TYKERB- lapatinib tablet
GlaxoSmithKline LLC
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use TYKERB safely and effectively. See full prescribing information for TYKERB.
TYKERB (lapatinib) tablets, for oral use Initial U.S. Approval: 2007 WARNING: HEPATOTOXICITYSee full prescribing information for complete boxed warning.Hepatotoxicity has been observed in clinical trials and postmarketing experience. The hepatotoxicity may be severe and deaths have been reported. Causality of the deaths is uncertain [see Warnings and Precautions (5.2)]. RECENT MAJOR CHANGES
INDICATIONS AND USAGETYKERB, a kinase inhibitor, is indicated in combination with: (1)
TYKERB in combination with an aromatase inhibitor has not been compared to a trastuzumab-containing chemotherapy regimen for the treatment of metastatic breast cancer. DOSAGE AND ADMINISTRATIONThe recommended dosage of TYKERB for advanced or metastatic breast cancer is 1,250 mg (5 tablets) given orally once daily on Days 1-21 continuously in combination with capecitabine 2,000 mg/m2/day (administered orally in 2 doses approximately 12 hours apart) on Days 1-14 in a repeating 21-day cycle. (2.1) The recommended dose of TYKERB for hormone receptor-positive, HER2-positive metastatic breast cancer is 1,500 mg (6 tablets) given orally once daily continuously in combination with letrozole. When TYKERB is coadministered with letrozole, the recommended dose of letrozole is 2.5 mg once daily. (2.1)
DOSAGE FORMS AND STRENGTHS250 mg tablets (3) CONTRAINDICATIONSKnown severe hypersensitivity (e.g., anaphylaxis) to this product or any of its components. (4) WARNINGS AND PRECAUTIONS
ADVERSE REACTIONSThe most common (>20%) adverse reactions during treatment with TYKERB plus capecitabine were diarrhea, palmar-plantar erythrodysesthesia, nausea, rash, vomiting, and fatigue. The most common (≥20%) adverse reactions during treatment with TYKERB plus letrozole were diarrhea, rash, nausea, and fatigue. (6.1) To report SUSPECTED ADVERSE REACTIONS, contact GlaxoSmithKline at 1-888-825-5249 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. DRUG INTERACTIONS
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling. Revised: 3/2015 |
FULL PRESCRIBING INFORMATION
WARNING: HEPATOTOXICITY
Hepatotoxicity has been observed in clinical trials and postmarketing experience. The hepatotoxicity may be severe and deaths have been reported. Causality of the deaths is uncertain [see Warnings and Precautions (5.2)].
1 INDICATIONS AND USAGE
TYKERB® is indicated in combination with:
- •
- capecitabine for the treatment of patients with advanced or metastatic breast cancer whose tumors overexpress HER2 and who have received prior therapy including an anthracycline, a taxane, and trastuzumab.
Limitation of Use: Patients should have disease progression on trastuzumab prior to initiation of treatment with TYKERB in combination with capecitabine. - •
- letrozole for the treatment of postmenopausal women with hormone receptor-positive metastatic breast cancer that overexpresses the HER2 receptor for whom hormonal therapy is indicated.
TYKERB in combination with an aromatase inhibitor has not been compared to a trastuzumab-containing chemotherapy regimen for the treatment of metastatic breast cancer.
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosing
HER2-Positive Metastatic Breast Cancer: The recommended dose of TYKERB is 1,250 mg given orally once daily on Days 1-21 continuously in combination with capecitabine 2,000 mg/m2/day (administered orally in 2 doses approximately 12 hours apart) on Days 1-14 in a repeating 21-day cycle. TYKERB should be taken at least one hour before or one hour after a meal. The dose of TYKERB should be once daily (5 tablets administered all at once); dividing the daily dose is not recommended [see Clinical Pharmacology (12.3)]. Capecitabine should be taken with food or within 30 minutes after food. If a day’s dose is missed, the patient should not double the dose the next day. Treatment should be continued until disease progression or unacceptable toxicity occurs.
Hormone Receptor-Positive, HER2-Positive Metastatic Breast Cancer: The recommended dose of TYKERB is 1,500 mg given orally once daily continuously in combination with letrozole. When coadministered with TYKERB, the recommended dose of letrozole is 2.5 mg once daily. TYKERB should be taken at least one hour before or one hour after a meal. The dose of TYKERB should be once daily (6 tablets administered all at once); dividing the daily dose is not recommended [see Clinical Pharmacology (12.3)].
2.2 Dose Modification Guidelines
Cardiac Events: TYKERB should be discontinued in patients with a decreased left ventricular ejection fraction (LVEF) that is Grade 2 or greater by National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE v3) and in patients with an LVEF that drops below the institution’s lower limit of normal [see Warnings and Precautions (5.1) and Adverse Reactions (6.1)]. TYKERB in combination with capecitabine may be restarted at a reduced dose (1,000 mg/day) and in combination with letrozole may be restarted at a reduced dose of 1,250 mg/day after a minimum of 2 weeks if the LVEF recovers to normal and the patient is asymptomatic.
