Label: CABERGOLINE tablet
- NDC Code(s): 0093-5420-88
- Packager: Teva Pharmaceuticals USA, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated July 31, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Cabergoline tablets USP contain cabergoline, USP, a dopamine receptor agonist. The chemical name for cabergoline, USP is 1-[(6-allylergolin-8β-yl)-carbonyl]-1-[3-(dimethylamino)propyl]-3-ethylurea and has the following structural formula:
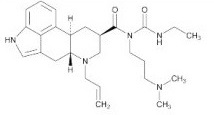
C26H37N5O2 M.W. 451.6
Cabergoline, USP is a white powder soluble in ethyl alcohol, chloroform, and N, N-dimethylformamide (DMF); slightly soluble in 0.1N hydrochloric acid; very slightly soluble in n-hexane; and insoluble in water.
Each cabergoline tablet USP, for oral administration, contains 0.5 mg of cabergoline, USP and has the following inactive ingredients: anhydrous lactose and leucine.
-
CLINICAL PHARMACOLOGY
Mechanism of Action
The secretion of prolactin by the anterior pituitary is mainly under hypothalamic inhibitory control, likely exerted through release of dopamine by tuberoinfundibular neurons. Cabergoline is a long-acting dopamine receptor agonist with a high affinity for D2 receptors. Results of in vitro studies demonstrate that cabergoline exerts a direct inhibitory effect on the secretion of prolactin by rat pituitary lactotrophs. Cabergoline decreased serum prolactin levels in reserpinized rats. Receptor-binding studies indicate that cabergoline has low affinity for dopamine D1, α1- and α2-adrenergic, and 5-HT1- and 5-HT2-serotonin receptors.
Clinical Studies
The prolactin-lowering efficacy of cabergoline was demonstrated in hyperprolactinemic women in two randomized, double-blind, comparative studies, one with placebo and the other with bromocriptine. In the placebo-controlled study (placebo n = 20; cabergoline n = 168), cabergoline produced a dose-related decrease in serum prolactin levels with prolactin normalized after 4 weeks of treatment in 29%, 76%, 74% and 95% of the patients receiving 0.125, 0.5, 0.75, and 1 mg twice weekly respectively.
In the 8-week, double-blind period of the comparative trial with bromocriptine (cabergoline n = 223; bromocriptine n = 236 in the intent-to-treat analysis), prolactin was normalized in 77% of the patients treated with cabergoline at 0.5 mg twice weekly compared with 59% of those treated with bromocriptine at 2.5 mg twice daily. Restoration of menses occurred in 77% of the women treated with cabergoline, compared with 70% of those treated with bromocriptine. Among patients with galactorrhea, this symptom disappeared in 73% of those treated with cabergoline compared with 56% of those treated with bromocriptine.
Pharmacokinetics
Absorption
Following single oral doses of 0.5 mg to 1.5 mg given to 12 healthy adult volunteers, mean peak plasma levels of 30 to 70 picograms (pg)/mL of cabergoline were observed within 2 to 3 hours. Over the 0.5 to 7 mg dose range, cabergoline plasma levels appeared to be dose-proportional in 12 healthy adult volunteers and nine adult parkinsonian patients. A repeat-dose study in 12 healthy volunteers suggests that steady-state levels following a once-weekly dosing schedule are expected to be two-fold to three-fold higher than after a single dose. The absolute bioavailability of cabergoline is unknown. A significant fraction of the administered dose undergoes a first-pass effect. The elimination half-life of cabergoline estimated from urinary data of 12 healthy subjects ranged between 63 to 69 hours. The prolonged prolactin-lowering effect of cabergoline may be related to its slow elimination and long half-life.
Distribution
In animals, based on total radioactivity, cabergoline (and/or its metabolites) has shown extensive tissue distribution. Radioactivity in the pituitary exceeded that in plasma by > 100-fold and was eliminated with a half-life of approximately 60 hours. This finding is consistent with the long-lasting prolactin-lowering effect of the drug. Whole body autoradiography studies in pregnant rats showed no fetal uptake but high levels in the uterine wall. Significant radioactivity (parent plus metabolites) detected in the milk of lactating rats suggests a potential for exposure to nursing infants. The drug is extensively distributed throughout the body. Cabergoline is moderately bound (40% to 42%) to human plasma proteins in a concentration-independent manner. Concomitant dosing of highly protein-bound drugs is unlikely to affect its disposition.
