RICE FOOD- oryza sativa solution
RYE FOOD- secale cereale solution
SESAME SEED- sesamum indicum solution
BROCCOLI- brassica oleracea var. botrytis solution
BUCKWHEAT- fagopyrum esculentum solution
CASHEW NUT- anacardium occidentale solution
CAULIFLOWER- brassica oleracea var. botrytis solution
CHOCOLATE CACAO BEAN- theobroma cacao solution
CANTALOUPE MUSKMELON- cucumis melo solution
CARROT- daucus carota solution
CELERY- apium graveolens var. dulce solution
LETTUCE- lactuca sativa solution
FILBERT HAZELNUT FOOD- corylus americana solution
VANILLA- vanilla planifolia solution
WHOLE WHEAT- triticum aestivum solution
APRICOT- prunus armeniaca solution
WHOLE GRAIN BARLEY- hordeum vulgare solution
NAVY BEAN- phaseolus vulgaris solution
STRING GREEN BEAN- phaseolus vulgaris solution
CABBAGE- brassica oleracea var. capitata solution
CINNAMON- cinnamomum verum solution
COFFEE- coffea arabica solution
MUSHROOM- agaricus campestris solution
MUSTARD FOOD- sinapis alba solution
BANANA- musa acuminata solution
COCONUT- cocos nucifera solution
CORN FOOD- zea mays solution
MALT- hordeum vulgare solution
BLACK PEPPER- piper nigrum solution
LEMON- citrus x limon solution
OAT FOOD- avena sativa solution
ONION- allium cepa solution
CUCUMBER- cucumis sativus solution
BLUEBERRY- vaccinium myrtilloides solution
GINGER- zingiber officinale solution
WHITE SEEDLESS GRAPE- vitis vinifera solution
TOMATO- solanum lycopersicum solution
GARLIC- allium sativum solution
SOYBEAN FOOD- glycine max solution
SPINACH- spinacia oleracea solution
GREEN PEPPER- piper nigrum solution
PINEAPPLE- ananas comosus solution
STRAWBERRY- fragaria x ananassa solution
YELLOW SUMMER SQUASH- cucurbita pepo var. ovifera solution
BRAZIL NUT- bertholletia excelsa solution
CRANBERRY- vaccinium macrocarpon solution
GREEN OLIVE- olea europaea solution
CHERRY- prunus avium solution
LIMA BEAN- phaseolus lunatus solution
HOPS FOOD- humulus lupulus solution
NUTMEG- myristica fragrans solution
PECAN FOOD- carya illinoinensis solution
SWEET POTATO- ipomoea batatas solution
WHITE POTATO- solanum tuberosum solution
RED RASPBERRY- rubus idaeus solution
ORANGE- citrus x sinensis solution
BLACK WALNUT FOOD- juglans nigra solution
WATERMELON- citrullus lanatus solution
GRAPEFRUIT- citrus x paradisi solution
PEACH- prunus persica solution
PEANUT- arachis hypogaea solution
GREEN ENGLISH PEA- pisum sativum solution
PEAR- pyrus communis solution
BEEF- bos taurus solution
CHICKEN MEAT- gallus gallus solution
CHICKEN EGG WHITE- gallus gallus solution
CHICKEN EGG YOLK- gallus gallus solution
CHICKEN EGG WHOLE- gallus gallus solution
LAMB- ovis aries solution
PORK- sus scrofa solution
TURKEY MEAT- meleagris gallopavo solution
COW MILK- bos taurus solution
BLACK BASS- centropristis striata solution
CATFISH- ictalurus punctatus solution
CLAM- mercenaria mercenaria solution
ATLANTIC COD- gadus morhua solution
CRAB- callinectes sapidus solution
FISH MIX- gadus morhua, paralichthys lethostigma, hippoglossus hippoglossus, scomber scombrus and thunnus albacares solution
SHELLFISH MIX- mercenaria mercenaria, callinectes sapidus, crassostrea virginica, placopecten magellanicus and farfantepenaeus aztecus solution
FLOUNDER- paralichthys lethostigma solution
MAINE LOBSTER- homarus americanus solution
MACKEREL- scomber scombrus solution
OYSTER- crassostrea virginica solution
OCEAN PERCH- sebastes alutus solution
SALMON- salmo salar solution
SCALLOPS- placopecten magellanicus solution
SHRIMP- farfantepenaeus aztecus solution
LAKE TROUT- oncorhynchus mykiss solution
TUNA- thunnus albacares solution
ALMOND FOOD- prunus dulcis solution
APPLE- malus pumila solution
ENGLISH WALNUT- juglans regia solution
Greer Laboratories, Inc.
----------
Non Standardized Allergenic Extracts
Pollens, Molds, Epidermals, Insects, Dusts, Foods, and Miscellaneous Inhalants
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use Non-Standardized Allergenic Extracts (Pollens, Molds, Epidermals, Insects, Foods and Miscellaneous Inhalants) safely and effectively. See full prescribing information for Non-Standardized Allergenic Extracts.
Non-Standardized Allergenic Extracts (Pollens, Molds, Epidermals, Insects, Foods, and Miscellaneous Inhalants)
Solutions for percutaneous, intradermal or subcutaneous administration.
Initial U.S. Approval: 1968
|
WARNING: SEVERE ALLERGIC REACTIONS See full prescribing information for complete boxed warning.
|
INDICATIONS AND USAGE
Non-Standardized Allergenic Extracts are indicated for:
- Skin test diagnosis of patients with a clinical history of allergies to one or more of the specific allergens. (1)
- Immunotherapy for reduction of allergen-induced allergic symptoms confirmed by appropriate positive skin tests or in vitro testing for allergen-specific IgE antibodies. (1)
Food extracts have not been proven safe or effective in allergen immunotherapy.
DOSAGE AND ADMINISTRATION
For percutaneous, intradermal or subcutaneous use only.
The extracts are diluted with sterile diluents when used for intradermal testing or subcutaneous immunotherapy. For percutaneous testing, the extracts are diluted with sterile diluents in patients expected to be at greater risk for systemic allergic reaction. Dosages vary by mode of administration and by individual response. See full prescribing information for instructions on preparation, administration, and adjustments of dose. (2.1)
DOSAGE FORMS AND STRENGTHS
Non-Standardized Allergenic Extracts are labeled in weight/volume and/or protein nitrogen units (PNU)/milliliter (a measure of total protein), and are supplied as sterile aqueous stock concentrates at up to 1:10 weight/volume or 40,000 PNU/milliliter, or 50% glycerin stock concentrates at up to 1:20 weight/volume. (3)
CONTRAINDICATIONS
- Severe, unstable or uncontrolled asthma. (4)
- History of any severe systemic or local allergic reaction to an allergen extract. (4)
WARNINGS AND PRECAUTIONS
Severe allergic reactions have occurred following the administration of other allergenic extracts and may occur in individuals following the administration of Non-Standardized Allergenic Extracts in the following situations:
- Extreme sensitivity to Non-Standardized Allergenic Extracts, receipt of high doses of Non-Standardized Allergenic Extracts, or concomitant exposure to similar environmental allergens. (5.1)
- Receiving an accelerated immunotherapy build-up schedule (e.g., “rush” immunotherapy), or changing from one allergenic lot to another. (5.1)
ADVERSE REACTIONS
The most common adverse reactions, occurring in 26 to 82% of all patients who receive subcutaneous immunotherapy, are local adverse reactions at the injection site (e.g., erythema, itching, swelling, tenderness, pain). (6)
Systemic adverse reactions, occurring in ≤ 7% of patients who receive subcutaneous immunotherapy, include generalized skin erythema, urticaria, pruritus, angioedema, rhinitis, wheezing, laryngeal edema, and hypotension.