Hepatic Impairment: Patients with severe hepatic impairment (Child-Pugh Class C) should have their dose of TYKERB reduced. A dose reduction from 1,250 mg/day to 750 mg/day (HER2-positive metastatic breast cancer indication) or from 1,500 mg/day to 1,000 mg/day (hormone receptor-positive, HER2-positive breast cancer indication) in patients with severe hepatic impairment is predicted to adjust the area under the curve (AUC) to the normal range and should be considered. However, there are no clinical data with this dose adjustment in patients with severe hepatic impairment.
Diarrhea: TYKERB should be interrupted in patients with diarrhea which is NCI CTCAE Grade 3 or Grade 1 or 2 with complicating features (moderate to severe abdominal cramping, nausea or vomiting ≥NCI CTCAE Grade 2, decreased performance status, fever, sepsis, neutropenia, frank bleeding, or dehydration). TYKERB may be reintroduced at a lower dose (reduced from 1,250 mg/day to 1,000 mg/day or from 1,500 mg/day to 1,250 mg/day) when diarrhea resolves to Grade 1 or less. TYKERB should be permanently discontinued in patients with diarrhea which is NCI CTCAE Grade 4 [see Warnings and Precautions (5.4) and Adverse Reactions (6.1)].
Concomitant Strong CYP3A4 Inhibitors: The concomitant use of strong CYP3A4 inhibitors should be avoided (e.g., ketoconazole, itraconazole, clarithromycin, atazanavir, indinavir, nefazodone, nelfinavir, ritonavir, saquinavir, telithromycin, voriconazole). Grapefruit may also increase plasma concentrations of lapatinib and should be avoided. If patients must be coadministered a strong CYP3A4 inhibitor, based on pharmacokinetic studies, a dose reduction to 500 mg/day of lapatinib is predicted to adjust the lapatinib AUC to the range observed without inhibitors and should be considered. However, there are no clinical data with this dose adjustment in patients receiving strong CYP3A4 inhibitors. If the strong inhibitor is discontinued, a washout period of approximately 1 week should be allowed before the lapatinib dose is adjusted upward to the indicated dose [see Drug Interactions (7.2)].
Concomitant Strong CYP3A4 Inducers: The concomitant use of strong CYP3A4 inducers should be avoided (e.g., dexamethasone, phenytoin, carbamazepine, rifampin, rifabutin, rifapentin, phenobarbital, St. John’s wort). If patients must be coadministered a strong CYP3A4 inducer, based on pharmacokinetic studies, the dose of lapatinib should be titrated gradually from 1,250 mg/day up to 4,500 mg/day (HER2-positive metastatic breast cancer indication) or from 1,500 mg/day up to 5,500 mg/day (hormone receptor-positive, HER2-positive breast cancer indication) based on tolerability. This dose of lapatinib is predicted to adjust the lapatinib AUC to the range observed without inducers and should be considered. However, there are no clinical data with this dose adjustment in patients receiving strong CYP3A4 inducers. If the strong inducer is discontinued the lapatinib dose should be reduced to the indicated dose [see Drug Interactions (7.2)].
Other Toxicities: Discontinuation or interruption of dosing with TYKERB may be considered when patients develop ≥Grade 2 NCI CTCAE toxicity and can be restarted at the standard dose of 1,250 or 1,500 mg/day when the toxicity improves to Grade 1 or less. If the toxicity recurs, then TYKERB in combination with capecitabine should be restarted at a lower dose (1,000 mg/day) and in combination with letrozole should be restarted at a lower dose of 1,250 mg/day.
See manufacturer’s prescribing information for the coadministered product dosage adjustment guidelines in the event of toxicity and other relevant safety information or contraindications.
3 DOSAGE FORMS AND STRENGTHS
250 mg tablets — oval, biconvex, orange, film-coated with GS XJG debossed on one side.
4 CONTRAINDICATIONS
TYKERB is contraindicated in patients with known severe hypersensitivity (e.g., anaphylaxis) to this product or any of its components.
5 WARNINGS AND PRECAUTIONS
5.1 Decreased Left Ventricular Ejection Fraction
TYKERB has been reported to decrease LVEF [see Adverse Reactions (6.1)]. In clinical trials, the majority (>57%) of LVEF decreases occurred within the first 12 weeks of treatment; however, data on long-term exposure are limited. Caution should be taken if TYKERB is to be administered to patients with conditions that could impair left ventricular function. LVEF should be evaluated in all patients prior to initiation of treatment with TYKERB to ensure that the patient has a baseline LVEF that is within the institution’s normal limits. LVEF should continue to be evaluated during treatment with TYKERB to ensure that LVEF does not decline below the institution’s normal limits [see Dosage and Administration (2.2)].
