaks are found, discard unit as sterility may be impaired.
Preparation for Administration:
Use aseptic technique.
- Suspend container from eyelet support.
- Remove protector from outlet port at bottom of container.
- Attach administration set. Refer to complete directions accompanying set.
3 DOSAGE FORMS AND STRENGTHS
Intravenous Injection: 32 mg in 50 mL (contains 32 mg ondansetron as Ondansetron Hydrochloride, USP).
4 CONTRAINDICATIONS
The concomitant use of apomorphine with ondansetron is contraindicated based on reports of profound hypotension and loss of consciousness when apomorphine was administered with ondansetron.
Ondansetron Injection, USP is contraindicated for patients known to have hypersensitivity to the drug.
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity
Hypersensitivity reactions have been reported in patients who have exhibited hypersensitivity to other selective 5-HT3 receptor antagonists.
5.2 Gastrointestinal
Ondansetron is not a drug that stimulates gastric or intestinal peristalsis. It should not be used instead of nasogastric suction. The use of ondansetron in patients following abdominal surgery or in patients with chemotherapy-induced nausea and vomiting may mask a progressive ileus and/or gastric distention.
6 ADVERSE REACTIONS
6.1 Chemotherapy-Induced Nausea and Vomiting
The adverse events in Table 1 have been reported in individuals receiving ondansetron at a dosage of three 0.15-mg/kg doses or as a single 32-mg dose in clinical trials. These patients were receiving concomitant chemotherapy, primarily cisplatin, and intravenous fluids. Most were receiving a diuretic.
|
Principal Adverse Events in Comparative Trials |
||||
| Number of Patients With Event | ||||
|
Ondansetron Injection 0.15 mg/kg x 3 n = 419 |
Ondansetron Injection 32 mg x 1 n = 220 |
Metoclopramide 2 mg/kg x 6 n = 156 |
Placebo n = 34 |
|
| Headache | 17% | 25% | 7% | 15% |
| Diarrhea | 16% | 8% | 44% | 18% |
| Pyrexia | 8% | 7% | 5% | 3% |
| Akathisia | 0% | 0% | 6% | 0% |
| Acute dystonic reactions* | 0% | 0% | 5% | 0% |
6.2 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Cardiac Disorders
Rare cases of angina pectoris (chest pain), hypotension, tachycardia, and electrocardiogram abnormalities have been reported. In many cases, the relationship to ondansetron injection was unclear.
Gastrointestinal Disorders
Constipation has been reported in 11% of chemotherapy patients receiving multiday ondansetron.
Hepatobiliary Disorders
In comparative trials in cisplatin chemotherapy patients with normal baseline values of aspartate aminotransferase (AST) and alanine aminotransferase (ALT), these enzymes have been reported to exceed twice the upper limit of normal in approximately 5% of patients. The increases were transient and did not appear to be related to dose or duration of therapy. On repeat exposure, similar transient elevations in aminotransferase values occurred in some courses, but symptomatic hepatic disease did not occur.
Skin and Subcutaneous Tissue Disorders
Rash has occurred in approximately 1% of patients receiving ondansetron.
Nervous System Disorders
There have been rare cases of grand mal convulsion and rare reports consistent with, but not diagnostic of, extrapyramidal disorders in patients receiving ondansetron injection. The relationship to ondansetron injection was unclear.
Metabolism and Nutrition Disorders
Rare cases of hypokalemia have been reported. The relationship to ondansetron injection was unclear.
6.3 Postmarketing Experience
In addition to adverse events reported from clinical trials, the following events have been identified during post-approval use of intravenous formulations of ondansetron injection. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. The events have been chosen for inclusion due to a combination of their seriousness, frequency of reporting, or potential causal connection to ondansetron.
Cardiac Disorders
Arrhythmias (including ventricular fibrillation, ventricular tachycardia, torsade de pointes, atrial fibrillation, supraventricular tachycardia, and premature ventricular contractions), electrocardiogram abnormalities (including QT prolonged, ST segment depression, and second degree atrioventricular block), syncope, chest discomfort, bradycardia, and palpitations have been reported.
Immune System Disorders
Rare cases of hypersensitivity reactions, sometimes severe (e.g., cardiopulmonary arrest, anaphylactic and anaphylactoid reactions, shock, laryngospasm, stridor, hypotension, bronchospasm, angioedema, laryngeal edema, dyspnea) have been reported.