Metabolism
In both animals and humans, cabergoline is extensively metabolized, predominately via hydrolysis of the acylurea bond or the urea moiety. Cytochrome P-450 mediated metabolism appears to be minimal. Cabergoline does not cause enzyme induction and/or inhibition in the rat. Hydrolysis of the acylurea or urea moiety abolishes the prolactin-lowering effect of cabergoline, and major metabolites identified thus far do not contribute to the therapeutic effect.
Excretion
After oral dosing of radioactive cabergoline to five healthy volunteers, approximately 22% and 60% of the dose was excreted within 20 days in the urine and feces, respectively. Less than 4% of the dose was excreted unchanged in the urine. Nonrenal and renal clearances for cabergoline are about 3.2 L/min and 0.08 L/min, respectively. Urinary excretion in hyperprolactinemic patients was similar.
Special Populations
Renal Insufficiency
The pharmacokinetics of cabergoline were not altered in 12 patients with moderate-to-severe renal insufficiency as assessed by creatinine clearance.
Hepatic Insufficiency
In 12 patients with mild-to-moderate hepatic dysfunction (Child-Pugh score ≤ 10), no effect on mean cabergoline Cmax or area under the plasma concentration curve (AUC) was observed. However, patients with severe insufficiency (Child-Pugh score > 10) show a substantial increase in the mean cabergoline Cmax and AUC, and thus necessitate caution.
Pharmacodynamics
Dose response with inhibition of plasma prolactin, onset of maximal effect, and duration of effect has been documented following single cabergoline doses to healthy volunteers (0.05 to 1.5 mg) and hyperprolactinemic patients (0.3 to 1 mg). In volunteers, prolactin inhibition was evident at doses > 0.2 mg, while doses ≥ 0.5 mg caused maximal suppression in most subjects. Higher doses produce prolactin suppression in a greater proportion of subjects and with an earlier onset and longer duration of action. In 12 healthy volunteers, 0.5, 1, and 1.5 mg doses resulted in complete prolactin inhibition, with a maximum effect within 3 hours in 92% to 100% of subjects after the 1 and 1.5 mg doses compared with 50% of subjects after the 0.5 mg dose.
In hyperprolactinemic patients (N = 51), the maximal prolactin decrease after a 0.6 mg single dose of cabergoline was comparable to 2.5 mg bromocriptine; however, the duration of effect was markedly longer (14 days vs. 24 hours). The time to maximal effect was shorter for bromocriptine than cabergoline (6 hours vs. 48 hours).
In 72 healthy volunteers, single or multiple doses (up to 2 mg) of cabergoline resulted in selective inhibition of prolactin with no apparent effect on other anterior pituitary hormones (GH, FSH, LH, ACTH, and TSH) or cortisol.
- INDICATIONS AND USAGE
-
CONTRAINDICATIONS
Cabergoline tablets are contraindicated in patients with:
- Uncontrolled hypertension or known hypersensitivity to ergot derivatives.
- History of cardiac valvular disorders, as suggested by anatomical evidence of valvulopathy of any valve, determined by pre-treatment evaluation including echocardiographic demonstration of valve leaflet thickening, valve restriction, or mixed valve restriction-stenosis (see WARNINGS).
- History of pulmonary, pericardial, or retroperitoneal fibrotic disorders (see WARNINGS).
-
WARNINGS
Pregnancy
Dopamine agonists in general should not be used in patients with pregnancy-induced hypertension, for example, preeclampsia, eclampsia, and postpartum hypertension, unless the potential benefit is judged to outweigh the possible risk.
Fibrotic Complications
Cardiac Valvulopathy
All patients should undergo a cardiovascular evaluation, including echocardiogram to assess the potential presence of valvular disease. If valvular disease is detected, the patient should not be treated with cabergoline (see CONTRAINDICATIONS). Postmarketing cases of cardiac valvulopathy have been reported in patients receiving cabergoline. These cases have generally occurred during administration of high doses of cabergoline (> 2 mg/day) for the treatment of Parkinson’s disease. Cases of cardiac valvulopathy have also been reported in patients receiving lower doses of cabergoline for the treatment of hyperprolactinemic disorders.