5.2 Hepatotoxicity
Hepatotoxicity (ALT or AST >3 times the upper limit of normal and total bilirubin >2 times the upper limit of normal) has been observed in clinical trials (<1% of patients) and postmarketing experience. The hepatotoxicity may be severe and deaths have been reported. Causality of the deaths is uncertain. The hepatotoxicity may occur days to several months after initiation of treatment. Liver function tests (transaminases, bilirubin, and alkaline phosphatase) should be monitored before initiation of treatment, every 4 to 6 weeks during treatment, and as clinically indicated. If changes in liver function are severe, therapy with TYKERB should be discontinued and patients should not be retreated with TYKERB [see Adverse Reactions (6.1)].
5.3 Patients With Severe Hepatic Impairment
If TYKERB is to be administered to patients with severe pre-existing hepatic impairment, dose reduction should be considered [see Dosage and Administration (2.2) and Use in Specific Populations (8.7)]. In patients who develop severe hepatotoxicity while on therapy, TYKERB should be discontinued and patients should not be retreated with TYKERB [see Warnings and Precautions (5.2)].
5.4 Diarrhea
Diarrhea has been reported during treatment with TYKERB [see Adverse Reactions (6.1)]. The diarrhea may be severe, and deaths have been reported. Diarrhea generally occurs early during treatment with TYKERB, with almost half of those patients with diarrhea first experiencing it within 6 days. This usually lasts 4 to 5 days. Lapatinib-induced diarrhea is usually low-grade, with severe diarrhea of NCI CTCAE Grades 3 and 4 occurring in <10% and <1% of patients, respectively. Early identification and intervention is critical for the optimal management of diarrhea. Patients should be instructed to report any change in bowel patterns immediately. Prompt treatment of diarrhea with anti-diarrheal agents (such as loperamide) after the first unformed stool is recommended. Severe cases of diarrhea may require administration of oral or intravenous electrolytes and fluids, use of antibiotics such as fluoroquinolones (especially if diarrhea is persistent beyond 24 hours, there is fever, or Grade 3 or 4 neutropenia), and interruption or discontinuation of therapy with TYKERB [see Dosage and Administration (2.2)].
5.5 Interstitial Lung Disease/Pneumonitis
Lapatinib has been associated with interstitial lung disease and pneumonitis in monotherapy or in combination with other chemotherapies [see Adverse Reactions (6.1)]. Patients should be monitored for pulmonary symptoms indicative of interstitial lung disease or pneumonitis. TYKERB should be discontinued in patients who experience pulmonary symptoms indicative of interstitial lung disease/pneumonitis which are ≥Grade 3 (NCI CTCAE).
5.6 QT Prolongation
QT prolongation was observed in an uncontrolled, open-label, dose-escalation study of lapatinib in advanced cancer patients [see Clinical Pharmacology (12.4)]. Lapatinib should be administered with caution to patients who have or may develop prolongation of QTc. These conditions include patients with hypokalemia or hypomagnesemia, with congenital long QT syndrome, patients taking anti-arrhythmic medicines or other medicinal products that lead to QT prolongation, and cumulative high-dose anthracycline therapy. Hypokalemia or hypomagnesemia should be corrected prior to lapatinib administration.
5.7 Severe Cutaneous Reactions
Severe cutaneous reactions have been reported with TYKERB. If life-threatening reactions such as erythema multiforme, Stevens-Johnson syndrome, or toxic epidermal necrolysis (e.g., progressive skin rash often with blisters or mucosal lesions) are suspected, discontinue treatment with TYKERB.
5.8 Use in Pregnancy
TYKERB can cause fetal harm when administered to a pregnant woman. Based on findings in animals, TYKERB is expected to result in adverse reproductive effects. Lapatinib administered to rats during organogenesis and through lactation led to death of offspring within the first 4 days after birth [see Use in Specific Populations (8.1)].
There are no adequate and well-controlled studies with TYKERB in pregnant women. Women should be advised not to become pregnant when taking TYKERB. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus.
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
HER2-Positive Metastatic Breast Cancer: The safety of TYKERB has been evaluated in more than 12,000 patients in clinical trials. The efficacy and safety of TYKERB in combination with capecitabine in breast cancer was evaluated in 198 patients in a randomized, Phase 3 trial [see Clinical Studies (14.1)]. Adverse reactions which occurred in at least 10% of patients in either treatment arm and were higher in the combination arm are shown in Table 1.
The most common adverse reactions (>20%) during therapy with TYKERB plus capecitabine were gastrointestinal (diarrhea, nausea, and vomiting), dermatologic (palmar-plantar erythrodysesthesia and rash), and fatigue. Diarrhea was the most common adverse reaction resulting in discontinuation of study medication.
