Label: IPRATROPIUM BROMIDE spray
- NDC Code(s): 0054-0046-41
- Packager: Hikma Pharmaceuticals USA Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated July 1, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
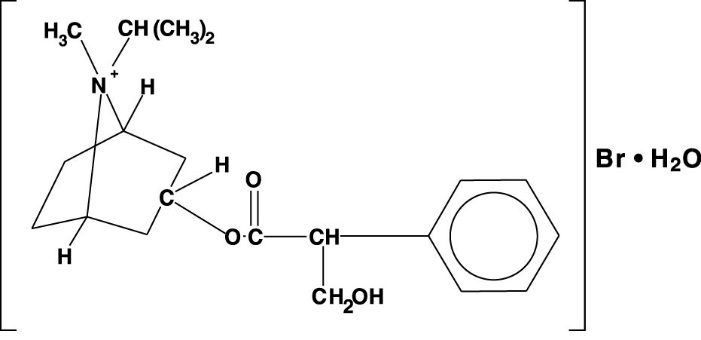
The active ingredient in Ipratropium Bromide Nasal Solution is ipratropium bromide (as the monohydrate). It is an anticholinergic agent chemically described as 8-azoniabicyclo [3.2.1] octane, 3-(3-hydroxy-1-oxo-2-phenylpropoxy)-8-methyl-8-(1-methylethyl)-, bromide monohydrate (3-endo, 8-syn)-: a synthetic quaternary ammonium compound, chemically related to atropine. The structural formula is:
ipratropium bromide C20H30BrNO3 • H2O Mol. Wt. 430.4
Ipratropium bromide is a white to off-white crystalline substance, freely soluble in water and methanol, sparingly soluble in ethanol, and insoluble in non-polar media. In aqueous solution, it exists in an ionized state as a quaternary ammonium compound.
Ipratropium Bromide Nasal Solution, 0.06% is a metered-dose, manual pump spray unit which delivers 42 mcg (70 mcL) ipratropium bromide per spray on an anhydrous basis in an isotonic, aqueous solution with pH adjusted to 4.7 with hydrochloric acid and/or sodium hydroxide (if needed). It also contains benzalkonium chloride, edetate disodium, purified water and sodium chloride. Each bottle contains 165 metered sprays.
-
CLINICAL PHARMACOLOGY
Mechanism of Action
Ipratropium bromide is an anticholinergic (para-sympatholytic) agent which, based on animal studies, appears to inhibit vagally-mediated reflexes by antagonizing the action of acetylcholine, the transmitter agent released at the neuromuscular junctions in the lung. In humans, ipratropium bromide has anti-secretory properties and, when applied locally, inhibits secretions from the serous and seromucous glands lining the nasal mucosa. Ipratropium bromide is a quaternary amine that minimally crosses the nasal and gastrointestinal membranes and the blood-brain barrier, resulting in a reduction of the systemic anticholinergic effects (e.g., neurologic, ophthalmic, cardiovascular, and gastrointestinal effects) that are seen with tertiary anticholinergic amines.
Pharmacokinetics
Absorption
Ipratropium bromide is poorly absorbed into the systemic circulation following oral administration (2 to 3%). Less than 20% of an 84 mcg per nostril dose was absorbed from the nasal mucosa of normal volunteers, induced-cold adult volunteers, naturally acquired common cold pediatric patients, or perennial rhinitis adult patients.
Distribution
Ipratropium bromide is minimally bound (0 to 9% in vitro) to plasma albumin and α1-acid glycoprotein. Its blood/plasma concentration ratio was estimated to be about 0.89. Studies in rats have shown that ipratropium bromide does not penetrate the blood-brain barrier.
Metabolism
Ipratropium bromide is partially metabolized to ester hydrolysis products, tropic acid, and tropane. These metabolites appear to be inactive based on in vitro receptor affinity studies using rat brain tissue homogenates.
Elimination
After intravenous administration of 2 mg ipratropium bromide to 10 healthy volunteers, the terminal half-life of ipratropium bromide was approximately 1.6 hours. The total body clearance and renal clearance were estimated to be 2,505 and 1,019 mL/min, respectively. The amount of the total dose excreted unchanged in the urine (Ae) within 24 hours was approximately one-half of the administered dose.
Pediatrics
Following administration of 84 mcg of ipratropium bromide per nostril three times a day in patients 5 to 18 years old (n=42) with a naturally acquired common cold, the mean amount of the total dose excreted unchanged in the urine of 7.8% was comparable to 84 mcg per nostril four times a day in an adult induced common cold population (n=22) of 7.3 to 8.1%. Plasma ipratropium concentrations were relatively low (ranging from undetectable up to 0.62 ng/mL). No correlation of the amount of the total dose excreted unchanged in the urine (Ae) with age or gen- der was observed in the pediatric population.
Special Populations
Gender does not appear to influence the absorption or excretion of nasally administered ipratropium bromide. The pharmacokinetics of ipratropium bromide have not been studied in patients with hepatic or renal insufficiency or in the elderly.
Drug-Drug Interactions
No specific pharmacokinetic studies were conducted to evaluate potential drug-drug interactions.
Pharmacodynamics
In two single dose trials (n=17), doses up to 336 mcg of ipratropium bromide did not significantly affect pupillary diameter, heart rate, or systolic/diastolic blood pressure. Similarly, ipratropium bromide 0.06% in adult patients (n=22) with induced-colds (84 mcg/nostril four times a day) and in pediatric patients (n=45) with naturally acquired common cold (84 mcg/nostril three times a day) had no significant effects on pupillary diameter, heart rate, or systolic/diastolic blood pressure.