Gastrointestinal Disorders
Nausea and vomiting have been reported.
Hepatobiliary Disorders
Hepatic necrosis, hepatitis, jaundice, hepatic enzyme abnormalities [including increased aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), gamma-glutamyl transferase (GGT), and bilirubin], and hepatic function abnormal have been reported. Hepatic failure and death have been reported in patients with cancer receiving concurrent medications including potentially hepatotoxic cytotoxic chemotherapy and antibiotics. The etiology of the hepatic failure is unclear.
General Disorders and Administrative Site Conditions
Pain, erythema, and irritation at site of injection have been reported. Chills and lethargy have also been reported.
Respiratory, Thoracic, and Mediastinal Disorders
Hiccups have been reported.
Nervous System Disorders
Oculogyric crisis appearing alone, as well as with other dystonic reactions have been reported.
Skin and Subcutaneous Tissue Disorders
Urticaria, pruritus, flushing, and hyperhidrosis have been reported.
Musculoskeletal and Connective Tissue Disorders
Arthralgia has been reported.
Special Senses
Transient dizziness during or shortly after intravenous infusion has been reported.
Eye Disorders
Transient blurred vision, in some cases associated with accommodation disorder. Rare cases of transient blindness, predominantly during intravenous administration, have been reported. These cases of transient blindness were reported to resolve within a few minutes up to 48 hours.
7 DRUG INTERACTIONS
7.1 Drugs Affecting the Hepatic Cytochrome P-450 Drug-metabolizing Enzyme System
Ondansetron does not itself appear to induce or inhibit the cytochrome P-450 drug-metabolizing enzyme system of the liver [see Clinical Pharmacology (12.3) ]. Because ondansetron is metabolized by hepatic cytochrome P-450 drug-metabolizing enzymes (CYP3A4, CYP2D6, CYP1A2), inducers or inhibitors of these enzymes may change the clearance and, hence, the half-life of ondansetron. On the basis of limited available data, no dosage adjustment is recommended for patients on these drugs.
7.2 Apomorphine
Based on reports of profound hypotension and loss of consciousness when apomorphine was administered with ondansetron, the concomitant use of apomorphine with ondansetron is contraindicated.
7.3 Phenytoin, Carbamazepine, and Rifampicin
In patients treated with potent inducers of CYP3A4 (i.e., phenytoin, carbamazepine, and rifampicin), the clearance of ondansetron was significantly increased and ondansetron blood concentrations were decreased. However, on the basis of available data, no dosage adjustment for ondansetron is recommended for patients on these drugs.2, 3
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Teratogenic Effects
Pregnancy Category B
Reproduction studies have been performed in rats at intravenous doses up to 4 mg/kg/day (about 1 times the recommended human intravenous dose based on body surface area) and in rabbits at intravenous doses up to 4 mg/kg/day (about 2 times the recommended human intravenous dose based on body surface area) and have revealed no evidence of impaired fertility or harm to the fetus due to ondansetron.
There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
8.3 Nursing Mothers
Ondansetron is excreted in the milk of rats. It is not known whether ondansetron is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when ondansetron is administered to a nursing woman.
8.4 Pediatric Use
The safety and effectiveness of Ondansetron Injection 32 mg, USP premixed injection has not been studied in pediatric patients and therefore should not be used in children [see Dosage and Administration (2.2), and Clinical Pharmacology (12.3) ].
8.5 Geriatric Use
Of the total number of subjects enrolled in cancer chemotherapy-induced and postoperative nausea and vomiting in US- and foreign-controlled clinical trials, 862 were 65 years of age and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out. Dosage adjustment is not needed in patients over the age of 65 [see Clinical Pharmacology (12.3) ].
8.6 Renal Impairment
Although plasma clearance is reduced in patients with severe renal impairment (creatine clearance <30 mL/min), no dosage adjustment is recommended [see Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
In patients with severe hepatic impairment, clearance is reduced and apparent volume of distribution is increased with a resultant increase in plasma half-life. In such patients, a total daily dose of 8 mg should not be exceeded [see Dosage and Administration (2.3), and Clinical Pharmacology (12.3)].