The most common Grade 3 and 4 adverse reactions (NCI CTCAE v3) were diarrhea and palmar-plantar erythrodysesthesia. Selected laboratory abnormalities are shown in Table 2.
|
TYKERB 1,250 mg/day + Capecitabine 2,000 mg/m2/day |
Capecitabine 2,500 mg/m2/day |
|||||
|
(N = 198) |
(N = 191) |
|||||
|
All Gradesa |
Grade 3 |
Grade 4 |
All Gradesa |
Grade 3 |
Grade 4 |
|
|
Reactions |
% |
% |
% |
% |
% |
% |
|
Gastrointestinal disorders | ||||||
|
Diarrhea |
65 |
13 |
1 |
40 |
10 |
0 |
|
Nausea |
44 |
2 |
0 |
43 |
2 |
0 |
|
Vomiting |
26 |
2 |
0 |
21 |
2 |
0 |
|
Stomatitis |
14 |
0 |
0 |
11 |
<1 |
0 |
|
Dyspepsia |
11 |
<1 |
0 |
3 |
0 |
0 |
|
Skin and subcutaneous tissue disorders | ||||||
|
Palmar-plantar erythrodysesthesia |
53 |
12 |
0 |
51 |
14 |
0 |
|
Rashb |
28 |
2 |
0 |
14 |
1 |
0 |
|
Dry skin |
10 |
0 |
0 |
6 |
0 |
0 |
|
General disorders and administrative site conditions | ||||||
|
Mucosal inflammation |
15 |
0 |
0 |
12 |
2 |
0 |
|
Musculoskeletal and connective tissue disorders | ||||||
|
Pain in extremity |
12 |
1 |
0 |
7 |
<1 |
0 |
|
Back pain |
11 |
1 |
0 |
6 |
<1 |
0 |
|
Respiratory, thoracic, and mediastinal disorders | ||||||
|
Dyspnea |
12 |
3 |
0 |
8 |
2 |
0 |
|
Psychiatric disorders | ||||||
|
Insomnia |
10 |
<1 |
0 |
6 |
0 |
0 |
a National Cancer Institute Common Terminology Criteria for Adverse Events, version 3.
b Grade 3 dermatitis acneiform was reported in <1% of patients in the group receiving TYKERB plus capecitabine.
|
TYKERB 1,250 mg/day + Capecitabine 2,000 mg/m2/day |
Capecitabine 2,500 mg/m2/day |
|||||
|
All Gradesa |
Grade 3 |
Grade 4 |
All Gradesa |
Grade 3 |
Grade 4 |
|
|
Parameters |
% |
% |
% |
% |
% |
% |
|
Hematologic | ||||||
|
Hemoglobin |
56 |
<1 |
0 |
53 |
1 |
0 |
|
Platelets |
18 |
<1 |
0 |
17 |
<1 |
<1 |
|
Neutrophils |
22 |
3 |
<1 |
31 |
2 |
1 |
|
Hepatic | ||||||
|
Total Bilirubin |
45 |
4 |
0 |
30 |
3 |
0 |
|
AST |
49 |
2 |
<1 |
43 |
2 |
0 |
|
ALT |
37 |
2 |
0 |
33 |
1 |
0 |
a National Cancer Institute Common Terminology Criteria for Adverse Events, version 3.
Hormone Receptor-Positive, Metastatic Breast Cancer: In a randomized clinical trial of patients (N = 1,286) with hormone receptor-positive, metastatic breast cancer, who had not received chemotherapy for their metastatic disease, patients received letrozole with or without TYKERB. In this trial, the safety profile of TYKERB was consistent with previously reported results from trials of TYKERB in the advanced or metastatic breast cancer population. Adverse reactions which occurred in at least 10% of patients in either treatment arm and were higher in the combination arm are shown in Table 3. Selected laboratory abnormalities are shown in Table 4.
|
TYKERB 1,500 mg/day + Letrozole 2.5 mg/day |
Letrozole 2.5 mg/day |
|||||
|
(N = 654) |
(N = 624) |
|||||
|
All Gradesa |
Grade 3 |
Grade 4 |
All Gradesa |
Grade 3 |
Grade 4 |
|
|
Reactions |
% |
% |
% |
% |
% |
% |
|
Gastrointestinal disorders | ||||||
|
Diarrhea |
64 |
9 |
<1 |
20 |
<1 |
0 |
|
Nausea |
31 |
<1 |
0 |
21 |
<1 |
0 |
|
Vomiting |
17 |
1 |
<1 |
11 |
<1 |
<1 |
|
Anorexia |
11 |
<1 |
0 |
9 |
<1 |
0 |
|
Skin and subcutaneous tissue disorders | ||||||
|
Rashb |
44 |
1 |
0 |
13 |
0 |
0 |
|
Dry skin |
13 |
<1 |
0 |
4 |
0 |
0 |
|
Alopecia |
13 |
<1 |
0 |
7 |
0 |
0 |
|
Pruritus |
12 |
<1 |
0 |
9 |
<1 |
0 |
|
Nail Disorder |
11 |
<1 |
0 |
<1 |
0 |
0 |
|
General disorders and administrative site conditions | ||||||
|
Fatigue |
20 |
2 |
0 |
17 |
<1 |
0 |
|
Asthenia |
12 |
<1 |
0 |
11 |
<1 |
0 |
|
Nervous system disorders | ||||||
|
Headache |
14 |
<1 |
0 |
13 |
<1 |
0 |
|
Respiratory, thoracic, and mediastinal disorders | ||||||
|
Epistaxis |
11 |
<1 |
0 |
2 |
<1 |
0 |
a National Cancer Institute Common Terminology Criteria for Adverse Events, version 3.