Controlled clinical trials demonstrated that intranasal fluorocarbon-propelled ipratropium bromide does not alter physiologic nasal functions (e.g., sense of smell, ciliary beat frequency, mucociliary clearance, or the air conditioning capacity of the nose).
Clinical Trials
Clinical trials for ipratropium bromide 0.06% were conducted in patients with rhinorrhea associated with naturally occurring common colds. In two controlled four day comparisons of ipratropium bromide 0.06% (84 mcg per nostril, administered three or four times daily; n=352) with its vehicle (n=351), there was a statistically significant reduction of rhinorrhea, as measured by both nasal discharge weight and the patients’ subjective assessment of severity of rhinorrhea using a visual analog scale. These significant differences were evident within one hour following dosing. There was no effect of ipratropium bromide 0.06% on degree of nasal congestion or sneezing. The response to ipratropium bromide 0.06% did not appear to be affected by age or gender. No controlled clinical trials directly compared the efficacy of three times daily versus four times daily treatment.
One clinical trial was conducted with ipratropium bromide 0.06%, administered four times daily for three weeks, in 218 patients with rhinorrhea associated with Seasonal Allergic Rhinitis (SAR), compared to its vehicle in 211 patients. Patients in this trial were adults and adolescents 12 years of age and above. Ipratropium bromide 0.06% was significantly more effective in reducing the severity and duration of rhinorrhea over the three weeks of the study, as measured by daily patient symptom scores. There was no difference between treatment groups in the effect on nasal congestion, sneezing or itching eyes.
-
INDICATIONS AND USAGE
Ipratropium bromide nasal solution 0.06% is indicated for the symptomatic relief of rhinorrhea associated with the common cold or seasonal allergic rhinitis for adults and children age 5 years and older. Ipratropium bromide 0.06% does not relieve nasal congestion or sneezing associated with the common cold or seasonal allergic rhinitis.
The safety and effectiveness of the use of ipratropium bromide 0.06% beyond four days in patients with the common cold or beyond three weeks in patients with seasonal allergic rhinitis has not been established.
- CONTRAINDICATIONS
-
WARNINGS
Immediate hypersensitivity reactions may occur after administration of ipratropium bromide, as demonstrated by urticaria, angioedema, rash, bronchospasm, anaphylaxis, and oropharyngeal edema. If such a reaction occurs, therapy with ipratropium bromide should be stopped at once and alternative treatment should be considered.
-
PRECAUTIONS
Information for Patients
Patients should be advised that temporary blurring of vision, precipitation or worsening of narrow-angle glaucoma, mydriasis, increased intraocular pressure, acute eye pain or discomfort, visual halos or colored images in association with red eyes from conjunctival and corneal congestion may result if ipratropium bromide comes into direct contact with the eyes. Patients should be instructed to avoid spraying ipratropium bromide in or around their eyes. Patients who experience eye pain, blurred vision, excessive nasal dryness or episodes of nasal bleeding should be instructed to contact their doctor. To ensure proper dosing, patients should be advised not to alter the size of the nasal spray opening. Patients should be reminded to carefully read and follow the accompanying “Patient’s Instructions for Use”.
Since dizziness, accommodation disorder, mydriasis, and blurred vision may occur with use of ipratropium bromide patients should be cautioned about engaging in activities requiring balance and visual acuity such as driving a car or operating appliances, machinery, etc.
Drug Interactions
No controlled clinical trials were conducted to investigate potential drug-drug interactions. There is potential for an additive interaction with other concomitantly administered medications with anticholinergic properties, including ipratropium bromide for oral inhalation.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Two-year oral carcinogenicity studies in rats and mice have revealed no carcinogenic activity at doses up to 6 mg/kg. This dose corresponds in rats and mice to approximately 70 and 35 times the maximum recommended daily intranasal dose in adults, respectively, and approximately 35 and 15 times the maximum recommended daily intranasal dose in children, respectively, on a mg/m2 basis. Results of various mutagenicity studies (Ames test, mouse dominant lethal test, mouse micronucleus test, and chromosome aberration of bone marrow in Chinese hamsters) were negative.
Fertility of male or female rats at oral doses up to 50 mg/kg (approximately 600 times the maximum recommended daily intranasal dose in adults on a mg/m2 basis) was unaffected by ipratropium bromide administration. At an oral dose of 500 mg/kg (approximately 6,000 times the maximum recommended daily intranasal dose in adults on a mg/m2 basis), ipratropium bromide produced a decrease in the conception rate.
Pregnancy
Teratogenic Effects
Pregnancy Category B
There are no adequate and well-controlled studies for ipratropium bromide in pregnant women. Because animal reproduction studies are not always predictive of human response, ipratropium bromide should be used during pregnancy only if clearly needed.
Oral reproduction studies were performed at ipratropium doses of 10 mg/kg in mice, 1,000 mg/kg in rats and 125 mg/kg in rabbits. These doses correspond, in each species respectively, to approximately 60, 12,000, and 3,000 times the maximum recommended daily intranasal dose (MRDID) in adults on a mg/m2 basis. Inhalation reproduction studies were conducted in rats and rabbits at doses of 1.5 and 1.8 mg/kg, respectively, (approximately 20 and 45 times, respectively, the MRDID in adults on a mg/m2 basis). These studies demonstrated no evidence of teratogenic effects as a result of ipratropium bromide. At oral doses 90 mg/ kg and above in rats (approximately 1,100 times the MRDID in adults on a mg/m2 basis) embryotoxicity was observed as increased resorption. This effect is not considered relevant to human use due to the large doses at which it was observed and the difference in route of administration.