These can be fatal. (6)
To report SUSPECTED ADVERSE REACTIONS, contact GREER Laboratories, Inc. at 1-855-274-1322 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- Antihistamines and other medications that suppress histamine, including topical corticosteroids, topical anesthetics and tricyclic antidepressants can interfere with skin test results. (7)
See 17 for PATIENT COUNSELING INFORMATION
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: SEVERE ALLERGIC REACTIONS
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Preparation for Administration
2.2 Diagnostic Testing
2.3 Immunotherapy
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Serious Systemic Adverse Reactions
5.2 Epinephrine
5.3 Cross-Reactions and Dose Sensitivity
6 ADVERSE REACTIONS
7 DRUG INTERACTIONS
7.1 Antihistamines
7.2 Topical Corticosteroids and Topical Anesthetics
7.3 Tricyclic Antidepressants
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
14 CLINICAL STUDIES
15 REFERENCES
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage and Handling
17 PATIENT COUNSELING INFORMATION
*Sections or subsections omitted from the Full Prescribing Information are not listed.
FULL PRESCRIBING INFORMATION
WARNING: SEVERE ALLERGIC REACTIONS
- Non-Standardized Allergenic Extracts can cause severe life-threatening systemic reactions, including anaphylaxis. (5.1)
- Do not administer these products to patients with severe, unstable or uncontrolled asthma. (4)
- Observe patients in the office for at least 30 minutes following treatment. Emergency measures and personnel trained in their use must be available immediately in the event of a life-threatening reaction. (5.1)
- Patients with extreme sensitivity to these products, those on an accelerated immunotherapy build-up schedule, those switching to another allergenic lot, those receiving high doses of Non-Standardized Allergenic Extracts, or those also exposed to similar allergens may be at increased risk of a severe allergic reaction. (5.1)
- These products may not be suitable for patients with certain underlying medical conditions that may reduce their ability to survive a serious allergic reaction. (5.1)
- These products may not be suitable for patients who may be unresponsive to epinephrine or inhaled bronchodilators, such as those taking beta-blockers. (5.2)
1 INDICATIONS AND USAGE
Non-Standardized Allergenic Extracts are indicated for:
- Skin test diagnosis of patients with a clinical history of allergies to one or more of the specific non-standardized allergens.
- Immunotherapy for the reduction of allergen-induced allergic symptoms confirmed by appropriate positive skin tests or by in vitro testing for allergen-specific IgE antibodies.
Food extracts have not been proven safe or effective in allergen immunotherapy.
2 DOSAGE AND ADMINISTRATION
For percutaneous, intradermal or subcutaneous use only.
The extracts are diluted with sterile diluents when used for intradermal testing or subcutaneous immunotherapy. For percutaneous testing, the extracts are diluted with sterile diluents in patients expected to be at greater risk for systemic allergic reaction. Dosages vary by mode of administration and by individual response.
2.1 Preparation for Administration
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Discard solution if either of these conditions exist.
The extracts are diluted with sterile diluents when used for percutaneous and intradermal testing, or for subcutaneous immunotherapy.
Extracts labeled “For Diagnostic Use Only” are intended for percutaneous and intradermal testing only. These extracts have not been shown by adequate data to be safe and effective for therapeutic use. The extracts labeled For Diagnostic Use Only are the foods Barley, Coffee, Oat, Pineapple, Rye, Spinach, Wheat, the insects Flea, House Fly, Mosquito, and the plant and plant parts Cottonseed and Flax.
Undiluted 50% glycerin stock concentrate is used for percutaneous testing. To prepare 10-fold dilutions for percutaneous testing in patients suspected to be at greater risk for systemic allergic reaction, start with the stock concentrate. Proceed as shown in Table 1. The 10-fold dilution series uses 0.5 milliliters of concentrate added to 4.5 milliliters of sterile 50% glycerin diluent. Subsequent dilutions are made in a similar manner.
To prepare 10-fold dilutions for intradermal testing and immunotherapy, start with a 1:10 weight/volume, 1:20 weight/volume, or up to a 40,000 PNU/milliliter stock concentrate. Proceed as shown in Table 1. The 10-fold dilution series uses 0.5 milliliter of concentrate added to 4.5 milliliters of sterile diluent (glycerin-free diluents for intradermal testing, glycerin-free or 10% glycerin-saline diluents for immunotherapy). Subsequent dilutions are made in a similar manner.
| Dilution | Extract | Milliliters of Diluent | Dilution Strength (w/v) | Dilution Strength (w/v) | Dilution Strength (PNU/milliliter) |
| 0 | Concentrate | 1:10 | 1:20 | 20,000 | |
| 1 | 0.5 mL Concentrate | 4.5 | 1:100 | 1:200 | 2,000 |
| 2 | 0.5 mL Dilution 1 | 4.5 | 1:1,000 | 1:2,000 | 200 |
| 3 | 0.5 mL Dilution 2 | 4.5 | 1:10,000 | 1:20,000 | 20 |
| 4 | 0.5 mL Dilution 3 | 4.5 | 1:100,000 | 1:200,000 | 2 |
| 5 | 0.5 mL Dilution 4 | 4.5 | 1:1,000,000 | 1:2,000,000 | 0.2 |
| 6 | 0.5 mL Dilution 5 | 4.5 | 1:10,000,000 | 1:20,000,000 | 0.02 |
*There is no direct potency correlation across the table between PNU/milliliter and w/v.
Undiluted 50% glycerin stock concentrate is used for percutaneous testing. To prepare 5-fold dilutions for percutaneous testing in patients suspected to be at greater risk for systemic allergic reaction, start with the stock concentrate. Proceed as shown in Table 2. The 5-fold dilution series uses 1 milliliter of concentrate added to 4 milliliters of sterile 50% glycerin diluent. Subsequent dilutions are made in a similar manner.
To prepare 5-fold dilutions for intradermal testing or immunotherapy, start with a 1:10 weight/volume, 1:20 weight/volume, or up to a 40,000 PNU/milliliter stock concentrate. Proceed as shown in Table 2. The 5-fold dilution series uses 1 milliliter of concentrate added to 4 milliliters of sterile diluent (glycerin-free diluents for intradermal testing, glycerin-free or 10% glycerin-saline diluents for immunotherapy). Subsequent dilutions are made in a similar manner.
| Dilution | Extract | Milliliters of Diluent | Dilution Strength (w/v) | Dilution Strength (w/v) | Dilution Strength (PNU/milliliter) |
| 0 | Concentrate | 1:10 | 1:20 | 20,000 | |
| 1 | 1 mL Concentrate | 4 | 1:50 | 1:100 | 4,000 |
| 2 | 1 mL Dilution 1 | 4 | 1:250 | 1:500 | 800 |
| 3 | 1 mL Dilution 2 | 4 | 1:1,250 | 1:2,500 | 160 |
| 4 | 1 mL Dilution 3 | 4 | 1:6,250 | 1:12,500 | 32 |
| 5 | 1 mL Dilution 4 | 4 | 1:31,250 | 1:62,500 | 6.4 |
| 6 | 1 mL Dilution 5 | 4 | 1:156,250 | 1:312,500 | 1.28 |
*There is no direct potency correlation across the table between PNU/milliliter and w/v.
2.2 Diagnostic Testing
Diagnostic testing can be performed via percutaneous or intradermal administration of the Non-Standardized Allergenic Extracts. A positive skin test reaction should be interpreted in relation to the patient’s history and known exposure to the specific allergen(s).
Percutaneous Skin Testing
Preparation and Dose
For percutaneous testing (prick or puncture), use glycerinated extract; use the extracts at the highest available stock concentration. In patients suspected to be at greater risk for systemic allergic reaction, use 10-fold or 5-fold dilutions of the concentrate.
Prick test: Place one drop of extract with appropriate controls on the skin and with a skin test device, pierce through the drop into the skin with a slight lifting motion. Alternatively, use skin test devices loaded with the extract from the storage trays in a similar manner or in accordance with the device manufacturer’s recommendations.
Puncture test: Place one drop of extract or control on the skin and pierce the skin through the drop with a skin test device perpendicular to the skin. Alternatively, use skin test devices loaded with the extract from the storage trays in a similar manner or in accordance with the device manufacturer’s recommendations.
Interpreting Results
When using percutaneous skin test devices, follow the directions provided with the test devices. A glycerinated histamine control solution (6 milligrams/milliliter or 1 milligram/milliliter histamine base) may be used as the positive control. A 50% glycerin-saline solution may be used as the negative control.