b In addition to the rash reported under "Skin and subcutaneous tissue disorders", 3 additional subjects in each treatment arm had rash under "Infections and infestations"; none were Grade 3 or 4.
|
TYKERB 1,500 mg/day + Letrozole 2.5 mg/day |
Letrozole 2.5 mg/day |
|||||
|
All Gradesa |
Grade 3 |
Grade 4 |
All Gradesa |
Grade 3 |
Grade 4 |
|
|
Hepatic Parameters |
% |
% |
% |
% |
% |
% |
|
AST |
53 |
6 |
0 |
36 |
2 |
<1 |
|
ALT |
46 |
5 |
<1 |
35 |
1 |
0 |
|
Total Bilirubin |
22 |
<1 |
<1 |
11 |
1 |
<1 |
a National Cancer Institute Common Terminology Criteria for Adverse Events, version 3.
Decreases in Left Ventricular Ejection Fraction: Due to potential cardiac toxicity with HER2 (ErbB2) inhibitors, LVEF was monitored in clinical trials at approximately 8-week intervals. LVEF decreases were defined as signs or symptoms of deterioration in left ventricular cardiac function that are ≥Grade 3 (NCI CTCAE), or a ≥20% decrease in left ventricular cardiac ejection fraction relative to baseline which is below the institution's lower limit of normal. Among 198 patients who received combination treatment with TYKERB/capecitabine, 3 experienced Grade 2 and one had Grade 3 LVEF adverse reactions (NCI CTCAE v3) [see Warnings and Precautions (5.1)]. Among 654 patients who received combination treatment with TYKERB/letrozole, 26 patients experienced Grade 1 or 2 and 6 patients had Grade 3 or 4 LVEF adverse reactions.
Hepatotoxicity: TYKERB has been associated with hepatotoxicity [see Boxed Warning and Warnings and Precautions (5.2)].
Interstitial Lung Disease/Pneumonitis: TYKERB has been associated with interstitial lung disease and pneumonitis in monotherapy or in combination with other chemotherapies [see Warnings and Precautions (5.5)].
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of TYKERB. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune System Disorders: Hypersensitivity reactions including anaphylaxis [see Contraindications (4)].
Skin and Subcutaneous Tissue Disorders: Nail disorders including paronychia.
7 DRUG INTERACTIONS
7.1 Effects of Lapatinib on Drug Metabolizing Enzymes and Drug Transport Systems
Lapatinib inhibits CYP3A4, CYP2C8, and P-glycoprotein (P-gp, ABCB1) in vitro at clinically relevant concentrations and is a weak inhibitor of CYP3A4 in vivo. Caution should be exercised and dose reduction of the concomitant substrate drug should be considered when dosing TYKERB concurrently with medications with narrow therapeutic windows that are substrates of CYP3A4, CYP2C8, or P-gp. Lapatinib did not significantly inhibit the following enzymes in human liver microsomes: CYP1A2, CYP2C9, CYP2C19, and CYP2D6 or UGT enzymes in vitro, however, the clinical significance is unknown.
Midazolam: Following coadministration of TYKERB and midazolam (CYP3A4 substrate), 24-hour systemic exposure (AUC) of orally administered midazolam increased 45%, while 24-hour AUC of intravenously administered midazolam increased 22%.
Paclitaxel: In cancer patients receiving TYKERB and paclitaxel (CYP2C8 and P-gp substrate), 24-hour systemic exposure (AUC) of paclitaxel was increased 23%. This increase in paclitaxel exposure may have been underestimated from the in vivo evaluation due to study design limitations.
Digoxin: Following coadministration of TYKERB and digoxin (P-gp substrate), systemic AUC of an oral digoxin dose increased approximately 2.8-fold. Serum digoxin concentrations should be monitored prior to initiation of TYKERB and throughout coadministration. If digoxin serum concentration is >1.2 ng/mL, the digoxin dose should be reduced by half.
7.2 Drugs That Inhibit or Induce Cytochrome P450 3A4 Enzymes
Lapatinib undergoes extensive metabolism by CYP3A4, and concomitant administration of strong inhibitors or inducers of CYP3A4 alter lapatinib concentrations significantly (see Ketoconazole and Carbamazepine sections, below). Dose adjustment of lapatinib should be considered for patients who must receive concomitant strong inhibitors or concomitant strong inducers of CYP3A4 enzymes [see Dosage and Administration (2.2)].
Ketoconazole: In healthy subjects receiving ketoconazole, a CYP3A4 inhibitor, at 200 mg twice daily for 7 days, systemic exposure (AUC) to lapatinib was increased to approximately 3.6-fold of control and half-life increased to 1.7-fold of control.
Carbamazepine: In healthy subjects receiving the CYP3A4 inducer, carbamazepine, at 100 mg twice daily for 3 days and 200 mg twice daily for 17 days, systemic exposure (AUC) to lapatinib was decreased approximately 72%.