9 DRUG ABUSE AND DEPENDENCE
Animal studies have shown that ondansetron is not discriminated as a benzodiazepine nor does it substitute for benzodiazepines in direct addiction studies.
10 OVERDOSAGE
There is no specific antidote for ondansetron overdose. Patients should be managed with appropriate supportive therapy. Individual doses as large as 150 mg and total daily dosages (three doses) as large as 252 mg have been administered intravenously without significant adverse events. These doses are more than 10 times the recommended daily dose.
In addition to the adverse events listed above, the following events have been described in the setting of ondansetron overdose: “Sudden blindness” (amaurosis) of 2 to 3 minutes’ duration plus severe constipation occurred in one patient that was administered 72 mg of ondansetron intravenously as a single dose. Hypotension (and faintness) occurred in another patient that took 48 mg of oral ondansetron. Following infusion of 32 mg over only a 4-minute period, a vasovagal episode with transient second-degree heart block was observed. In all instances, the events resolved completely.
11 DESCRIPTION
The active ingredient in Ondansetron Injection, USP is Ondansetron Hydrochloride USP, the racemic form of ondansetron and a selective blocking agent of the serotonin 5-HT3 receptor type. Chemically it is (±) 1, 2, 3, 9-tetrahydro-9-methyl-3-[(2-methyl-1H-imidazol-1-yl)methyl]-4H-carbazol-4-one, monohydrochloride, dihydrate. It has the following structural formula:
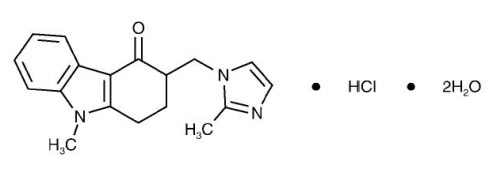
The empirical formula is C18H19N3O•HCl•2H2O, representing a molecular weight of 365.86.
Sterile, Premixed Solution for Intravenous Administration in Single-Dose, Flexible Plastic Containers: A ready to use iso-osmotic formulation of Ondansetron Injection, USP in 50 mL of sodium chloride diluent is available in the INTRAVIA plastic container. Each 50 mL contains 32 mg ondansetron (as Ondansetron Hydrochloride, USP); 450 mg Sodium Chloride, USP; 25 mg Citric Acid Monohydrate, USP; and 12.5 mg Sodium Citrate Dihydrate, USP. Ondansetron Injection, USP is a clear, colorless, nonpyrogenic sterile solution that contains no preservatives. The pH is 3.3 to 4.0.
The flexible plastic container is fabricated from a specially designed multilayer plastic
(PL 2408). Solutions in contact with the plastic container leach out certain chemical components from the plastic in very small amounts; however the plastic container materials conform to USP Biological Reactivity Testing requirements. The flexible container has a foil overwrap. Water can permeate the plastic into the overwrap, but the amount is insufficient to significantly affect the premixed solution.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Ondansetron is a selective 5-HT3 receptor antagonist. While ondansetron’s mechanism of action has not been fully characterized, it is not a dopamine-receptor antagonist.
12.2 Pharmacodynamics
Serotonin receptors of the 5-HT3 type are present both peripherally on vagal nerve terminals and centrally in the chemoreceptor trigger zone of the area postrema. It is not certain whether ondansetron’s antiemetic action in chemotherapy-induced emesis is mediated centrally, peripherally, or in both sites. However, cytotoxic chemotherapy appears to be associated with release of serotonin from the enterochromaffin cells of the small intestine. In humans, urinary 5-HIAA (5-hydroxyindoleacetic acid) excretion increases after cisplatin administration in parallel with the onset of emesis. The released serotonin may stimulate the vagal afferents through the 5-HT3 receptors and initiate the vomiting reflex.
In animals, the emetic response to cisplatin can be prevented by pretreatment with an inhibitor of serotonin synthesis, bilateral abdominal vagotomy and greater splanchnic nerve section, or pretreatment with a serotonin 5-HT3 receptor antagonist.
In normal volunteers, single intravenous doses of 0.15 mg/kg of ondansetron had no effect on esophageal motility, gastric motility, lower esophageal sphincter pressure, or small intestinal transit time. In another study in six normal male volunteers, a 16-mg dose infused over 5 minutes showed no effect of the drug on cardiac output, heart rate, stroke volume, blood pressure, or electrocardiogram (ECG). Multiday administration of ondansetron has been shown to slow colonic transit in normal volunteers. Ondansetron has no effect on plasma prolactin concentrations.