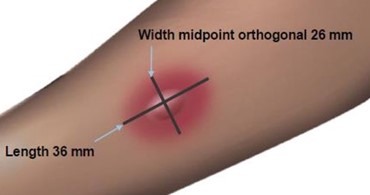
Read and record skin test responses 15 to 20 minutes after exposure. Individual patient reactivity can vary with time, allergen potency, and/or immunotherapy, as well as testing technique. The most reliable method of recording a skin test reaction is to measure the largest diameter of both wheal and erythema. While some correlation exists between the size of the skin test reaction and the degree of sensitivity, other factors should be considered in the diagnosis of allergy to specific allergens (see Figure 1 below).
Figure 1: Measurement of Wheal and Flare
Use a paper or plastic millimeter skin reaction guide as shown below.

Fifteen minutes after application of the skin test, measure the length and midpoint orthogonal width of each flare and wheal from the inner edge of the reaction.

The length of the skin test is defined as the largest diameter and the width of the skin test is defined as the diameter perpendicular to the length at its midpoint. Consider the wheal and flare as separate entities. First, measure the flare and then independently measure the wheal.
Measuring the Flare
Measuring the Wheal
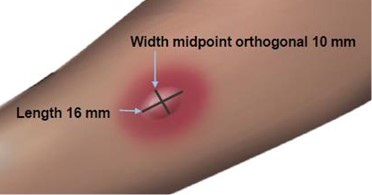
The average diameter measurement in the example above of the flare is (26 mm + 36 mm)/2 = 31 mm and the average diameter of the wheal is (10 mm + 16 mm)/2 = 13 mm.
Responses to positive controls should be at least 3 millimeters larger than responses to the negative controls.
Negative controls should elicit no reaction or only reactions of small diameter (less than 2 millimeters wheal, less than 5 millimeters erythema).
If either the positive or negative control response does not meet the above criteria, results for the allergenic extracts tested at the same time should be considered invalid and be repeated.
Intradermal Skin Testing
Preparation and Dose
For intradermal testing, dilute stock concentrate to 1:100 to 1:1000 volume to volume of Non-Standardized Allergenic Extracts stock concentrate solution. Dilute the stock concentrate solution with sterile diluent [see Dosage and Administration (2.1)]. Use normal or buffered saline or normal saline with human serum albumin (HSA) diluent. If the result from the initial test dose is negative, subsequent intradermal tests using increasingly stronger doses may be performed up to the maximum recommended strength of 1:25 volume to volume dilution of the extract concentrate solution.
Inject 0.02 milliliters of the extract solution intradermally according to the algorithms shown in Figure 2.
Figure 2: Algorithm for Dilution of Stock Concentrate Solution of Non-Standardized Allergenic Extracts for Intradermal Skin Testing
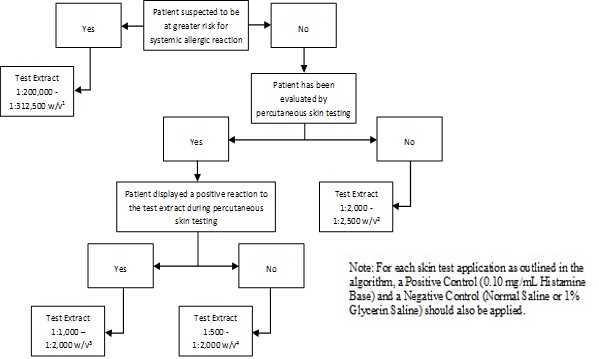
1 Corresponds to 1:10,000 - 1:15,625 volume to volume dilution of 1:20 weight/volume glycerinated extract concentrates
2 Corresponds to 1:100 - 1:125 volume to volume dilution of 1:20 weight/volume glycerinated extract concentrates
3 Corresponds to 1:50 - 1:100 volume to volume dilution of 1:20 weight/volume glycerinated extract concentrates
4 Corresponds to 1:25 - 1:100 volume to volume dilution of 1:20 weight/volume glycerinated extract concentrates
2.3 Immunotherapy
For subcutaneous administration only.
Preparation and Dose
Stock concentrates of Non-Standardized Allergenic Extracts are available in aqueous (up to 1:10 weight/volume or 40,000 PNU/milliliter) and 50% glycerin (up to 1:20 weight/volume) strengths for immunotherapy. Stock concentrates are diluted in normal saline, buffered saline, HSA-saline, or 10% glycerin-saline, depending on the patient’s reactivity to the diluent. See Table 1 and Table 2 for dilution preparation.
Administration of Immunotherapy
Administer immunotherapy by subcutaneous injection in the lateral aspect of the upper arm or thigh. Avoid injection directly into any blood vessel.
The optimal interval between doses of allergenic extract varies among individuals. Injections are usually given 1 to 2 times per week until the maintenance dose is reached, at which time the injection interval is increased to 2, then 3, and finally 4 weeks. Dosages vary by mode of administration, and by clinical response and tolerance. The minimum course of treatment may be three to five years, depending on the clinical response.
Guidelines for Immunotherapy
The initial dose of the extract should be based on the skin test reactivity. In patients suspected to be at greater risk for systemic allergic reaction by history and skin test, the initial dose of the extract should be 0.05 milliliter of a 1:20,000,000 to 1:2,000,000 weight/volume extract dilution. Patients not suspected to be at greater risk for systemic allergic reaction may be started at 0.1 milliliter of a 1:200,000 to 1:20,000 weight/volume extract dilution.
The dose of allergenic extract is increased at each injection by no more than 50% of the previous dose, and the next increment is governed by the response to the last injection.
Select the maximum tolerated maintenance dose based on the patient’s clinical response and tolerance. Doses larger than 0.2 milliliter of the stock concentrate are rarely administered because an extract in 50% glycerin diluent can cause discomfort upon injection.
Dosage Modification Guidelines for Immunotherapy
The following conditions may indicate a need to withhold or reduce the dosage of immunotherapy.
- Symptoms of rhinitis and/or asthma
- Infection accompanied by fever
- Exposure to excessive amounts of clinically relevant environmental allergen prior to a scheduled injection
- Large local reactions that persist for longer than 24 hours can be an indication for repeating the previous dose or reducing the dose at the next administration
Any evidence of a systemic reaction is an indication for a significant reduction (at least 75%) in the subsequent dose. Repeated systemic adverse reactions are sufficient reason for the cessation of further attempts to increase the dose.
Local adverse reactions require a decrease in the next dose by at least 50%. Proceed cautiously in subsequent dosing. In situations prompting dose reduction, once the reduced dose is tolerated, a cautious increase in dosage can be attempted.
Changing extract to a different lot or from a different manufacturer: When switching to a different lot of extract, or from another manufacturer’s extract, decrease the starting dose. Because manufacturing processes and sources of raw materials differ among manufacturers, the interchangeability of extracts from different manufacturers cannot be assured. In general, a dose reduction of 50 to 75% of the previous dose should be adequate, but each situation must be evaluated separately, considering the patient’s history of sensitivity, tolerance of previous injections, and other factors. Dose intervals should not exceed one week when rebuilding the dose.
Unscheduled gaps between treatments: Patients can lose tolerance to allergen injections during prolonged periods between doses, which increases their risk for an adverse reaction. The duration of tolerance between injections varies from patient to patient.
During the build-up phase, when patients receive injections 1 to 2 times per week, repeat or reduce the extract dosage if there
has been a substantial time interval between injections. This depends on: 1) the concentration of allergen immunotherapy extract that is to be administered; 2) a previous history of systemic reactions; and 3) the degree of variation from the prescribed interval of time, with longer intervals since the last injection leading to greater reductions in the dose to be administered.
This suggested approach to dose modification, due to unscheduled gaps between treatments during the build-up phase, is not based on published evidence. The individual physician should use this or a similar protocol for the specific clinical setting.
Similarly, if unscheduled gaps occur during maintenance therapy, it may be necessary to reduce the dosage and bring the patient up to maintenance dosing using an established build-up protocol.