Nursing Mothers
It is known that some ipratropium bromide is systemically absorbed following nasal administration; however the portion which may be excreted in human milk is unknown. Because lipid-insoluble quaternary cations pass into breast milk, caution should be exercised when ipratropium bromide is administered to a nursing mother.
Pediatric Use
The safety of ipratropium bromide nasal solution 0.06% at a dose of two sprays (84 mcg) per nostril three times a day (total dose 504 mcg/day) for two to four days has been demonstrated in two clinical trials involving 362 pediatric patients 5 to 11 years of age with naturally acquired common colds. In this pediatric population ipratropium bromide nasal solution 0.06% had an adverse event profile similar to that observed in adolescent and adult patients. When ipratropium bromide was concomitantly administered with an oral decongestant (pseudoephedrine HCl) in 122 children ages 5 to 12 years, and concomitantly administered with an oral decongestant/antihistamine combination (pseudoephedrine HCl/ chlorpheniramine maleate) in 123 children ages 5 to 12 years, adverse event profiles were similar to ipratropium bromide alone. The safety of ipratropium bromide nasal solution 0.06% at a dose of two sprays (84 mcg) per nostril four times a day (total dose 672 mcg/day) for three weeks in pediatric seasonal allergic rhinitis patients down to 5 years is based upon the safety demonstrated in the pediatric common cold trials and the trial in adult and adolescent patients 12 to 75 years of age with seasonal allergic rhinitis. The effectiveness of ipratropium bromide nasal solution 0.06% for the treatment of rhinorrhea associated with the common cold and seasonal allergic rhinitis in this pediatric age group is based on extrapolation of the demonstrated efficacy of ipratropium bromide nasal solution 0.06% in adolescents and adults with the conditions and the likelihood that the disease course, pathophysiology, and the drug’s effects are substantially similar to that of adults. The recommended dose for common cold for the pediatric population is based on cross-study comparisons of the efficacy of ipratropium bromide nasal solution 0.06% in adult and pediatric patients and on its safety profile in both adults and pediatric common cold patients. The recommended dose for seasonal allergic rhinitis for the pediatric population down to 5 years is based upon the efficacy and safety of ipratropium bromide nasal solution 0.06% in adults and adolescents 12 years of age and above with seasonal allergic rhinitis and the safety profile of this dose in both adult and pediatric common cold patients. The safety and effectiveness of ipratropium bromide nasal solution 0.06% in pediatric patients under 5 years of age have not been established.
-
ADVERSE REACTIONS
Adverse reaction information on ipratropium bromide 0.06% in patients with the common cold was derived from two multicenter, vehicle-controlled clinical trials involving 1,276 patients (195 patients on ipratropium bromide 0.03%, 352 patients on ipratropium bromide 0.06%, 189 patients on ipratropium bromide 0.12%, 351 patients on vehicle and 189 patients receiving no treatment).
Table 1 shows adverse events reported for patients who received ipratropium bromide 0.06% at the recommended dose of 84 mcg per nostril, or vehicle, administered three or four times daily, where the incidence is 1% or greater in the ipratropium bromide group and higher in the ipratropium bromide group than in the vehicle group.
Table 1: % of Patients with Common Cold Reporting Events1
Ipratropium Bromide 0.06% Vehicle Control No. of Patients
352
351
Epistaxis2
8.2%
2.3%
Nasal Dryness
4.8%
2.8%
Dry Mouth/Throat
1.4%
0.3%
Nasal Congestion
1.1%
0.0%
1 This table includes adverse events for which the incidence was 1% or greater in the ipratropium bromide group and higher in the ipratropium bromide group than in the vehicle group.
2 Epistaxis reported by 5.4% of ipratropium bromide patients and 1.4% of vehicle patients, blood tinged nasal mucus by 2.8% of ipratropium bromide patients and 0.9% of vehicle patients.
Ipratropium bromide 0.06% was well tolerated by most patients. The most frequently reported adverse events were transient episodes of nasal dryness or epistaxis. The majority of these adverse events (96%) were mild or moderate in nature, none was considered serious, and none resulted in hospitalization. No patient required treatment for nasal dryness, and only three patients (<1%) required treatment for epistaxis, which consisted of local application of pressure or a moisturizing agent (e.g., petroleum jelly). No patient receiving ipratropium bromide 0.06% was discontinued from the trial due to either nasal dryness or bleeding.
Adverse events reported by less than 1% of the patients receiving ipratropium bromide 0.06% during the controlled clinical trials that are potentially related to ipratropium bromide’s local effects or systemic anticholinergic effects include: taste perversion, nasal burning, conjunctivitis, coughing, dizziness, hoarseness, palpitation, pharyngitis, tachycardia, thirst, tinnitus, and blurred vision. No controlled trial was conducted to address the relative incidence of adverse events for three times daily versus four times daily therapy.
Nasal adverse events seen in the clinical trial with seasonal allergic rhinitis (SAR) patients (see Table 2) were similar to those seen in the common cold trials. Additional events were reported at a higher rate in the SAR trial due in part to the longer duration of the trial and the inclusion of Upper Respiratory Tract Infection (URI) as an adverse event. In common cold trials, URI was the disease under study and not an adverse event.