Ondansetron does not alter the respiratory depressant effects produced by alfentanil or the degree of neuromuscular blockade produced by atracurium. Interactions with general or local anesthetics have not been studied.
12.3 Pharmacokinetics
Ondansetron is extensively metabolized in humans, with approximately 5% of a radiolabeled dose recovered as the parent compound from the urine. The primary metabolic pathway is hydroxylation on the indole ring followed by glucuronide or sulfate conjugations.
Although some nonconjugated metabolites have pharmacologic activity, these are not found in plasma at concentrations likely to significantly contribute to the biological activity of ondansetron.
In vitro metabolism studies have shown that ondansetron is a substrate for human hepatic cytochrome P-450 enzymes, including CYP1A2, CYP2D6, and CYP3A4. In terms of overall ondansetron turnover, CYP3A4 played the predominant role. Because of the multiplicity of metabolic enzymes capable of metabolizing ondansetron, it is likely that inhibition or loss of one enzyme (e.g., CYP2D6 genetic deficiency) will be compensated by others and may result in little change in overall rates of ondansetron elimination. Ondansetron elimination may be affected by cytochrome P-450 inducers. In a pharmacokinetic study of 16 epileptic patients maintained chronically on CYP3A4 inducers, carbamazepine, or phenytoin, reduction in AUC, Cmax, and T½ of ondansetron was observed.2 This resulted in a significant increase in clearance. However, on the basis of available data, no dosage adjustment for ondansetron is recommended [see Drug Interactions (7) ].
In humans, carmustine, etoposide, and cisplatin do not affect the pharmacokinetics of ondansetron.
In normal volunteers, the following mean pharmacokinetic data have been determined following a single 0.15-mg/kg intravenous dose (see Table 2).
|
Pharmacokinetics in Normal Adult Volunteers |
||||
| Age-group | n |
Peak Plasma Concentration (ng/mL) |
Mean Elimination Half-life (h) |
Plasma Clearance (L/h/kg) |
| 19-40 | 11 | 102 | 3.5 | 0.381 |
| 61-74 | 12 | 106 | 4.7 | 0.319 |
| >75 | 11 | 170 | 5.5 | 0.262 |
Geriatric: A reduction in clearance and increase in elimination half-life are seen in patients over 75 years of age. In clinical trials with cancer patients, safety and efficacy were similar in patients over 65 years of age and those under 65 years of age; there was an insufficient number of patients over 75 years of age to permit conclusions in that age-group. No dosage adjustment is recommended in the elderly [see Use in Specific Populations (8.5) ].
Renal Impairment: Due to the very small contribution (5%) of renal clearance to the overall clearance, renal impairment was not expected to significantly influence the total clearance of ondansetron. However, ondansetron mean plasma clearance was reduced by about 41% in patients with severe renal impairment (creatinine clearance <30 mL/min). This reduction in clearance is variable and was not consistent with an increase in half-life. No reduction in dose or dosing frequency in these patients is warranted [see Use in Specific Populations (8.6) ].
Hepatic Impairment: In patients with mild-to-moderate hepatic impairment, clearance is reduced twofold and mean half-life is increased to 11.6 hours compared to 5.7 hours in normals. In patients with severe hepatic impairment (Child-Pugh1 Class C), clearance is reduced twofold to threefold and apparent volume of distribution is increased with a resultant increase in half-life to 20 hours. In patients with severe hepatic impairment, a total daily dose of 8 mg should not be exceeded [see Use in Specific Populations (8.7) ].
Pediatric: The pharmacokinetics of single dose 32 mg ondansetron pre-mixed injection have not been characterized in pediatric patients [see Use in Specific Populations (8.4) ].
In normal volunteers (19 to 39 years old, n = 23), the peak plasma concentration was 264 ng/mL following a single 32-mg dose administered as a 15-minute intravenous infusion. The mean elimination half-life was 4.1 hours. Systemic exposure to 32 mg of ondansetron was not proportional to dose as measured by comparing dose-normalized AUC values to an 8-mg dose. This is consistent with a small decrease in systemic clearance with increasing plasma concentrations.