Changing from non-stabilized to human serum albumin (HSA) stabilized diluents: Allergenic extracts prepared with diluents containing HSA and 0.4% phenol are more stable than those prepared with diluents that do not contain stabilizers. When switching from a non-stabilized to an HSA-stabilized diluent, consider lowering the dose for immunotherapy.
3 DOSAGE FORMS AND STRENGTHS
Non-Standardized Allergenic Extracts are labeled in weight/volume and/or protein nitrogen units (PNU)/milliliter (a measure of total protein), and are supplied as sterile aqueous stock concentrates at up to 1:10 weight/volume or 40,000 PNU/milliliter, or 50% glycerin stock concentrates at up to 1:20 weight/volume.
4 CONTRAINDICATIONS
Non-Standardized Allergenic Extracts are contraindicated in patients with:
- Severe, unstable or uncontrolled asthma
- History of any severe systemic or local allergic reaction to an allergen extract
5 WARNINGS AND PRECAUTIONS
5.1 Serious Systemic Adverse Reactions
Serious systemic adverse reactions have occurred following the administration of other allergenic extracts and may occur in individuals following the administration of Non-Standardized Allergenic Extracts in the following situations:
- Extreme sensitivity to the specific allergen(s)
- Receipt of an accelerated immunotherapy build-up schedule (e.g., “rush” immunotherapy)
- Receipt of high doses of allergenic extracts or concomitant exposure to similar environmental allergens
- Change from one allergenic extract lot to another allergenic extract lot
High-risk patients have had fatal reactions. Consider using more dilute preparations in patients suspected to be at greater risk of systemic allergic reaction [see Dosage and Administration (2.1)].
Administer Non-Standardized Allergenic Extracts in a healthcare setting under the supervision of a physician prepared to manage a severe systemic or a severe local allergic reaction.Observe patients in the office for at least 30 minutes following administration. 1
5.2 Epinephrine
Non-Standardized Allergenic Extracts may not be suitable for patients with certain medical conditions that may reduce the ability to survive a serious allergic reaction or increase the risk of adverse reactions after epinephrine administration. Examples of these medical conditions include but are not limited to: markedly compromised lung function (either chronic or acute), unstable angina, recent myocardial infarction, significant arrhythmia, and uncontrolled hypertension.
These products may not be suitable for patients who are taking medications that can potentiate or inhibit the effect of epinephrine. These medications include:
Βeta-adrenergic blockers: Patients taking beta-adrenergic blockers may be unresponsive to the usual doses of epinephrine used to treat serious systemic reactions, including anaphylaxis. Specifically, beta-adrenergic blockers antagonize the cardiostimulating and bronchodilating effects of epinephrine.
Alpha-adrenergic blockers, ergot alkaloids: Patients taking alpha-adrenergic blockers may be unresponsive to the usual doses of epinephrine used to treat serious systemic reactions, including anaphylaxis. Specifically, alpha-adrenergic blockers antagonize the vasoconstricting and hypertensive effects of epinephrine. Similarly, ergot alkaloids may reverse the pressor effects of epinephrine.
Tricyclic antidepressants, levothyroxine sodium, monoamine oxidase inhibitors, and certain antihistamines: The adverse effects of epinephrine may be potentiated in patients taking tricyclic antidepressants, levothyroxine sodium, monoamine oxidase inhibitors, and the antihistamines chlorpheniramine, and diphenhydramine.
Cardiac glycosides, diuretics: Patients who receive epinephrine while taking cardiac glycosides or diuretics should be observed carefully for the development of cardiac arrhythmias.
5.3 Cross-Reactions and Dose Sensitivity
When determining the final dose of an allergen mixture for immunotherapy, consider cross-reactivity among component extracts.
Determine the initial dilution of allergenic extract, starting dose, and progression of dosage based on the patient’s history and results of skin tests 2 [see Dosage and Administration (2.1)]. Strongly positive skin tests can be indicators for potential adverse reactions.
6 ADVERSE REACTIONS
The most common adverse reactions, occurring in 26 to 82% of all patients who receive subcutaneous immunotherapy are local adverse reactions at the injection site (e.g., erythema, itching, swelling, tenderness, pain). 1 Systemic adverse reactions, occurring in ≤ 7% of patients who receive subcutaneous immunotherapy, 3 include generalized skin erythema, urticaria, pruritus, angioedema, rhinitis, wheezing, laryngeal edema, and hypotension. These adverse reactions can be fatal. 1
The allergenic extracts labeled “For Diagnostic Use Only” that contain sodium formaldehyde sulfoxylate (SFS) can cause slight discoloration of the skin at the site of administration. This discoloration can remain for extended amounts of time.
7 DRUG INTERACTIONS
7.1 Antihistamines
Do not perform skin testing with allergenic extracts within 3 to 10 days of use of first-generation H1-histamine receptor blockers (e.g., clemastine, diphenhydramine) and second-generation antihistamines (e.g., loratadine, cetirizine). These products suppress histamine skin test reactions and could mask a positive response. 2
7.2 Topical Corticosteroids and Topical Anesthetics
Topical corticosteroids can suppress skin reactivity; therefore, discontinue use at the skin test site for 2 to 3 weeks before skin testing. Avoid use of topical local anesthetics at skin test sites as they can suppress flare responses. 2
7.3 Tricyclic Antidepressants
Tricyclic antidepressants can have potent antihistamine effects that can affect skin testing. If tricyclic medication has been recently discontinued, allow 7 to 14 days before initiating skin testing. 2
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
All pregnancies have a risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively. There are no human or animal data to establish the presence or absence of Non-Standardized Allergenic Extracts-associated risks during pregnancy.
8.2 Lactation
Risk Summary
It is not known whether Non-Standardized Allergenic Extracts are present in human milk. Data are not available to assess the effects of these extracts on the breastfed child or on milk production/excretion. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Non-Standardized Allergenic Extracts and any potential adverse effects on the breastfed child from the extracts or from the underlying maternal condition.
8.4 Pediatric Use
For use of these products in children younger than 5 years of age, consideration should be given to the patient’s ability to comply and cooperate with allergen immunotherapy and the potential for difficulty in communicating with the child regarding systemic reactions. 1
8.5 Geriatric Use
Data are not available to determine if subjects 65 years of age and older respond differently to allergen immunotherapy than younger subjects.
11 DESCRIPTION
Non-Standardized Allergenic Extracts are sterile solutions used for percutaneous testing, intradermal testing, or subcutaneous immunotherapy. Aqueous extracts contain the soluble extractants of the source material in water for injection, 0.5% sodium chloride, 0.54% sodium bicarbonate, and 0.4% phenol. Glycerinated extracts contain the soluable extractants of the source material in water for injection and 50% glycerin, 0.25% sodium chloride, 0.27% sodium bicarbonate, and 0.2% phenol. The pH of the extracts range from 6 to 9.
Certain food extracts (Barley, Oat, Pineapple, Rye, Spinach, and Wheat), labeled “For Diagnostic Use Only”, contain 0.1% sodium formaldehyde sulfoxylate as an antioxidant.
Source materials used in the manufacture of allergenic extracts are collected from natural sources or from laboratory cultures.
Non-Standardized Allergenic Extracts appear as clear and colorless to dark brown solutions that should be free of particulate matter.
Extracts are labeled either as weight-to-volume based on the weight of the source material to the volume of the extracting fluid, or as PNU/milliliter with one PNU representing 0.00001 mg of protein nitrogen per milliliter.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The skin test reaction results from interaction of the introduced allergen and allergen-specific IgE antibodies bound to mast cells, leading to mast cell degranulation and release of histamine, tryptase and other mediators, which results in the formation of the wheal and flare.
The precise mechanisms of action of allergen immunotherapy are not known. Immunologic responses to immunotherapy include changes in allergen-specific IgE levels, allergen-specific IgG levels, and regulatory T cell responses. 1
14 CLINICAL STUDIES
Specific immunotherapy with allergenic extracts is helpful in reducing symptoms associated with exposure to the offending allergens. A summary of effectiveness by the Panel on Review of Allergenic Extracts, an advisory committee to the U.S. Food and Drug Administration, has been published. 4
15 REFERENCES
- Cox LJ, Nelson H, Lockey R.Allergen immunotherapy: A practice parameter third update. J Allergy Clin Immunol. 2011;127:(1)S1-55.