Table 2: % of Patients with SAR Reporting Events1 Ipratropium Bromide 0.06% Vehicle Control No. of Patients
218
211
Epistaxis2
6.0%
3.3%
Pharyngitis
5.0%
3.8%
URI
5.0%
3.3%
Nasal Dryness
4.6%
0.9%
Headache
4.1%
0.5%
Dry Mouth/Throat
4.1%
0.0%
Taste Perversion
3.7%
1.4%
Sinusitis
2.8%
2.8%
Pain
1.8%
0.9%
Diarrhea
1.8%
0.5%
1 This table includes adverse events for which the incidence was 1% or greater in the ipratropium bromide group and higher in the ipratropium bromide group than in the vehicle group.
2 Epistaxis reported by 3.7% of ipratropium bromide patients and 2.4% of vehicle patients, blood tinged nasal mucus by 2.3% of ipratropium bromide patients and 1.9% of vehicle patients.
There were no reports of allergic-type reactions in the controlled clinical common cold and SAR trials.
Post-Marketing Experience
Allergic type reactions such as skin rash, angioedema, including that of the throat, tongue, lips and face, generalized urticaria (including giant urticaria), laryngospasm, and anaphylactic reactions have been reported with ipratropium bromide 0.06% and for other ipratropium bromide-containing products, with positive rechallenge in some cases.
Additional side effects identified from the published literature and/or post-marketing surveillance on the use of ipratropium bromide-containing products (singly or in combination with albuterol), include: urinary retention, prostatic disorders, mydriasis, cases of precipitation or worsening of narrow-angle glaucoma, acute eye pain, ocular irritation, wheezing, dryness of the oropharynx, tachycardia, edema, gastrointestinal distress (diarrhea, nausea, vomiting), bowel obstruction, constipation, nasal discomfort, throat irritation, hypersensitivity, accommodation disorder, intraocular pressure increased, glaucoma, halo vision, conjunctival hyperaemia, corneal edema, heart rate increased, bronchospasm, pharyngeal edema, gastrointestinal motility disorder, mouth edema, stomatitis, and pruritus.
After oral inhalation of ipratropium bromide in patients suffering from COPD/Asthma supraventricular tachycardia and atrial fibrillation have been reported.
-
OVERDOSAGE
Acute overdosage by intranasal administration is unlikely since ipratropium bromide is not well absorbed systemically after intranasal or oral administration. Following administration of a 20 mg oral dose (equivalent to ingesting more than two bottles of ipratropium bromide 0.06%) to 10 male volunteers, no change in heart rate or blood pressure was noted. Following a 2 mg intravenous infusion over 15 minutes to the same 10 male volunteers, plasma ipratropium concentrations of 22 to 45 ng/mL were observed (>100 times the concentrations observed following intranasal administration). Following intravenous infusion these 10 volunteers had a mean increase of heart rate of 50 bpm and less than 20 mmHg change in systolic or diastolic blood pressure at the time of peak ipratropium levels.
-
DOSAGE AND ADMINISTRATION
For Symptomatic Relief of Rhinorrhea Associated with the Common Cold
The recommended dose of ipratropium bromide nasal solution 0.06% is two sprays (84 mcg) per nostril three or four times daily (total dose 504 to 672 mcg/day) in adults and children age 12 years and older. Optimum dosage varies with response of the individual patient. The recommended dose of ipratropium bromide nasal solution 0.06% for children age 5 to 11 years is two sprays (84 mcg) per nostril three times daily (total dose of 504 mcg/day).
The safety and effectiveness of the use of ipratropium bromide nasal solution 0.06% beyond four days in patients with the common cold have not been established.
For Symptomatic Relief of Rhinorrhea Associated with Seasonal Allergic Rhinitis
The recommended dose of ipratropium bromide nasal solution, 0.06% is two sprays (84 mcg) per nostril four times daily (total dose 672 mcg/day) in adults and children age 5 years and older.
The safety and effectiveness of the use of ipratropium bromide nasal solution 0.06% beyond three weeks in patients with seasonal allergic rhinitis have not been established.
Initial pump priming requires seven sprays of the pump. If used regularly as recommended, no further priming is required. If not used for more than 24 hours, the pump will require two sprays, or if not used for more than seven days, the pump will require seven sprays to reprime. Avoid spraying into eyes.