Plasma protein binding of ondansetron as measured in vitro was 70% to 76%, with binding constant over the pharmacologic concentration range (10 to 500 ng/mL). Circulating drug also distributes into erythrocytes.
A positive lymphoblast transformation test to ondansetron has been reported, which suggests immunologic sensitivity to ondansetron.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 2-year oral carcinogenicity study in mice, ondansetron was not carcinogenic at doses up to 30 mg/kg/day (about 3.8 times the recommended human intravenous dose of 32 mg based on body surface area).
In a 2-year oral carcinogenicity study in rats, ondansetron was not carcinogenic at doses up to 10 mg/kg/day (about 2.5 times the recommended human intravenous dose based on body surface area).
Ondansetron was not mutagenic in standard tests for mutagenicity.
Ondansetron at oral doses up to 15 mg/kg/day (about 3.8 times the recommended human intravenous dose based on body surface area) was found to have no effect on fertility and reproductive performance of male or female rats.
14 CLINICAL STUDIES
14.1 Chemotherapy-Induced Nausea and Vomiting
In a double-blind study of three different dosing regimens of ondansetron injection, 0.015 mg/kg, 0.15 mg/kg, and 0.3 mg/kg, each given three times during the course of cancer chemotherapy, the 0.15 mg/kg dosing regimen was more effective than the 0.015 mg/kg dosing regimen. The 0.3 mg/kg dosing regimen was not shown to be more effective than the 0.15 mg/kg dosing regimen.
14.2 Cisplatin-Based Chemotherapy
In a double-blind study in 28 patients, ondansetron injection (three 0.15-mg/kg doses) was significantly more effective than placebo in preventing nausea and vomiting induced by cisplatin-based chemotherapy. Treatment response was as shown in Table 3.
|
|||
|
Treatment Response of Chemotherapy-Induced Nausea and Emesis |
|||
|
Ondansetron Injection 0.15 mg/kg x 3 | Placebo | P Value† | |
| Number of patients | 14 | 14 | |
|
Treatment response 0 Emetic episodes 1-2 Emetic episodes 3-5 Emetic episodes More than 5 emetic episodes/rescued |
2 (14%) 8 (57%) 2 (14%) 2 (14%) |
0 (0%) 0 (0%) 1 (7%) 13 (93%) | 0.001 |
|
Median number of emetic episodes | 1.5 | Undefined‡ | |
|
Median time to first emetic episode (h) | 11.6 | 2.8 | 0.001 |
|
Median nausea scores (0-100)§ | 3 | 59 | 0.034 |
|
Global satisfaction with control of nausea and vomiting (0-100)¶ | 96 | 10.5 | 0.009 |
Ondansetron injection (0.15 mg/kg x 3 doses) was compared with metoclopramide (2 mg/kg x 6 doses) in a single-blind trial in 307 patients receiving cisplatin >100 mg/m2 with or without other chemotherapeutic agents. Patients received the first dose of ondansetron or metoclopramide 30 minutes before cisplatin. Two additional ondansetron doses were administered 4 and 8 hours later, or five additional metoclopramide doses were administered 2, 4, 7, 10, and 13 hours later. Cisplatin was administered over a period of 3 hours or less. Episodes of vomiting and retching were tabulated over the period of 24 hours after cisplatin. The results of this study are summarized in Table 4.
|
|||
|
Treatment Response of Emesis Induced by Cisplatin (>100 mg/m2) |
|||
| Ondansetron Injection | Metoclopramide | P Value | |
| Dose | 0.15 mg/kg x 3 | 2 mg/kg x 6 | |
| Number of patients in efficacy population | 136 | 138 | |
|
Treatment response 0 Emetic episodes 1-2 Emetic episodes 3-5 Emetic episodes More than 5 emetic episodes/rescued |
54 (40%) 34 (25%) 19 (14%) 29 (21%) |
41 (30%) 30 (22%) 18 (13%) 49 (36%) | |
|
Comparison of treatments with respect to 0 Emetic episodes More than 5 emetic episodes/rescued |
54/136 29/136 |
41/138 49/138 |
0.083 0.009 |
| Median number of emetic episodes | 1 | 2 | 0.005 |
| Median time to first emetic episode (h) | 20.5 | 4.3 | <0.001 |
| Global satisfaction with control of nausea and vomiting (0-100)† | 85 | 63 | 0.001 |
| Acute dystonic reactions | 0 | 8 | 0.005 |
| Akathisia | 0 | 10 | 0.002 |
In a stratified, randomized, double-blind, parallel-group, multicenter study, a single 32 mg dose of ondansetron was compared with three 0.15 mg/kg doses in patients receiving cisplatin doses of either 50 to 70 mg/m2 or >100 mg/m2. Patients received the first ondansetron dose 30 minutes before cisplatin. Two additional ondansetron doses were administered 4 and 8 hours later to the group receiving three 0.15-mg/kg doses. In both strata, significantly fewer patients on the single 32-mg dose than those receiving the three-dose regimen failed (see Table 5).