- Bernstein IL, Li JT, Bernstein DI, et al.Allergy diagnostic testing: an updated practice parameter. Ann Allergy Asthma Immunol. 2008;100:S1-148.
- Greenberg MA, Kaufman CR, Gonzalez GE, et al. Late and immediate systemic-allergic reactions to inhalant allergen immunotherapy. J Allergy Clin Immunol. 1986;77:865-870.
- Federal Register Proposed Rule: Biological Products: Implementation of Efficacy Review, Allergenic Extracts, Federal Register 1985;50: 3082-3288.
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
Non-Standardized Allergenic Extracts and mixes may be supplied as aqueous stock concentrates of up to 1:10 weight/volume or 40,000 PNU/milliliter for intradermal and subcutaneous testing. The extracts may also be supplied as 50% glycerin stock concentrates of up to 1:20 weight/volume for use in percutaneous skin testing and subcutaneous immunotherapy. Non-Standardized Allergenic Extracts are labeled in weight/volume and/or PNU/milliliter and may be provided in 5, 10, and 50 milliliter vials. Glycerinated extracts are also supplied in 5 milliliter dropper vials for prick or puncture testing.
Non-Standardized Allergenic Extracts available are as follows:
| Pollens ‑ Grasses |
| Bahia Grass, Paspalum notatum |
| Brome, Smooth, Bromus inermis |
| Canarygrass, Reed, Phalaris arundinacea |
| Johnson Grass, Sorghum halepense |
| Quack (Couch) Grass, Elymus repens |
| Ryegrass, Giant Wild, Leymus condensatus |
| Velvetgrass, Holcus lanatus |
| Wheatgrass, Western, Pascopyrum smithii |
| Pollens - Trees |
| Acacia, Acacia dealbata |
| Alder, Hazel, Alnus serrulata |
| Alder, Red, Alnus rubra |
| Alder, White, Alnus rhombifolia |
| Ash, Arizona (Velvet), Fraxinus velutina |
| Ash, Green, Fraxinus pennsylvanica |
| Ash Mix (Equal parts Fraxinus pennsylvanica, Fraxinus americana) |
| Ash, Oregon, Fraxinus latifolia |
| Ash, White, Fraxinus americana |
| Aspen, Populus tremuloides |
| Beech, American, Fagus grandifolia |
| Birch, Black-Sweet, Betula lenta |
| Birch, Mix (Equal parts Betula lenta, Betula nigra, Betula populifolia) |
| Birch, River, Betula nigra |
| Birch, Spring, Betula occidentalis |
| Birch, White, Betula populifolia |
| Box Elder, Acer negundo |
| Cedar, Mountain, Juniperus ashei |
| Cedar, Red, Juniperus virginiana |
| Cedar, Salt (Tamarisk), Tamarix gallica |
| Central/Eastern 4 Tree Mix (Equal parts Ulmus americana, Acer negundo, Carya illinoinensis, Quercus virginiana) |
| Cottonwood, Arizona (Fremont), Populus fremontii |
| Cottonwood, Black, Populus trichocarpa |
| Cottonwood, Eastern, Populus deltoides |
| Cottonwood, Western, Populus deltoides ssp. monilifera |
| Cypress, Arizona, Callitropsis arizonica |
| Cypress, Bald, Taxodium distichum |
| Eastern Oak Mix (Equal parts Quercus velutina, Quercus rubra, Quercus alba) |
| Eastern 6 Tree Mix (Equal parts Fagus grandifolia, Populus deltoides, Quercus rubra, Betula nigra, Carya ovata, Fraxinus americana) |
| Eastern 7 Tree Mix (Equal parts Ulmus americana, Fagus grandifolia, Populus deltoides, Quercus rubra, Betula nigra, Carya ovata, Fraxinus americana) |
| Eastern 8 Tree Mix (Equal parts Ulmus americana, Fagus grandifolia, Populus deltoides, Quercus rubra, Betula nigra, Carya ovata, Fraxinus americana, Acer saccharum) |
| Eastern 10 Tree Mix (Equal parts Platanus occidentalis, Ulmus americana, Fagus grandifolia, Populus deltoides, Quercus rubra, Betula nigra, Carya ovata, Fraxinus americana, Acer saccharum, Liquidambar styraciflua) |
| Elm, American, Ulmus americana |
| Elm, Cedar, Ulmus crassifolia |
| Elm Mix (Equal parts Ulmus americana, Ulmus pumila) |
| Elm, Siberian, Ulmus pumila |
| Eucalyptus, Bluegum, Eucalyptus globulus |
| Hackberry, Celtis occidentalis |
| Hazelnut, American, Corylus americana |
| Hickory Mix (Equal parts Carya glabra, Carya ovata, Carya laciniosa, Carya tomentosa) |
| Hickory-Pecan Mix (Equal parts Carya illinoinensis, Carya ovata) |
| Hickory, Shagbark, Carya ovata |
| Hickory, Shellbark, Carya laciniosa |
| Hickory, White, Carya tomentosa |
| Juniper Mix (Equal parts Juniperus monosperma, Juniperus scopulorum) |
| Juniper, Oneseed, Juniperus monosperma |
| Juniper, Pinchot, Juniperus pinchotii |
| Juniper, Rocky Mountain, Juniperus scopulorum |
| Juniper, Utah, Juniperus osteosperma |
| Juniper, Western, Juniperus occidentalis |
| Locust Blossom, Black, Robinia pseudoacacia |
| Mango Blossom, Mangifera indica |
| Maple-Box Elder Mix (Equal parts Acer saccharum, Acer negundo) |
| 2 Maple Mix (Equal parts Acer rubrum, Acer saccharum) |
| 3 Maple Mix (Equal parts Acer rubrum, Acer saccharinum, Acer saccharum) |
| Maple, Red, Acer rubrum |
| Maple, Silver/Soft, Acer saccharinum |
| Maple, Sugar/Hard, Acer saccharum |
| Melaleuca, Melaleuca quinquenervia |
| Mesquite, Velvet Prosopis velutina |
| Mulberry, Paper, Broussonetia papyrifera |
| Mulberry, Red, Morus rubra |
| Mulberry, White, Morus alba |
| Oak, Arizona (Gambel), Quercus gambelii |
| Oak, Black, Quercus velutina |
| Oak, Bur, Quercus macrocarpa |
| Oak, California Black, Quercus kelloggii |
| Oak, California Live, Quercus agrifolia |
| Oak, California White, Quercus lobata |
| Oak, Post, Quercus stellata |
| Oak, Red, Quercus rubra |
| Oak, Virginia Live, Quercus virginiana |
| Oak, Water, Quercus nigra |
| Oak, Western White, Quercus garryana |
| Oak, White, Quercus alba |
| Olive, Olea europaea |
| Olive, Russian, Elaeagnus angustifolia |
| Orange Pollen, Citrus X sinensis |
| Palm, Queen, Syagrus romanzoffiana |
| Pecan, Carya illinoinensis |
| Peppertree Mix (Equal parts Schinus molle, Schinus terebinthifolius) |
| Pine, Australian (Beefwood), Casuarina equisetifolia |
| Pine, Loblolly, Pinus taeda |
| Pine, Longleaf, Pinus palustris |
| Pine Mix (Equal parts Pinus taeda, Pinus strobus, Pinus echinata) |
| Pine, Ponderosa, Pinus ponderosa |
| Pine, Virginia Scrub, Pinus virginiana |
| Pine, White (Eastern), Pinus strobus |
| Pine, White (Western), Pinus monticola |
| Pine, Yellow, Pinus echinata |
| Poplar, Lombardy’s, Populus nigra |
| Poplar, White, Populus alba |
| Privet, Ligustrum vulgare |
| Sweetgum, Liquidambar styraciflua |
| Sycamore, American, Platanus occidentalis |
| Sycamore, California (Western), Platanus racemosa |