-
HOW SUPPLIED
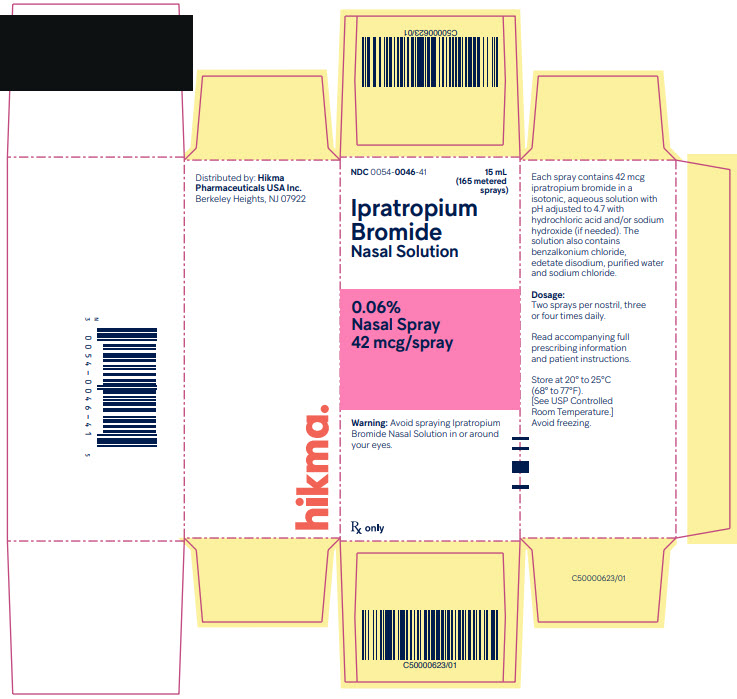
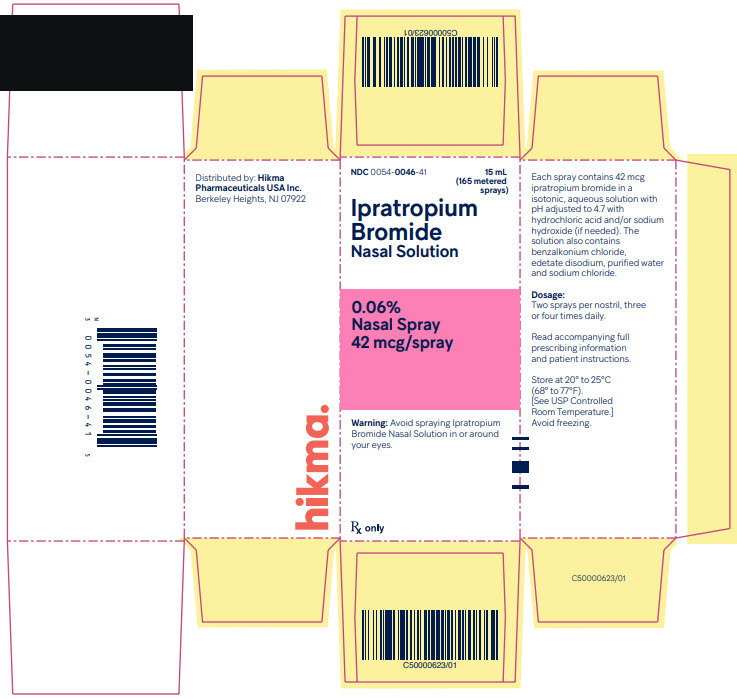
Ipratropium Bromide Nasal Solution 0.06% is supplied as a clear, colorless solution in a white high density polyethylene (HDPE) bottle fitted with a white and clear metered nasal spray pump, a green safety clip to prevent accidental discharge of the spray, and a clear plastic dust cap. It contains 16.6 g of product formulation, 165 sprays, each delivering 42 mcg (70 mcL) of ipratropium per spray, or 10 days of therapy at the maximum recommended dose (two sprays per nostril four times a day).
Ipratopium Bromide Nasal Solution, 0.06%
Nasal Spray, 42 mcg/spray
NDC 0054-0046-41: Bottle of 15 mL (165 metered sprays)
Storage
Store at 20° to 25°C (68° to 77°F).[See USP Controlled Room Temperature.] Avoid freezing. Keep out of reach of children. Do not spray in the eyes.
Address medical inquiries to Hikma Pharmaceuticals USA Inc. at 1-800-962-8364.
Patients should be reminded to read and follow the accompanying “Patient’s Instructions for Use”, which should be dispensed with the product.
Distributed by:
Hikma Pharmaceuticals USA Inc.Berkeley Heights, NJ 07922
C50000618/01
Revised July 2022
-
PATIENT’S INSTRUCTIONS FOR USE
Ipratropium Bromide Nasal Solution 0.06%
Nasal Spray, 42 mcg/sprayRx Only
Read complete instructions carefully before using.
In order to ensure proper dosing, do not attempt to change the size of the spray opening.
Ipratropium bromide nasal solution 0.06% is indicated for the symptomatic relief of rhinorrhea (runny nose) associated with the common cold or seasonal allergic rhinitis for adults and children age 5 years and older. Ipratropium bromide nasal solution 0.06% does not relieve nasal congestion or sneezing, associated with the common cold or seasonal allergic rhinitis. Do not use ipratropium bromide nasal solution 0.06 % for longer than four days for a common cold or three weeks for seasonal allergic rhinitis unless instructed by your physician.
Read complete instructions carefully and use only as directed.
To Use:
- 1.