7.3 Drugs That Inhibit Drug Transport Systems
Lapatinib is a substrate of the efflux transporter P-glycoprotein (P-gp, ABCB1). If TYKERB is administered with drugs that inhibit P-gp, increased concentrations of lapatinib are likely, and caution should be exercised.
7.4 Acid-Reducing Agents
The aqueous solubility of lapatinib is pH dependent, with higher pH resulting in lower solubility. However, esomeprazole, a proton pump inhibitor, administered at a dose of 40 mg once daily for 7 days, did not result in a clinically meaningful reduction in lapatinib steady-state exposure.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category D [see Warnings and Precautions (5.8)].
Based on findings in animals, TYKERB can cause fetal harm when administered to a pregnant woman. Lapatinib administered to rats during organogenesis and through lactation led to death of offspring within the first 4 days after birth. When administered to pregnant animals during the period of organogenesis, lapatinib caused fetal anomalies (rats) or abortions (rabbits) at maternally toxic doses. There are no adequate and well-controlled studies with TYKERB in pregnant women. Women should be advised not to become pregnant when taking TYKERB. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus.
In a study where pregnant rats were dosed with lapatinib during organogenesis and through lactation, at a dose of 120 mg/kg/day (approximately 6.4 times the human clinical exposure based on AUC following 1,250-mg dose of lapatinib plus capecitabine), 91% of the pups had died by the fourth day after birth, while 34% of the 60 mg/kg/day pups were dead. The highest no-effect dose for this study was 20 mg/kg/day (approximately equal to the human clinical exposure based on AUC).
Lapatinib was studied for effects on embryo-fetal development in pregnant rats and rabbits given oral doses of 30, 60, and 120 mg/kg/day. There were no teratogenic effects; however, minor anomalies (left-sided umbilical artery, cervical rib, and precocious ossification) occurred in rats at the maternally toxic dose of 120 mg/kg/day (approximately 6.4 times the human clinical exposure based on AUC following 1,250-mg dose of lapatinib plus capecitabine). In rabbits, lapatinib was associated with maternal toxicity at 60 and 120 mg/kg/day (approximately 0.07 and 0.2 times the human clinical exposure, respectively, based on AUC following 1,250-mg dose of lapatinib plus capecitabine) and abortions at 120 mg/kg/day. Maternal toxicity was associated with decreased fetal body weights and minor skeletal variations.
8.3 Nursing Mothers
It is not known whether lapatinib is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from TYKERB, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
8.4 Pediatric Use
The safety and effectiveness of TYKERB in pediatric patients have not been established.
8.5 Geriatric Use
Of the total number of metastatic breast cancer patients in clinical studies of TYKERB in combination with capecitabine (N = 198), 17% were 65 years of age and older, and 1% were 75 years of age and older. Of the total number of hormone receptor-positive, HER2-positive metastatic breast cancer patients in clinical studies of TYKERB in combination with letrozole (N = 642), 44% were 65 years of age and older, and 12% were 75 years of age and older. No overall differences in safety or effectiveness were observed between elderly subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
8.6 Renal Impairment
Lapatinib pharmacokinetics have not been specifically studied in patients with renal impairment or in patients undergoing hemodialysis. There is no experience with TYKERB in patients with severe renal impairment. However, renal impairment is unlikely to affect the pharmacokinetics of lapatinib given that less than 2% (lapatinib and metabolites) of an administered dose is eliminated by the kidneys.
8.7 Hepatic Impairment
The pharmacokinetics of lapatinib were examined in subjects with pre-existing moderate (n = 8) or severe (n = 4) hepatic impairment (Child-Pugh Class B/C, respectively) and in 8 healthy control subjects. Systemic exposure (AUC) to lapatinib after a single oral 100-mg dose increased approximately 14% and 63% in subjects with moderate and severe pre-existing hepatic impairment, respectively. Administration of TYKERB in patients with severe hepatic impairment should be undertaken with caution due to increased exposure to the drug. A dose reduction should be considered for patients with severe pre-existing hepatic impairment [see Dosage and Administration (2.2)]. In patients who develop severe hepatotoxicity while on therapy, TYKERB should be discontinued and patients should not be retreated with TYKERB [see Warnings and Precautions (5.2)].
10 OVERDOSAGE
There is no known antidote for overdoses of TYKERB. The maximum oral doses of lapatinib that have been administered in clinical trials are 1,800 mg once daily. More frequent ingestion of TYKERB could result in serum concentrations exceeding those observed in clinical trials and could result in increased toxicity. Therefore, missed doses should not be replaced and dosing should resume with the next scheduled daily dose.
Asymptomatic and symptomatic cases of overdose have been reported. The doses ranged from 2,500 to 9,000 mg daily and where reported, the duration varied between 1 and 17 days. Symptoms observed include lapatinib-associated events [see Adverse Reactions (6.1)] and in some cases sore scalp, sinus tachycardia (with otherwise normal ECG), and/or mucosal inflammation.
Because lapatinib is not significantly renally excreted and is highly bound to plasma proteins, hemodialysis would not be expected to be an effective method to enhance the elimination of lapatinib.