| 11 Tree Mix (Equal parts Fagus grandifolia, Platanus occidentalis, Ulmus americana, Juglans nigra, Salix nigra, Populus deltoides,Quercus rubra, Betula nigra, Carya ovata, Acer saccharum, Fraxinus americana) |
| Walnut, Black, Juglans nigra |
| Walnut, California Black, Juglans californica |
| Walnut, English, Juglans regia |
| Wax Myrtle, Morella cerifera |
| Western Oak Mix (Equal parts Quercus kelloggii, Quercus agrifolia, Quercus garryana) |
| Western 3 Tree Mix (Equal parts Olea europaea, Ulmus pumila, Platanus racemosa) |
| Western 10 Tree Mix (Equal parts Acacia dealbata, Acer negundo, Populus fremontii, Olea europaea, Ulmus pumila, Betula occidentalis, Juniperus occidentalis, Platanus racemosa, Quercus garryana, Morus alba) |
| Western Walnut Mix (Equal parts Juglans californica, Juglans regia) |
| Willow, Arroyo, Salix lasiolepis |
| Willow, Black, Salix nigra |
| Pollens - Weeds and Garden Plants |
| Allscale, Atriplex polycarpa |
| Amaranth, Green, Amaranthus hybridus |
| Baccharis Mix (Equal parts Baccharis sarothroides, Baccharis halimifolia) |
| Burningbush (Kochia), Kochia scoparia spp. scoparia |
| Burrobrush, Ambrosia salsola |
| Central/Western Weed Mix (Equal parts Kochia scoparia ssp. scoparia, Chenopodium album, Salsola kali) |
| Cocklebur, Xanthium strumarium |
| Common Weed Mix (Equal parts Xanthium strumarium, Plantago lanceolata, Chenopodium album, Amaranthus retroflexus, Salsola kali) |
| Dock-Sorrel Mix (Equal parts Rumex acetosella, Rumex crispus) |
| Dock, Yellow (Curly), Rumex crispus |
| Dogfennel, Eupatorium capillifolium |
| Goldenrod, Solidago canadensis |
| Iodinebush, Allenrolfea occidentalis |
| Lamb's Quarters, Chenopodium album |
| Lenscale (Quailbrush), Atriplex lentiformis |
| Marsh Elder, True (Rough), Iva annua |
| Marshelder, Burweed (Giant Poverty), Cyclachaena xanthiifolia |
| Mixed Amaranths (Equal parts Amaranthus hybridus, Amaranthus palmeri, Amaranthus retroflexus) |
| Mugwort, Common, Artemisia vulgaris |
| National Weed Mix (Equal parts Xanthium strumarium, Ambrosia trifida, Chenopodium album, Amaranthus retroflexus, Ambrosia artemisiifolia) |
| Nettle, Urtica dioica |
| Palmer's Amaranth, Amaranthus palmeri |
| Pigweed, Rough Redroot, Amaranthus retroflexus |
| Pigweed, Spiny, Amaranthus spinosus |
| Plantain, English, Plantago lanceolata |
| Plantain-Sorrel Mix (Equal parts Plantago lanceolata, Rumex acetosella) |
| Rabbit Bush, Ambrosia deltoidea |
| Ragweed, Desert, Ambrosia dumosa |
| Ragweed, False, Ambrosia acanthicarpa |
| Ragweed, Giant (Tall), Ambrosia trifida |
| Ragweed, Lanceleaf, Ambrosia bidentata |
| Ragweed, Slender, Ambrosia confertiflora |
| Ragweed, Western, Ambrosia psilostachya |
| Russian Thistle, Salsola kali |
| Sagebrush, Common, Artemisia tridentata |
| Sage Mix (Equal parts Artemisia tridentata, Artemisia ludoviciana) |
| Sage, Prairie, Artemisia ludoviciana |
| Saltbush, Annual, Atriplex wrightii |
| Scale/Atriplex Mix (Equal parts Atriplex polycarpa, Atriplex lentiformis, Atriplex canescens) |
| Sorrel, Sheep (Red), Rumex acetosella |
| Waterhemp, Tall, Amaranthus tuberculatus |
| 3 Weed Mix (Equal parts Xanthium strumarium, Chenopodium album, Amaranthus retroflexus) |
| Western Ragweed Mix (Equal parts Ambrosia acanthicarpa, Ambrosia psilostachya) |
| Wingscale, Atriplex canescens |
| Plants and Plant Parts |
| Cotton Linters, Gossypium hirsutum |
| Cottonseed, Gossypium hirsutum (For Diagnostic Use Only) |
| Flax, Linum usitatissimum (For Diagnostic Use Only) |
| Gum, Arabic, Acacia senegal |
| Gum, Karaya, Sterculia urens |
| Gum, Tragacanth, Astragalus gummifer |
| Kapok, Ceiba pentandra |
| Orris Root, Iris germanica |
| Pyrethrum, Chrysanthemum cinerariifolium |
| Tobacco, Cultivated, Leaf, Nicotiana tabacum |
| Pollens - Cultivated Farm Plants |
| Alfalfa, Medicago sativa |
| Beet, Sugar, Beta vulgaris |
| Corn, Cultivated, Zea mays |
| Oat, Cultivated, Avena sativa |
| Rape (Mustard), Brassica napus |
| Red Clover, Trifolium pratense |
| Rye, Cultivated, Secale cereale |
| Wheat, Cultivated, Triticum aestivum |
| Pollens - Flowers |
| Daisy, Leucanthemum vulgare |
| Dandelion, Taraxacum officinale |
| Sunflower, Helianthus annuus |
| Molds, Rusts and Smuts |
| AHH Mold Mix (Equal parts Alternaria alternata, Bipolaris sorokiniana, Cladosporium sphaerospermum) |
| Alternaria alternata |
| Alternaria/Hormodendrum Mix (Equal parts Alternaria alternata, Cladosporium sphaerospermum) |
| Aspergillus amstelodami |
| Aspergillus flavus |
| Aspergillus fumigatus |
| Aspergillus Mix (Equal parts Aspergillus amstelodami, Aspergillus flavus, Aspergillus fumigatus, Aspergillus nidulans, Aspergillus niger) |
| Aspergillus nidulans |
| Aspergillus niger |
| Aureobasidium pullulans |
| Bermuda Grass Smut, Ustilago cynodontis |
| Bipolaris sorokiniana |
| Botrytis cinerea |
| Candida albicans |
| Chaetomium globosum |
| Cladosporium herbarum |
| Cladosporium sphaerospermum |
| Corn Smut, Ustilago maydis |
| Curvularia spicifera |
| Dematiaceae Mix (Equal parts Alternaria alternata, Aureobasidium pullulans, Bipolaris sorokiniana, Cladosporium herbarum, Curvularia spicifera, Helminthosporium solani) |
| Epicoccum nigrum |
| Epidermophyton floccosum |
| Fusarium Mix (Equal parts Gibberella fujikuroi, Fusarium solani) |
| Fusarium solani |
| Geotrichum candidum |
| Gibberella fujikuroi |
| Gliocladium viride |
| Grain Smut Mix (Equal parts Ustilago maydis, Ustilago tritici, Ustilago nuda, Ustilago avenae) |
| Grass Smut Mix (Equal parts Ustilago cynodontis, Sporisorium cruentum) |
| Helminthosporium solani |
| Hypomyces perniciousus |
| Loose Kernel Smut, Sporisorium cruentum |
| Loose Smut, Wheat, Ustilago tritici |
| Microsporum canis |
| Mold Mix #1 (Equal parts Alternaria alternata, Aspergillus niger, Bipolaris sorokiniana, Cladosporium sphaerospermum, Penicillium chrysogenum var. chrysogenum) |
| Mold Mix #2 (Equal parts Aureobasidium pullulans, Curvularia spicifera, Gibberella fujikuroi, Mucor plumbeus, Rhizopus stolonifer) |
| Mold Mix #3 (Equal parts Alternaria alternata, Aspergillus niger, Cladosporium sphaerospermum, Penicillium chrysogenum var. chrysogenum) |
| Monilia Mix (Equal parts Candida albicans, Neurospora intermedia) |
| Mucor circinelloides f. circinelloides |
| Mucor circinelloides f. lusitanicus |
| Mucor Mix (Equal parts Mucor circinelloides f. lusitanicus, Mucor plumbeus) |