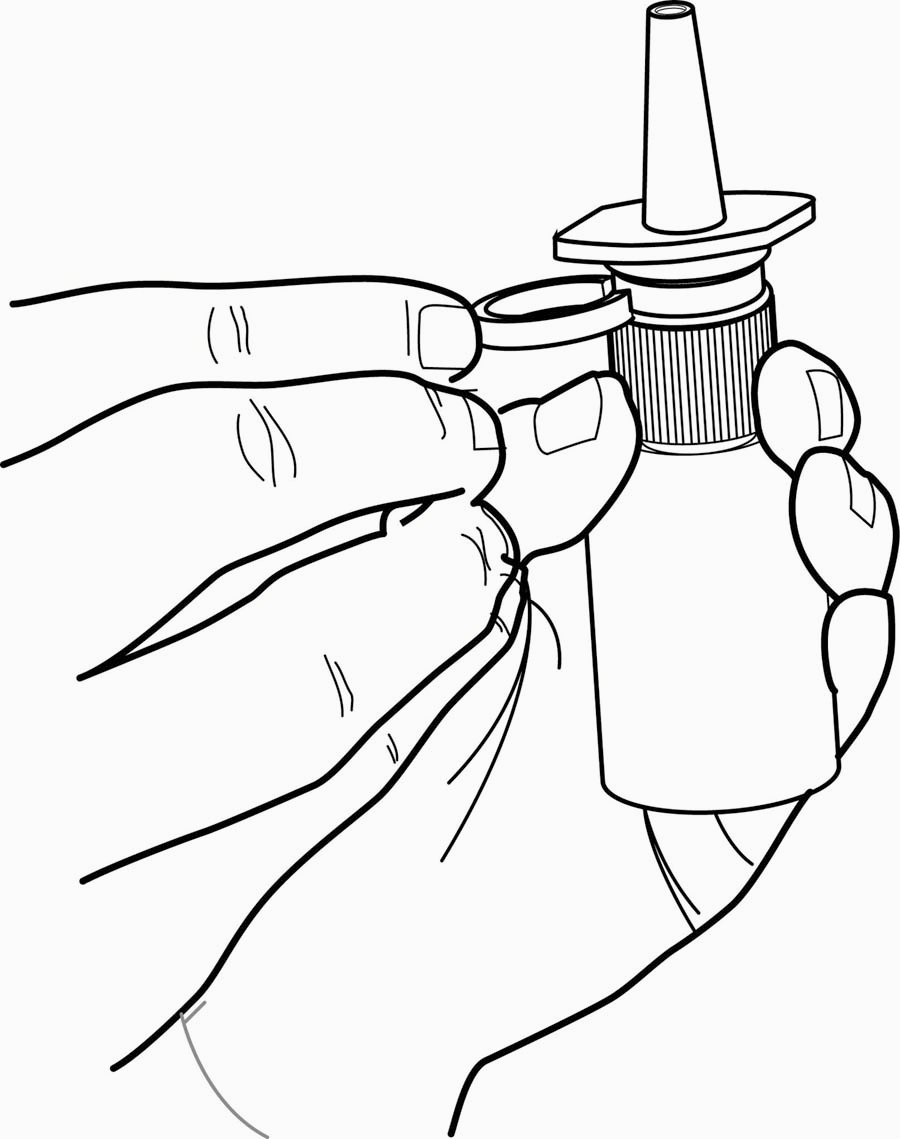
- Remove the clear plastic dust cap and the green safety clip from the nasal spray pump (Figure 1). The safety clip prevents the accidental discharge of the spray in your pocket or purse.
Figure 1
- 2.
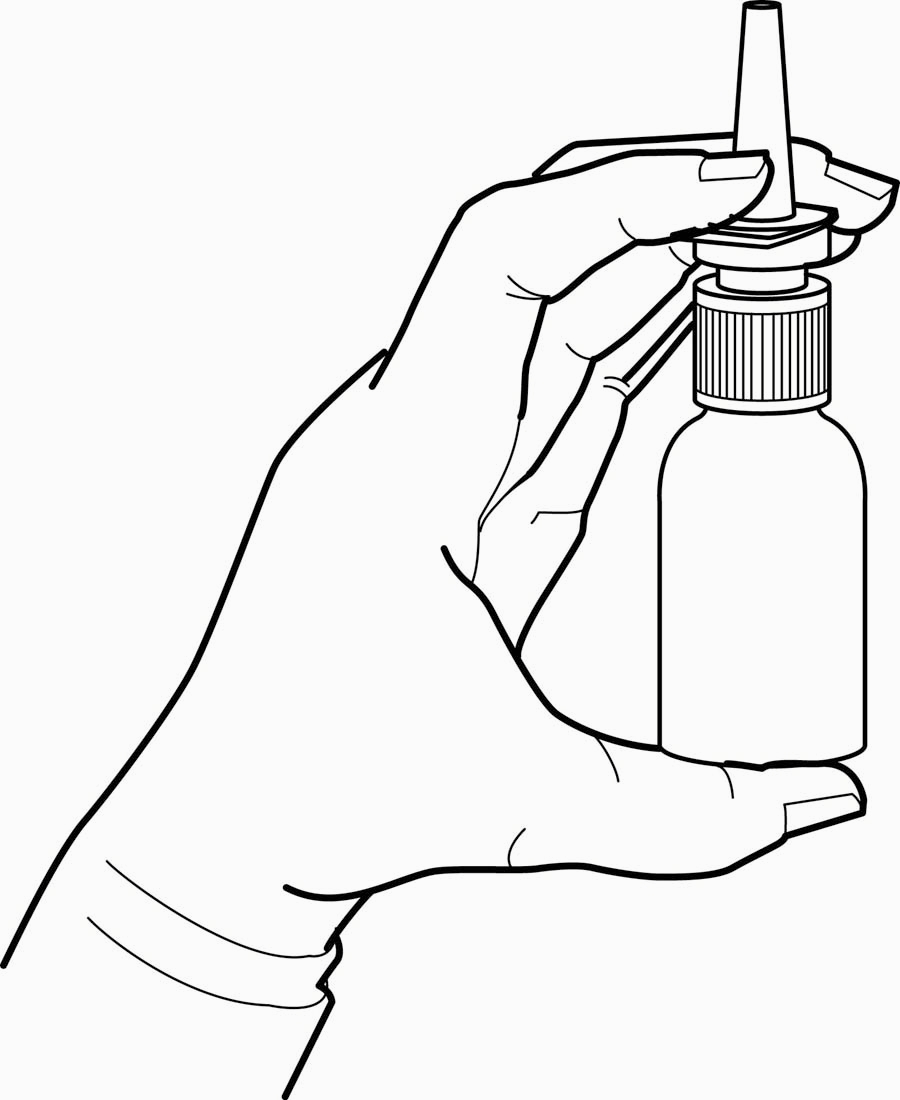
- The nasal spray pump must be primed before ipratropium bromide nasal solution, 0.06% is used for the first time. To prime the pump, hold the bottle with your thumb at the base and your index and middle fingers on the white shoulder area. Make sure the bottle points upright and away from your eyes. Press your thumb firmly and quickly against the bottle seven times (Figure 2). The pump is now primed and can be used. Your pump should not have to be reprimed unless you have not used the medication for more than 24 hours; repriming the pump will only require two sprays. If you have not used your nasal spray for more than seven days, repriming the pump will require seven sprays.