Treatment of overdose with TYKERB should consist of general supportive measures.
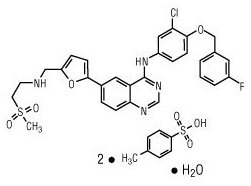
11 DESCRIPTION
Lapatinib is a small molecule and a member of the 4-anilinoquinazoline class of kinase inhibitors. It is present as the monohydrate of the ditosylate salt, with chemical name N-(3-chloro-4-{[(3-fluorophenyl)methyl]oxy}phenyl)-6-[5-({[2-(methylsulfonyl)ethyl]amino}methyl)-2-furanyl]-4-quinazolinamine bis(4-methylbenzenesulfonate) monohydrate. It has the molecular formula C29H26ClFN4O4S (C7H8O3S)2 H2O and a molecular weight of 943.5. Lapatinib ditosylate monohydrate has the following chemical structure:
Lapatinib is a yellow solid, and its solubility in water is 0.007 mg/mL and in 0.1N HCl is 0.001 mg/mL at 25°C.
Each 250 mg tablet of TYKERB contains 405 mg of lapatinib ditosylate monohydrate, equivalent to 398 mg of lapatinib ditosylate or 250 mg lapatinib free base.
The inactive ingredients of TYKERB are: Tablet Core: Magnesium stearate, microcrystalline cellulose, povidone, sodium starch glycolate. Coating: Orange film-coat: FD&C yellow No. 6/sunset yellow FCF aluminum lake, hypromellose, macrogol/PEG 400, polysorbate 80, titanium dioxide.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Lapatinib is a 4-anilinoquinazoline kinase inhibitor of the intracellular tyrosine kinase domains of both Epidermal Growth Factor Receptor (EGFR [ErbB1]) and of Human Epidermal Receptor Type 2 (HER2 [ErbB2]) receptors (estimated Kiapp values of 3nM and 13nM, respectively) with a dissociation half-life of ≥300 minutes. Lapatinib inhibits ErbB-driven tumor cell growth in vitro and in various animal models.
An additive effect was demonstrated in an in vitro study when lapatinib and 5-FU (the active metabolite of capecitabine) were used in combination in the 4 tumor cell lines tested. The growth inhibitory effects of lapatinib were evaluated in trastuzumab-conditioned cell lines. Lapatinib retained significant activity against breast cancer cell lines selected for long-term growth in trastuzumab-containing medium in vitro. These in vitro findings suggest non–cross-resistance between these two agents.
Hormone receptor-positive breast cancer cells (with ER [Estrogen Receptor] and/or PgR [Progesterone Receptor]) that coexpress the HER2 tend to be resistant to established endocrine therapies. Similarly, hormone receptor-positive breast cancer cells that initially lack EGFR or HER2 upregulate these receptor proteins as the tumor becomes resistant to endocrine therapy.
12.3 Pharmacokinetics
Absorption: Absorption following oral administration of TYKERB is incomplete and variable. Serum concentrations appear after a median lag time of 0.25 hours (range 0 to 1.5 hours). Peak plasma concentrations (Cmax) of lapatinib are achieved approximately 4 hours after administration. Daily dosing of TYKERB results in achievement of steady state within 6 to 7 days, indicating an effective half-life of 24 hours.
At the dose of 1,250 mg daily, steady-state geometric mean (95% confidence interval) values of Cmax were 2.43 mcg/mL (1.57 to 3.77 mcg/mL) and AUC were 36.2 mcg.h/mL (23.4 to 56 mcg.h/mL).
Divided daily doses of TYKERB resulted in approximately 2-fold higher exposure at steady state (steady-state AUC) compared to the same total dose administered once daily.
Systemic exposure to lapatinib is increased when administered with food. Lapatinib AUC values were approximately 3- and 4-fold higher (Cmax approximately 2.5- and 3-fold higher) when administered with a low-fat (5% fat-500 calories) or with a high-fat (50% fat-1,000 calories) meal, respectively.
Distribution: Lapatinib is highly bound (>99%) to albumin and alpha-1 acid glycoprotein. In vitro studies indicate that lapatinib is a substrate for the transporters breast cancer-resistance protein (BCRP, ABCG2) and P-glycoprotein (P-gp, ABCB1). Lapatinib has also been shown to inhibit P-gp, BCRP, and the hepatic uptake transporter OATP 1B1, in vitro at clinically relevant concentrations.
Metabolism: Lapatinib undergoes extensive metabolism, primarily by CYP3A4 and CYP3A5, with minor contributions from CYP2C19 and CYP2C8 to a variety of oxidated metabolites, none of which accounts for more than 14% of the dose recovered in the feces or 10% of lapatinib concentration in plasma.
Elimination: At clinical doses, the terminal phase half-life following a single dose was 14.2 hours; accumulation with repeated dosing indicates an effective half-life of 24 hours.
Elimination of lapatinib is predominantly through metabolism by CYP3A4/5 with negligible (<2%) renal excretion. Recovery of parent lapatinib in feces accounts for a median of 27% (range 3% to 67%) of an oral dose.