| Mucor plumbeus |
| Neurospora intermedia |
| New Stock Fungi Mix (Equal parts Sarocladium strictum, Alternaria alternata, Aspergillus niger, Aureobasidium pullulans, Bipolaris sorokiniana, Botrytis cinerea, Candida albicans, Chaetomium globosum, Cladosporium sphaerospermum, Epicoccum nigrum, Gibberella fujikuroi, Mucor plumbeus, Penicillium chrysogenum var. chrysogenum, Phoma betae, Rhizopus stolonifer, Trichophyton mentagrophytes) |
| Oat Smut, Ustilago avenae |
| Paecilomyces variotii |
| Penicillium chrysogenum var. chrysogenum |
| Penicillium digitatum |
| Penicillium Mix (Equal parts Penicillium camemberti, Penicillium chrysogenum, Penicillium digitatum, Penicillium chrysogenum var. chrysogenum, Penicillium roqueforti) |
| Phoma betae |
| Phycomycetes Mix (Equal parts Mucor circinelloides f. lusitanicus, Rhizopus stolonifer) |
| Rhizopus arrhizus |
| Rhizopus Mix (Equal parts Rhizopus stolonifer, Rhizopus arrhizus) |
| Rhizopus stolonifer |
| Rhodotorula mucilaginosa |
| Saccharomyces cerevisiae |
| Sarocladium strictum |
| Stemphylium solani |
| Trichoderma harzianum |
| Trichophyton mentagrophytes |
| Trichophyton rubrum |
| Trichothecium roseum |
| Animal Allergens |
| Canary Feathers, Serinus canaria |
| Cattle Epithelia, Bos taurus |
| Chicken Feathers, Gallus gallus |
| Dog Epithelia, Canis lupus familiaris |
| Duck Feathers, Anas platyrhynchos |
| Gerbil Epithelia, Meriones unguiculatus |
| Goat Epithelia, Capra hircus |
| Goose Feathers, Anser anser |
| Guinea Pig Epithelia, Cavia porcellus |
| Hamster Epithelia, Mesocricetus auratus |
| Hog Epithelia, Sus scrofa |
| Horse Epithelia, Equus caballus |
| Mixed Feathers (Equal parts Gallus gallus, Anas platyrhynchos, Anser anser) |
| Mouse Epithelia, Mus musculus |
| Parakeet Feathers, Melopsittacus undulatus |
| Rabbit Epithelia, Oryctolagus cuniculus |
| Rat Epithelia, Rattus norvegicus |
| Silk Worm Cocoon, Bombyx mori |
| Insects (Whole Body) |
| Ant, Black Carpenter, Camponotus pennsylvanicus |
| Ant, Fire, Solenopsis invicta |
| Ant, Fire, Solenopsis richteri |
| Cockroach, American, Periplaneta americana |
| Cockroach, German, Blattella germanica |
| 2 Cockroach Mix (Equal parts Periplaneta americana, Blattella germanica) |
| Deer Fly, Chrysops vittatus |
| Flea, Ctenocephalis felis (For Dagnostic Use Only) |
| House Fly, Musca domestica (For Dagnostic Use Only) |
| Mosquito, Aedes taeniorhynchus (For Diagnostic Use Only) |
| Food - Animal Products and Poultry Products |
| Beef, Bos taurus |
| Chicken Meat, Gallus gallus |
| Egg, White, Chicken, Gallus gallus |
| Egg, Whole, Chicken, Gallus gallus |
| Egg, Yolk, Chicken, Gallus gallus |
| Lamb, Ovis aries |
| Pork, Sus scrofa |
| Turkey Meat, Meleagris gallopavo |
| Food - Dairy Products |
| Milk, Cow, Bos taurus |
| Food - Fish and Shellfish |
| Bass, Black, Centropristis striata |
| Catfish, Channel, Ictalurus punctatus |
| Clam, Northern Quahog, Mercenaria mercenaria |
| Cod, Atlantic, Gadus morhua |
| Crab, Blue, Callinectes sapidus |
| Fish Mix (Equal parts Gadus morhua, Paralichthys lethostigma, Hippoglossus hippoglossus, Scomber scombrus, Thunnus albacares) |
| Flounder, Southern, Paralichthys lethostigma |
| Lobster, American, Homarus americanus |
| Mackerel, King/Atlantic, Scomber scombrus |
| Oyster, Atlantic/Eastern, Crassostrea virginica |
| Perch, Ocean, Sebastes alutus |
| Salmon, Atlantic, Salmo salar |
| Scallops, Sea, Placopecten magellanicus |
| Shellfish Mix (Equal parts Mercenaria mercenaria, Callinectes sapidus, Crassostrea virginica, Placopecten magellanicus, Farfantepenaeus aztecus) |
| Shrimp, Brown, Farfantepenaeus aztecus |
| Trout, Rainbow, Oncorhynchus mykiss |
| Tuna, Yellowfin, Thunnus albacares |
| Food - Plant Source |
| Almond, Prunus dulcis |
| Apple, Malus pumila |
| Apricot, Prunus armeniaca |
| Barley, Whole Grain, Hordeum vulgare (For Diagnostic Use Only, Contains SFS*) |
| Bean, Lima, Phaseolus lunatus |
| Bean, Navy, Phaseolus vulgaris |
| Bean, String Green, Phaseolus vulgaris |
| Blueberry, Velvetleaf, Vaccinium myrtilloides |
| Brazil Nut, Bertholletia excelsa |
| Broccoli, Brassica oleracea var. botrytis |
| Buckwheat, Fagopyrum esculentum |
| Cabbage, Brassica oleracea var. capitata |
| Cacao Bean, Theobroma cacao |
| Cantaloupe, Cucumis melo |
| Carrot, Daucus carota |
| Cashew Nut, Anacardium occidentale |
| Cauliflower, Brassica oleracea var. botrytis |
| Celery, Apium graveolens var. dulce |
| Cherry, Sweet, Prunus avium |
| Cinnamon, Cinnamomum verum |
| Coconut, Cocos nucifera |
| Coffee, Coffea arabica (For Diagnostic Use Only) |
| Corn, Zea mays |
| Cranberry, Vaccinium macrocarpon |
| Cucumber, Cucumis sativus |
| Garlic, Allium sativum |
| Ginger, Zingiber officinale |
| Grape, White Seedless, Vitis vinifera |
| Grapefruit, Citrus X paradisi |
| Hazelnut (Filbert), Corylus americana |
| Hops, Humulus lupulus |
| Lemon, Citrus X limon |
| Lettuce, Lactuca sativa |
| Malt (Barley), Hordeum vulgare |
| Mushroom, Agaricus campestris |
| Mustard Seed, Sinapis alba |
| Nutmeg, Myristica fragrans |
| Oat, Avena sativa (For Diagnostic Use Only, Contains SFS*) |
| Olive, Green, Olea europaea |
| Onion, Allium cepa |
| Orange, Citrus X sinensis |
| Pea, Green or English, Pisum sativum |
| Peach, Prunus persica |
| Peanut, Arachis hypogaea |
| Pear, Pyrus communis |
| Pecan, Carya illinoinensis |
| Pepper, Black, Piper nigrum |
| Pepper, Green, Capsicum annuum |
| Pineapple, Ananas comosus (For Diagnostic Use Only, Contains SFS*) |
| Potato, Sweet, Ipomoea batatas |
| Potato, White, Solanum tuberosum |
| Raspberry, Red, Rubus idaeus |
| Rice, Oryza sativa |
| Rye, Secale cereale (For Diagnostic Use Only, Contains SFS*) |
| Sesame Seed, Sesamum indicum |
| Soybean, Glycine max |
| Spinach, Spinacia oleracea (For Diagnostic Use Only, Contains SFS*) |
| Squash, Yellow Summer, Cucurbita pepo var. ovifera |
| Strawberry, Fragaria X ananassa |
| Tomato, Solanum lycopersicum |
| Vanilla, Vanilla planifolia |
| Walnut, Black, Juglans nigra |
| Walnut, English, Juglans regia |
| Watermelon, Citrullus lanatus |
| Wheat, Whole, Triticum aestivum (For Diagnostic Use Only, Contains SFS*) |
*SFS – Sodium Formadehyde Sulfoxylate
16.2 Storage and Handling
Maintain at 2 to 8°C (36 to 46°F) during storage and use.