A multi-country, retrospective cohort study using general practice records and record linkage systems in the UK, Italy and the Netherlands was conducted to assess the association between new use of dopamine agonists including cabergoline (n = 27,812) for Parkinson’s disease and hyperprolactinemia and cardiac valvular regurgitation (CVR), other fibroses, and other cardiopulmonary events over a maximum of 12 years of follow up. In this study, the use of cabergoline among persons with Parkinson's disease was associated with an increased risk of CVR when compared to non-ergot-derived dopamine agonists (DAs) and levodopa [Incidence Rate (IR) per 10,000 person years of 68.1 (95% confidence interval (CI): 37.2 to 115.3) for cabergoline vs. 10 (95% CI: 5.2 to 19.4) for non-ergot DAs and 11.3 (95% CI: 7.2 to 17) for levodopa]. In the study analysis confined to persons with dopamine agonist-treated hyperprolactinemia (n = 8,386), when compared to non-use (n = 15,147), persons exposed to cabergoline did not have an elevated risk of CVR. The findings with respect to the risk of CVR associated with cabergoline treatment for persons with Parkinson’s disease (increased risk) and those with hyperprolactinemia (no increased risk) are consistent with the findings in other published studies.
Physicians should use the lowest effective dose of cabergoline for the treatment of hyperprolactinemic disorders and should periodically reassess the need for continuing therapy with cabergoline. Following treatment initiation, clinical and diagnostic monitoring (for example, chest x-ray, CT scan and cardiac echocardiogram) should be conducted to assess the risk of cardiac valvulopathy. The recommended frequency of routine echocardiographic monitoring is every 6 to 12 months or as clinically indicated with the presence of signs and symptoms such as edema, new cardiac murmur, dyspnea, or congestive heart failure.
Cabergoline should be discontinued if an echocardiogram reveals new valvular regurgitation, valvular restriction or valve leaflet thickening.
Cabergoline should be used with caution in patients exposed to other medications associated with valvulopathy.
Extracardiac Fibrotic Reactions
Postmarketing cases of pleural, pericardial, and retroperitoneal fibrosis have been reported following administration of cabergoline. Some reports were in patients previously treated with other ergotinic dopamine agonists. Cabergoline should not be used in patients with a history of cardiac or extracardiac fibrotic disorders.
Fibrotic disorders can have an insidious onset and patients should be monitored for manifestations of progressive fibrosis. Therefore, during treatment, attention should be paid to the signs and symptoms of:
- Pleuro-pulmonary disease such as dyspnea, shortness of breath, persistent cough or chest pain.
- Renal insufficiency or ureteral/abdominal vascular obstruction that may occur with pain in the loin/flank and lower limb edema as well as any possible abdominal masses or tenderness that may indicate retroperitoneal fibrosis.
- Cardiac failure: Cases of valvular and pericardial fibrosis have often manifested as cardiac failure. Therefore, valvular fibrosis (and constrictive pericarditis) should be excluded if such symptoms occur.
Clinical and diagnostic monitoring such as erythrocyte sedimentation rate, chest x-ray, serum creatinine measurements, and other investigations should be considered at baseline and as necessary while patients are treated with cabergoline.
Following diagnosis of pleural effusion or pulmonary fibrosis, the discontinuance of cabergoline was reported to result in improvement of signs and symptoms.
-
PRECAUTIONS
General
Initial doses higher than 1 mg may produce orthostatic hypotension. Care should be exercised when administering cabergoline with other medications known to lower blood pressure.
Postpartum Lactation Inhibition or Suppression
Cabergoline tablets are not indicated for the inhibition or suppression of physiologic lactation. Use of bromocriptine, another dopamine agonist for this purpose, has been associated with cases of hypertension, stroke, and seizures.
Hepatic Impairment
Since cabergoline is extensively metabolized by the liver, caution should be used, and careful monitoring exercised, when administering cabergoline to patients with hepatic impairment.
Psychiatric
Impulse control/compulsive behaviors, including pathological gambling, increased libido, and hypersexuality have been reported in patients treated with dopamine agonists including cabergoline. This has been generally reversible upon reduction of the dose or treatment discontinuation (see Postmarketing Surveillance Data). Prescribers should consider dose reduction or stopping the medication if a patient develops such urges while taking cabergoline.
Information for Patients
Patients should be instructed to notify their physician if they suspect they are pregnant, become pregnant, or intend to become pregnant during therapy. A pregnancy test should be done if there is any suspicion of pregnancy and continuation of treatment should be discussed with their physician.
Patients should notify their physician if they develop shortness of breath, persistent cough, difficulty with breathing when lying down, or swelling in their extremities.