Figure 2
- 3.
- Before using ipratropium bromide nasal solution 0.06%, blow your nose gently to clear your nostrils if necessary.
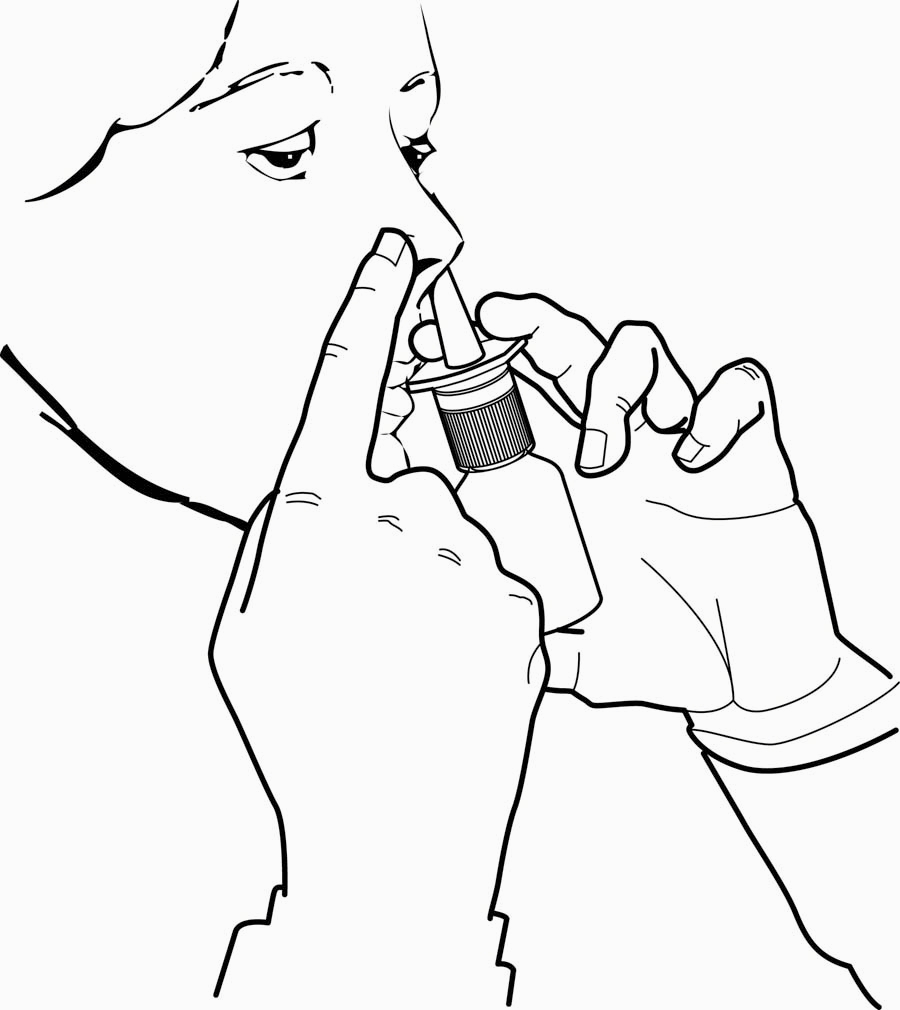
- 4.
- Close one nostril by gently placing your finger against the side of your nose, tilt your head slightly forward and, keeping the bottle upright, insert the nasal tip into the other nostril (Figure 3). Point the tip toward the back and outer side of the nose.
Figure 3
- 5.
- Press firmly and quickly upwards with the thumb at the base while holding the white shoulder portion of the pump between your index and middle fingers. Following each spray, sniff deeply and breathe out through your mouth.
- 6.
- After spraying the nostril and removing the unit, tilt your head backwards for a few seconds to let the spray spread over the back of the nose.
- 7.
- Repeat steps 4 through 6 in the same nostril.
- 8.
- Repeat steps 4 through 7 in the other nostril (i.e., two sprays per nostril).
- 9.
- Replace the clear plastic dust cap and safety clip.
- 10.
- At some time before the medication is completely used up, you should consult your physician or pharmacist to determine whether a refill is needed. You should not take extra doses or stop using ipratropium bromide nasal solution 0.06% without consulting your physician.