Dilutions of concentrated extracts that result in a glycerin content of less than 50% can reduce extract stability. Extract dilutions at 1:100 volume to volume should be kept no longer than a month, and more dilute solutions no more than a week. The potency of a dilution can be checked by skin test comparison to a fresh dilution of the extract on a known allergic patient.
17 PATIENT COUNSELING INFORMATION
Instruct patient to remain under observation in the office for 30 minutes or longer after an injection.
Caution patient that reactions can occur more than 30 minutes after skin testing or an injection.
Instruct patient to recognize the following symptoms as adverse reactions and to immediately return to the office or immediately seek other medical attention if any of these symptoms occur following skin testing or an injection:
- Unusual swelling and/or tenderness at the injection site
- Hives or itching of the skin
- Swelling of the face and/or mouth
- Sneezing, coughing or wheezing
- Shortness of breath
- Nausea
- Dizziness or faintness
Manufacturer:
U.S. License No. 308
Greer Laboratories, Inc.
Lenoir, NC 28645 U.S.A
| RICE FOOD
oryza sativa solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| RYE FOOD
secale cereale solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| SESAME SEED
sesamum indicum solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| BROCCOLI
brassica oleracea var. botrytis solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| BUCKWHEAT
fagopyrum esculentum solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| CASHEW NUT
anacardium occidentale solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| CAULIFLOWER
brassica oleracea var. botrytis solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| CHOCOLATE CACAO BEAN
theobroma cacao solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| CANTALOUPE MUSKMELON
cucumis melo solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| CARROT
daucus carota solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| CELERY
apium graveolens var. dulce solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| LETTUCE
lactuca sativa solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| FILBERT HAZELNUT FOOD
corylus americana solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| VANILLA
vanilla planifolia solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| WHOLE WHEAT
triticum aestivum solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| APRICOT
prunus armeniaca solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| WHOLE GRAIN BARLEY
hordeum vulgare solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| NAVY BEAN
phaseolus vulgaris solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| STRING GREEN BEAN
phaseolus vulgaris solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| CABBAGE
brassica oleracea var. capitata solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| CINNAMON
cinnamomum verum solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| COFFEE
coffea arabica solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| MUSHROOM
agaricus campestris solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| MUSTARD FOOD
sinapis alba solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| BANANA
musa acuminata solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| COCONUT
cocos nucifera solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| CORN FOOD
zea mays solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| MALT
hordeum vulgare solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| BLACK PEPPER
piper nigrum solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| LEMON
citrus x limon solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| OAT FOOD
avena sativa solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| ONION
allium cepa solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| CUCUMBER
cucumis sativus solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| BLUEBERRY
vaccinium myrtilloides solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| GINGER
zingiber officinale solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| WHITE SEEDLESS GRAPE
vitis vinifera solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| TOMATO
solanum lycopersicum solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| GARLIC
allium sativum solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| SOYBEAN FOOD
glycine max solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| SPINACH
spinacia oleracea solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| GREEN PEPPER
piper nigrum solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| PINEAPPLE
ananas comosus solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| STRAWBERRY
fragaria x ananassa solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| YELLOW SUMMER SQUASH
cucurbita pepo var. ovifera solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| BRAZIL NUT
bertholletia excelsa solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| CRANBERRY
vaccinium macrocarpon solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| GREEN OLIVE
olea europaea solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| CHERRY
prunus avium solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| LIMA BEAN
phaseolus lunatus solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| HOPS FOOD
humulus lupulus solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| NUTMEG
myristica fragrans solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| PECAN FOOD
carya illinoinensis solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| SWEET POTATO
ipomoea batatas solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| WHITE POTATO
solanum tuberosum solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| RED RASPBERRY
rubus idaeus solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| ORANGE
citrus x sinensis solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| BLACK WALNUT FOOD
juglans nigra solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| WATERMELON
citrullus lanatus solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| GRAPEFRUIT
citrus x paradisi solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| PEACH
prunus persica solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| PEANUT
arachis hypogaea solution |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| GREEN ENGLISH PEA
pisum sativum solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| PEAR
pyrus communis solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| BEEF
bos taurus solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| CHICKEN MEAT
gallus gallus solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| CHICKEN EGG WHITE
gallus gallus solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| CHICKEN EGG YOLK
gallus gallus solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| CHICKEN EGG WHOLE
gallus gallus solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| LAMB
ovis aries solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| PORK
sus scrofa solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| TURKEY MEAT
meleagris gallopavo solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| COW MILK
bos taurus solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| BLACK BASS
centropristis striata solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| CATFISH
ictalurus punctatus solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| CLAM
mercenaria mercenaria solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| ATLANTIC COD
gadus morhua solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| CRAB
callinectes sapidus solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| FISH MIX
gadus morhua, paralichthys lethostigma, hippoglossus hippoglossus, scomber scombrus and thunnus albacares solution |
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
| SHELLFISH MIX
mercenaria mercenaria, callinectes sapidus, crassostrea virginica, placopecten magellanicus and farfantepenaeus aztecus solution |
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
| FLOUNDER
paralichthys lethostigma solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| MAINE LOBSTER
homarus americanus solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| MACKEREL
scomber scombrus solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| OYSTER
crassostrea virginica solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| OCEAN PERCH
sebastes alutus solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| SALMON
salmo salar solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| SCALLOPS
placopecten magellanicus solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| SHRIMP
farfantepenaeus aztecus solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| LAKE TROUT
oncorhynchus mykiss solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| TUNA
thunnus albacares solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| ALMOND FOOD
prunus dulcis solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| APPLE
malus pumila solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| ENGLISH WALNUT
juglans regia solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Greer Laboratories, Inc. (024671414) |
| Registrant - Greer Laboratories, Inc. (024671414) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Greer Laboratories, Inc. | 024671414 | manufacture(22840-5746, 22840-5747, 22840-5748, 22840-5708, 22840-5709, 22840-5783, 22840-5713, 22840-5716, 22840-5711, 22840-5712, 22840-5714, 22840-5728, 22840-5785, 22840-5754, 22840-5756, 22840-5701, 22840-5703, 22840-5705, 22840-5706, 22840-5710, 22840-5717, 22840-5718, 22840-5730, 22840-5731, 22840-5702, 22840-5784, 22840-5719, 22840-5729, 22840-5740, 22840-5727, 22840-5733, 22840-5735, 22840-5721, 22840-5707, 22840-5723, 22840-5724, 22840-5753, 22840-5722, 22840-5749, 22840-5750, 22840-5741, 22840-5742, 22840-5752, 22840-5751, 22840-5782, 22840-5720, 22840-5734, 22840-5715, 22840-5704, 22840-5726, 22840-5732, 22840-5787, 22840-5743, 22840-5744, 22840-5745, 22840-5736, 22840-5788, 22840-5755, 22840-5725, 22840-5738, 22840-5786, 22840-5737, 22840-5739, 22840-5757, 22840-5760, 22840-5761, 22840-5763, 22840-5762, 22840-5758, 22840-5759, 22840-5764, 22840-5765, 22840-5766, 22840-5767, 22840-5768, 22840-5769, 22840-5770, 22840-9700, 22840-9701, 22840-5771, 22840-5772, 22840-5773, 22840-5774, 22840-5775, 22840-5776, 22840-5777, 22840-5778, 22840-5779, 22840-5780, 22840-5781, 22840-5700, 22840-5789) | |