Patients should be alerted to the possibility that patients may experience intense urges to spend money uncontrollably, intense urges to gamble, increased sexual urges, and other intense urges and the inability to control these urges while taking cabergoline. Advise patients to inform their healthcare provider if they develop new or increased uncontrolled spending, gambling urges, sexual urges, or other urges while being treated with cabergoline (see PRECAUTIONS).
Drug Interactions
Cabergoline should not be administered concurrently with D2-antagonists, such as phenothiazines, butyrophenones, thioxanthenes, or metoclopramide.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies were conducted in mice and rats with cabergoline given by gavage at doses up to 0.98 mg/kg/day and 0.32 mg/kg/day, respectively. These doses are 7 times and 4 times the maximum recommended human dose calculated on a body surface area basis using total mg/m2/week in rodents and mg/m2/week for a 50 kg human.
There was a slight increase in the incidence of cervical and uterine leiomyomas and uterine leiomyosarcomas in mice. In rats, there was a slight increase in malignant tumors of the cervix and uterus and interstitial cell adenomas. The occurrence of tumors in female rodents may be related to the prolonged suppression of prolactin secretion because prolactin is needed in rodents for the maintenance of the corpus luteum. In the absence of prolactin, the estrogen/progesterone ratio is increased, thereby increasing the risk for uterine tumors. In male rodents, the decrease in serum prolactin levels was associated with an increase in serum luteinizing hormone, which is thought to be a compensatory effect to maintain testicular steroid synthesis. Since these hormonal mechanisms are thought to be species-specific, the relevance of these tumors to humans is not known.
The mutagenic potential of cabergoline was evaluated and found to be negative in a battery of in vitro tests. These tests included the bacterial mutation (Ames) test with Salmonella typhimurium, the gene mutation assay with Schizosaccharomyces pombe P1 and V79 Chinese hamster cells, DNA damage and repair in Saccharomyces cerevisiae D4, and chromosomal aberrations in human lymphocytes. Cabergoline was also negative in the bone marrow micronucleus test in the mouse.
In female rats, a daily dose of 0.003 mg/kg for 2 weeks prior to mating and throughout the mating period inhibited conception. This dose represents approximately 1/28 the maximum recommended human dose calculated on a body surface area basis using total mg/m2/week in rats and mg/m2/week for a 50 kg human.
Pregnancy
Reproduction studies have been performed with cabergoline in mice, rats, and rabbits administered by gavage.
(Multiples of the maximum recommended human dose in this section are calculated on a body surface area basis using total mg/m2/week for animals and mg/m2/week for a 50 kg human.)
There were maternotoxic effects but no teratogenic effects in mice given cabergoline at doses up to 8 mg/kg/day (approximately 55 times the maximum recommended human dose) during the period of organogenesis.
A dose of 0.012 mg/kg/day (approximately 1/7 the maximum recommended human dose) during the period of organogenesis in rats caused an increase in post-implantation embryo-fetal losses. These losses could be due to the prolactin inhibitory properties of cabergoline in rats. At daily doses of 0.5 mg/kg/day (approximately 19 times the maximum recommended human dose) during the period of organogenesis in the rabbit, cabergoline caused maternotoxicity characterized by a loss of body weight and decreased food consumption. Doses of 4 mg/kg/day (approximately 150 times the maximum recommended human dose) during the period of organogenesis in the rabbit caused an increased occurrence of various malformations. However, in another study in rabbits, no treatment-related malformations or embryofetotoxicity were observed at doses up to 8 mg/kg/day (approximately 300 times the maximum recommended human dose).
In rats, doses higher than 0.003 mg/kg/day (approximately 1/28 the maximum recommended human dose) from 6 days before parturition and throughout the lactation period inhibited growth and caused death of offspring due to decreased milk secretion.
There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from cabergoline, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother. Use of cabergoline for the inhibition or suppression of physiologic lactation is not recommended (see PRECAUTIONS section).
The prolactin-lowering action of cabergoline suggests that it will interfere with lactation. Due to this interference with lactation, cabergoline should not be given to women postpartum who are breastfeeding or who are planning to breastfeed.
Pediatric Use
Safety and effectiveness of cabergoline in pediatric patients have not been established.
Geriatric Use
Clinical studies of cabergoline did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger patients. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
-
ADVERSE REACTIONS
The safety of cabergoline tablets has been evaluated in more than 900 patients with hyperprolactinemic disorders. Most adverse events were mild or moderate in severity.