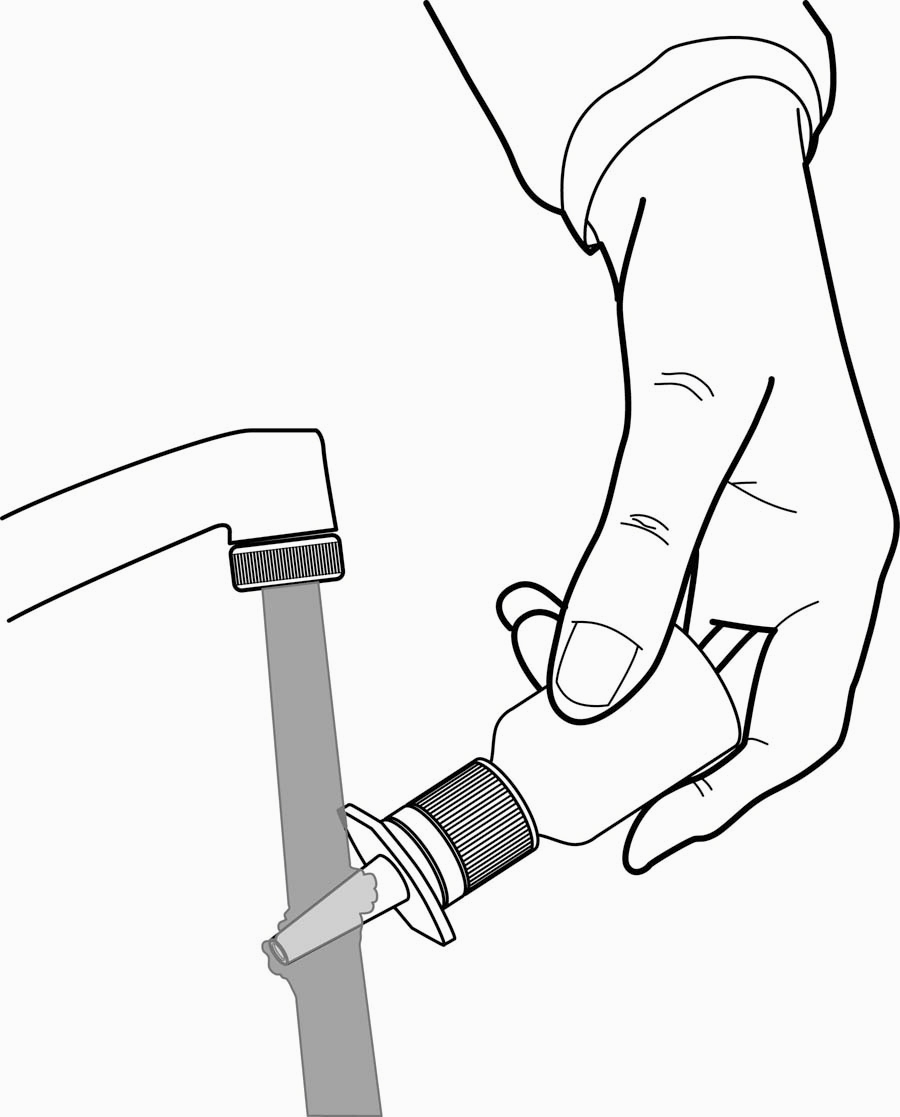
To Clean:
If the nasal tip becomes clogged, remove the clear plastic dust cap and safety clip. Hold the nasal tip under running, warm tap water (Figure 4) for about a minute. Dry the nasal tip, reprime the nasal spray pump (step 2 above), and replace the plastic dust cap and safety clip.
Figure 4
Caution:
Ipratropium bromide nasal solution 0.06% is intended to relieve your rhinorrhea (runny nose) with regular use. It is therefore important that you use ipratropium bromide nasal solution 0.06% as prescribed by your physician. For most patients, some improvement in runny nose is usually apparent following the first dose of treatment with ipratropium bromide nasal solution 0.06%. Do not use ipratropium bromide nasal solution 0.06% for longer than four days for your cold or three weeks for seasonal allergic rhinitis unless instructed by your physician.
Do not spray ipratropium bromide nasal solution 0.06% in your eyes. Should this occur, immediately flush your eye with cool tap water for several minutes. If you accidentally spray ipratropium bromide nasal solution 0.06% in your eyes, you may experience a temporary blurring of vision, visual halos or colored images in association with red eyes from conjunctival and corneal congestion, development or worsening of narrow-angle glaucoma, pupil dilation, or acute eye pain/discomfort, and increased sensitivity to light, which may last a few hours. Should eye pain or blurred vision occur, contact your doctor.
Should you experience excessive nasal dryness or episodes of nasal bleeding, contact your doctor.
If you have glaucoma or difficulty urinating due to an enlargement of the prostate, be sure to tell your physician prior to using ipratropium bromide nasal solution 0.06%.
If you are pregnant or breast feeding your baby, be sure to tell your physician prior to using ipratropium bromide nasal solution 0.06%.
Storage
Store at 20° to 25°C (68° to 77°F).[See USP Controlled Room Temperature.] Avoid freezing. Keep out of reach of children.
Address medical inquiries to Hikma Pharmaceuticals USA Inc. at 1-800-962-8364.
Distributed by:
Hikma Pharmaceuticals USA Inc.
Berkeley Heights, NJ 07922
C50000618/01
Revised July 2022
- PACKAGE/LABEL PRINCIPAL DISPLAY PANEL
- PACKAGE/LABEL PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
IPRATROPIUM BROMIDE
ipratropium bromide sprayProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0054-0046 Route of Administration NASAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength IPRATROPIUM BROMIDE (UNII: J697UZ2A9J) (IPRATROPIUM - UNII:GR88G0I6UL) IPRATROPIUM BROMIDE ANHYDROUS 42 ug Inactive Ingredients Ingredient Name Strength HYDROXYETHYL CELLULOSE (140 MPA.S AT 5%) (UNII: 8136Y38GY5) BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) EDETATE CALCIUM DISODIUM (UNII: 25IH6R4SGF) HYDROCHLORIC ACID (UNII: QTT17582CB) WATER (UNII: 059QF0KO0R) SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0054-0046-41 1 in 1 CARTON 11/05/2003 1 165 in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA076598 11/05/2003 Labeler - Hikma Pharmaceuticals USA Inc. (080189610) Establishment Name Address ID/FEI Business Operations West-Ward Columbus Inc. 058839929 MANUFACTURE(0054-0046)