|
Treatment Response of Chemotherapy-Induced Nausea and Emesis in 32 mg Single-Dose Therapy in Adults |
|||
|
Ondansetron Dose 0.15 mg/kg x 3 |
Ondansetron Dose 32 mg x 1 | P Value | |
| High-dose cisplatin ( >100 mg/m2) | |||
| Number of patients | 100 | 102 | |
|
Treatment response 0 Emetic episodes 1-2 Emetic episodes 3-5 Emetic episodes More than 5 emetic episodes/rescued |
41 (41 %) 19 (19%) 4 (4%) 36 (36%) |
49 (48%) 25 (25%) 8 (8%) 20 (20%) |
0.315 0.009 |
| Median time to first emetic episode (h) | 21.7 | 23 | 0.173 |
| Median nausea scores (0-100)* | 28 | 13 | 0.004 |
| Medium-dose cisplatin (50-70 mg/m2) | |||
| Number of patients | 101 | 93 | |
|
Treatment response 0 Emetic episodes 1-2 Emetic episodes 3-5 Emetic episodes More than 5 emetic episodes/rescued |
62 (61%) 11 (11%) 6 (6%) 22 (22%) |
68 (73%) 14 (15%) 3 (3%) 8 (9%) |
0.083 0.011 |
| Median time to first emetic episode (h) | Undefined† | Undefined | |
| Median nausea scores (0-100)* | 9 | 3 | 0.131 |
14.3 Cyclophosphamide-Based Chemotherapy
In a double-blind, placebo-controlled study of ondansetron injection, (three 0.15-mg/kg doses) in 20 patients receiving cyclophosphamide (500 to 600 mg/m2) chemotherapy, ondansetron injection was significantly more effective than placebo in preventing nausea and vomiting. The results are summarized in Table 6.
|
|||
|
Treatment Response of Chemotherapy-Induced Nausea and Emesis in |
|||
|
Ondansetron Injection | Placebo | P Value† | |
| Number of patients | 10 | 10 | |
|
Treatment response 0 Emetic episodes 1-2 Emetic episodes 3-5 Emetic episodes More than 5 emetic episodes/rescued |
7 (70%) 0 (0%) 2 (20%) 1 (10%) |
0 (0%) 2 (20%) 4 (40%) 4 (40%) |
0.001 0.131 |
| Median number of emetic episodes | 0 | 4 | 0.008 |
| Median time to first emetic episode (h) | Undefined‡ | 8.79 | |
| Median nausea scores (0-100)§ | 0 | 60 | 0.001 |
|
Global satisfaction with control of nausea and vomiting (0-100)¶ | 100 | 52 | 0.008 |
14.4 Re-treatment
In uncontrolled trials, 127 patients receiving cisplatin (median dose, 100 mg/m2) and ondansetron who had two or fewer emetic episodes were re-treated with ondansetron and chemotherapy, mainly cisplatin, for a total of 269 re-treatment courses (median, 2; range, 1 to 10). No emetic episodes occurred in 160 (59%), and two or fewer emetic episodes occurred in 217 (81%) re-treatment courses.
15 REFERENCES
- Pugh RNH, Murray-Lyon IM, Dawson JL, Pietroni MC, Williams R. Transection of the oesophagus for bleeding oesophageal varices. Brit J Surg. 1973;60:646-649.
- Britto MR, Hussey EK, Mydlow P, et al. Effect of enzyme inducers on ondansetron (OND) metabolism in humans. Clin Pharmacol Ther 1997;61:228.