In a 4-week, double-blind, placebo-controlled study, treatment consisted of placebo or cabergoline at fixed doses of 0.125, 0.5, 0.75, or 1 mg twice weekly. Doses were halved during the first week. Since a possible dose-related effect was observed for nausea only, the four cabergoline treatment groups have been combined. The incidence of the most common adverse events during the placebo-controlled study is presented in the following table.
Incidence of Reported Adverse Events During the 4-Week, Double-Blind, Placebo-Controlled Trial *Reported at ≥ 1% for cabergoline Adverse Event*
Cabergoline (n = 168)
0.125 to 1 mg two times a weekPlacebo (n = 20)
Number (percent)
Gastrointestinal
Nausea
45 (27)
4 (20)
Constipation
16 (10)
0
Abdominal pain
9 (5)
1 (5)
Dyspepsia
4 (2)
0
Vomiting
4 (2)
0
Central and Peripheral Nervous System
Headache
43 (26)
5 (25)
Dizziness
25 (15)
1 (5)
Paresthesia
2 (1)
0
Vertigo
2 (1)
0
Body As a Whole
Asthenia
15 (9)
2 (10)
Fatigue
12 (7)
0
Hot flashes
2 (1)
1 (5)
Psychiatric
Somnolence
9 (5)
1 (5)
Depression
5 (3)
1 (5)
Nervousness
4 (2)
0
Autonomic Nervous System
Postural hypotension
6 (4)
0
Reproductive – Female
Breast pain
2 (1)
0
Dysmenorrhea
2 (1)
0
Vision
Abnormal vision
2 (1)
0
In the 8-week, double-blind period of the comparative trial with bromocriptine, cabergoline (at a dose of 0.5 mg twice weekly) was discontinued because of an adverse event in 4 of 221 patients (2%) while bromocriptine (at a dose of 2.5 mg two times a day) was discontinued in 14 of 231 patients (6%). The most common reasons for discontinuation from cabergoline were headache, nausea and vomiting (3, 2 and 2 patients respectively); the most common reasons for discontinuation from bromocriptine were nausea, vomiting, headache, and dizziness or vertigo (10, 3, 3, and 3 patients respectively). The incidence of the most common adverse events during the double-blind portion of the comparative trial with bromocriptine is presented in the following table.Incidence of Reported Adverse Events During the 8-Week, Double-Blind Period of the Comparative Trial With Bromocriptine *Reported at ≥ 1% for cabergoline
Adverse Event*Cabergoline (n = 221)
Bromocriptine (n = 231)
Number (percent)
Gastrointestinal
Nausea
63 (29)
100 (43)
Constipation
15 (7)
21 (9)
Abdominal pain
12 (5)
19 (8)
Dyspepsia
11 (5)
16 (7)
Vomiting
9 (4)
16 (7)
Dry mouth
5 (2)
2 (1)
Diarrhea
4 (2)
7 (3)
Flatulence
4 (2)
3 (1)
Throat irritation
2 (1)
0
Toothache
2 (1)
0
Central and Peripheral Nervous System
Headache
58 (26)
62 (27)
Dizziness
38 (17)
42 (18)
Vertigo
9 (4)
10 (4)
Paresthesia
5 (2)
6 (3)
Body As a Whole
Asthenia
13 (6)
15 (6)
Fatigue
10 (5)
18 (8)
Syncope
3 (1)
3 (1)
Influenza-like symptoms
2 (1)
0
Malaise
2 (1)
0
Periorbital edema
2 (1)
2 (1)
Peripheral edema
2 (1)
1
Psychiatric
Depression
7 (3)
5 (2)
Somnolence
5 (2)
5 (2)
Anorexia
3 (1)
3 (1)
Anxiety
3 (1)
3 (1)
Insomnia
3 (1)
2 (1)
Impaired concentration
2 (1)
1
Nervousness
2 (1)
5 (2)
Cardiovascular
Hot flashes
6 (3)
3 (1)
Hypotension
3 (1)
4 (2)
Dependent edema
2 (1)
1
Palpitation
2 (1)
5 (2)
Reproductive – Female
Breast pain
5 (2)
8 (3)
Dysmenorrhea
2 (1)
1
Skin and Appendages
Acne
3 (1)
0
Pruritus
2 (1)
1
Musculoskeletal
Pain
4 (2)
6 (3)
Arthralgia
2 (1)
0
Respiratory
Rhinitis
2 (1)
9 (4)
Vision
Abnormal vision
2 (1)
2 (1)
Other adverse events that were reported at an incidence of < 1% in the overall clinical studies follow.Body As a Whole: Facial edema, influenza-like symptoms, malaise
Cardiovascular System: Hypotension, syncope, palpitations
Digestive System: Dry mouth, flatulence, diarrhea, anorexia
Metabolic and Nutritional System: Weight loss, weight gain
Nervous System: Somnolence, nervousness, paresthesia, insomnia, anxiety
Respiratory System: Nasal stuffiness, epistaxis
Skin and Appendages: Acne, pruritus
Special Senses: Abnormal vision
Urogenital System: Dysmenorrhea, increased libido
The safety of cabergoline has been evaluated in approximately 1,200 patients with Parkinson’s disease in controlled and uncontrolled studies at dosages of up to 11.5 mg/day which greatly exceeds the maximum recommended dosage of cabergoline for hyperprolactinemic disorders. In addition to the adverse events that occurred in the patients with hyperprolactinemic disorders, the most common adverse events in patients with Parkinson’s disease were dyskinesia, hallucinations, confusion, and peripheral edema. Heart failure, pleural effusion, pulmonary fibrosis, and gastric or duodenal ulcer occurred rarely. One case of constrictive pericarditis has been reported.
Postmarketing Surveillance Data
The following events have been reported in association with cabergoline: cardiac valvulopathy and extracardiac fibrotic reactions (see WARNINGS, Cardiac Valvulopathy and Extracardiac Fibrotic Reactions).
Other events have been reported in association with cabergoline: impulse control/compulsive behavior symptoms, including hypersexuality, increased libido and pathological gambling (see PRECAUTIONS, Psychiatric). In addition, cases of alopecia, aggression and psychotic disorder have been reported in patients taking cabergoline. Some of these reports have been in patients who have had prior adverse reactions to dopamine agonist products.
To report SUSPECTED ADVERSE REACTIONS, contact Teva Pharmaceuticals USA, Inc. at 1-888-838-2872 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch for voluntary reporting of adverse reactions.
- OVERDOSAGE
-
DOSAGE AND ADMINISTRATION
The recommended dosage of cabergoline tablets for initiation of therapy is 0.25 mg twice a week. Dosage may be increased by 0.25 mg twice weekly up to a dosage of 1 mg twice a week according to the patient’s serum prolactin level. Before initiating treatment, cardiovascular evaluation should be performed and echocardiography should be considered to assess for valvular disease.
Dosage increases should not occur more rapidly than every 4 weeks, so that the physician can assess the patient’s response to each dosage level. If the patient does not respond adequately, and no additional benefit is observed with higher doses, the lowest dose that achieved maximal response should be used and other therapeutic approaches considered. Patients receiving long-term treatment with cabergoline should undergo periodic assessment of their cardiac status and echocardiography should be considered.
After a normal serum prolactin level has been maintained for 6 months, cabergoline may be discontinued, with periodic monitoring of the serum prolactin level to determine whether or when treatment with cabergoline should be reinstituted. The durability of efficacy beyond 24 months of therapy with cabergoline has not been established.
-
HOW SUPPLIED
Cabergoline Tablets USP, 0.5 mg are available as white, oval-shaped, scored tablets, debossed “hourglass logo”, “0.5” with a score on one side and “5420” on the other side containing 0.5 mg cabergoline, USP packaged in unit-of-use bottles of 8 tablets (NDC 0093-5420-88).
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Dispense in original container.
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
Manufactured In Czech Republic By:
Teva Czech Industries s.r.o.
Opava-Komarov, Czech Republic
Manufactured For:
Teva Pharmaceuticals USA, Inc.
North Wales, PA 19454
Rev. G 7/2020
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CABERGOLINE
cabergoline tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0093-5420 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CABERGOLINE (UNII: LL60K9J05T) (CABERGOLINE - UNII:LL60K9J05T) CABERGOLINE 0.5 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) LEUCINE (UNII: GMW67QNF9C) Product Characteristics Color white Score 2 pieces Shape OVAL Size 8mm Flavor Imprint Code 0;5;5420 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0093-5420-88 1 in 1 CARTON 03/07/2007 1 8 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA077750 03/07/2007 Labeler - Teva Pharmaceuticals USA, Inc. (001627975)