- Villikka K, Kivisto KT, Neuvonen PJ. The effect of rifampin on the pharmacokinetics of oral and intravenous ondansetron. Clin Pharmacol Ther 1999;65:377-381.
- De Witte JL, Schoenmaekers B, Sessler DI, et al. Anesth Analg 2001;92:1319-1321.
- Arcioni R, della Rocca M, Romanò R, et al. Anesth Analg 2002;94:1553-1557.
16 HOW SUPPLIED/STORAGE AND HANDLING
Ondansetron Injection, USP premix in INTRAVIA Plastic Containers is supplied as follows:
2J1421 NDC 0338-1762-41 32 mg in 50 mL 10 Pack
Store between 2°C and 30°C (36°F and 86°F). Brief exposure up to 40°C (104°F) does not adversely affect the product. Protect from light until immediately prior to use. Avoid excessive heat. Protect from freezing.
17 PATIENT COUNSELING INFORMATION
To ensure the safe and effective use of Ondansetron Injection, USP Premix, this information should be discussed with the patient.
Inform patients of the following:
- Report any hypersensitivity reactions to this and other selective 5-HT3 receptor antagonists to your health care provider.
- Report the use of all medications especially apomorphine or any drug of the 5-HT3 antagonist class to your health care provider. Concomitant use of apomorphine and ondansetron may cause a significant drop in blood pressure and loss of consciousness.
- Intravenous ondansetron has been reported to cause transient ECG changes including QT interval prolongation.
- Ondansetron may cause headache, diarrhea, and fever.
Baxter Healthcare Corporation
Deerfield, IL 60015 USA
Printed in USA
Baxter and Intravia are registered trademarks of Baxter International Inc.
07-19-60-177
PACKAGE LABEL - PRINCIPAL DISPLAY PANEL
Container Label

LOT
EXP
50 mL
NDC 0338-1762-41
Ondansetron
INJECTION, USP
PREMIXED ISO-OSMOTIC SOLUTION WITH SODIUM
CHLORIDE IN SINGLE-DOSE INTRAVIA CONTAINER
RX ONLY STERILE NONPYROGENIC
EACH 50 ML CONTAINS 32 MG ONDANSETRON AS ONDANSETRON
HYDROCHLORIDE USP 450 MG SODIUM CHLORIDE USP 25 MG
CITRIC ACID MONOHYDRATE USP 12.5 MG SODIUM CITRATE
DIHYDRATE USP PH 3.3 TO 4.0
CONSULT PHARMACIST FOR ADDITIVE COMPATIBILITY IF ADDITIVES ARE
INTRODUCED USE ASEPTIC TECHNIQUE MIX THOROUGHLY AND USE
IMMEDIATELY USUAL DOSAGE INTRAVENOUSLY AS DIRECTED BY A
PHYSICIAN SEE PACKAGE INSERT CAUTIONS MUST NOT BE USED IN
SERIES CONNECTIONS DO NOT USE UNLESS SOLUTION IS CLEAR AND
COLORLESS CHECK FOR MINUTE LEAKS BY SQUEEZING CONTAINER
FIRMLY IF LEAKS ARE FOUND DISCARD CONTAINER AS STERILITY MAY BE
IMPAIRED PROTECT FROM LIGHT UNTIL IMMEDIATELY PRIOR TO USE
2J1421
Carton Label

Lot
Exp
10 x 50 mL single dose INTRAVIA containers
Code 2J1421
50 mL
NDC 0338-1762-41
Ondansetron Injection, USP
32 mg/50 mL
Premixed iso-osmotic solution with
sodium chloride in single-dose containers
Recommended Storage: Store between 2° and 30° C (36° and 86° F). Protect from light until
immediately prior to use. Protect from freezing. Avoid excessive heat.
Rx only
07-06-50-119
Baxter
| ONDANSETRON
ondansetron injection, solution |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| NDA | NDA021915 | 12/27/2006 | 07/25/2012 |
| Labeler - Baxter Healthcare Corporation (005083209) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Baxter Healthcare Corporation | 189326168 | ANALYSIS, LABEL, MANUFACTURE, PACK, STERILIZE | |
Revised: 04/2010 Baxter Healthcare Corporation