Effects of Age, Gender, or Race: Studies of the effects of age, gender, or race on the pharmacokinetics of lapatinib have not been performed.
12.4 QT Prolongation
The QT prolongation potential of lapatinib was assessed as part of an uncontrolled, open-label, dose-escalation study in advanced cancer patients. Eighty-one patients received daily doses of lapatinib ranging from 175 mg/day to 1,800 mg/day. Serial ECGs were collected on Day 1 and Day 14 to evaluate the effect of lapatinib on QT intervals. Analysis of the data suggested a consistent concentration-dependent increase in QTc interval.
12.5 Pharmacogenomics
The HLA alleles DQA1*02:01 and DRB1*07:01 were associated with hepatotoxicity reactions in a genetic substudy of a monotherapy trial with TYKERB (n = 1,194). Severe liver injury (ALT >5 times the upper limit of normal, NCI CTCAE Grade 3) occurred in 2% of patients overall; the incidence of severe liver injury among DQA1*02:01 or DRBI*07:01 allele carriers was 8% versus 0.5% in non-carriers. These HLA alleles are present in approximately 15% to 25% of Caucasian, Asian, African, and Hispanic populations and 1% in Japanese populations. Liver function should be monitored in all patients receiving therapy with TYKERB regardless of genotype.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In carcinogenicity studies, lapatinib was administered orally for up to 104 weeks at doses of 75 and 150 mg/kg/day in male mice and 75, 150, and 300 mg/kg/day in female mice (approximately 0.7 to 2 times the expected human clinical exposure based on AUC for a clinical dose of 1,250 mg/day) and 60, 120, 240, and 500 mg/kg/day (approximately 0.6 to 2.3 times the expected human clinical exposure based on AUC) in male rats, and 20, 60, and 180 mg/kg/day (approximately 1.4 to 10 times the expected human clinical exposure based on AUC) in female rats. There was no evidence of carcinogenicity in mice. In male rats, there was an increased incidence of whole body combined hemangiomas and hemangiosarcomas.
Lapatinib was not clastogenic or mutagenic in the Chinese hamster ovary chromosome aberration assay, microbial mutagenesis (Ames) assay, human lymphocyte chromosome aberration assay or the in vivo rat bone marrow chromosome aberration assay at single doses up to 2,000 mg/kg. However, an impurity in the drug product (up to 4 ppm or 8 mcg/day) was genotoxic when tested alone in both in vitro and in vivo assays.
There were no effects on male or female rat mating or fertility at doses up to 120 mg/kg/day in females and 180 mg/kg/day in males (approximately 6.4 times and 2.6 times the expected human clinical exposure based on AUC following 1,250-mg dose of lapatinib plus capecitabine, respectively). The effect of lapatinib on human fertility is unknown. However, when female rats were given oral doses of lapatinib during breeding and through the first 6 days of gestation, a significant decrease in the number of live fetuses was seen at 120 mg/kg/day and in the fetal body weights at ≥60 mg/kg/day (approximately 6.4 times and 3.3 times the expected human clinical exposure based on AUC following 1,250-mg dose of lapatinib plus capecitabine, respectively).
13.2 Animal Toxicology and/or Pharmacology
In 104-week repeat-dose studies in rodents, severe skin lesions that led to lethality were seen at the highest doses tested (300 mg/kg/day) in male mice and female rats. There was also an increase in renal infarcts and papillary necrosis in female rats at ≥60 mg/kg/day and ≥180 mg/kg/day, respectively (approximately 7 and 10 times the expected human clinical exposure based on AUC, respectively). The relevance of these findings for humans is uncertain.
14 CLINICAL STUDIES
14.1 HER2-Positive Metastatic Breast Cancer
The efficacy and safety of TYKERB in combination with capecitabine in breast cancer were evaluated in a randomized, Phase 3 trial. Patients eligible for enrollment had HER2 (ErbB2) overexpressing (IHC 3+ or IHC 2+ confirmed by FISH), locally advanced or metastatic breast cancer, progressing after prior treatment that included anthracyclines, taxanes, and trastuzumab.
Patients were randomized to receive either TYKERB 1,250 mg once daily (continuously) plus capecitabine 2,000 mg/m2/day on Days 1-14 every 21 days, or to receive capecitabine alone at a dose of 2,500 mg/m2/day on Days 1-14 every 21 days. The endpoint was time to progression (TTP). TTP was defined as time from randomization to tumor progression or death related to breast cancer. Based on the results of a pre-specified interim analysis, further enrollment was discontinued. Three hundred and ninety-nine (399) patients were enrolled in this study. The median age was 53 years and 14% were older than 65 years. Ninety-one percent (91%) were Caucasian. Ninety-seven percent (97%) had stage IV breast cancer, 48% were estrogen receptor+ (ER+) or progesterone receptor+ (PR+), and 95% were ErbB2 IHC 3+ or IHC 2+ with FISH confirmation. Approximately 95% of patients had prior treatment with anthracyclines, taxanes, and trastuzumab.
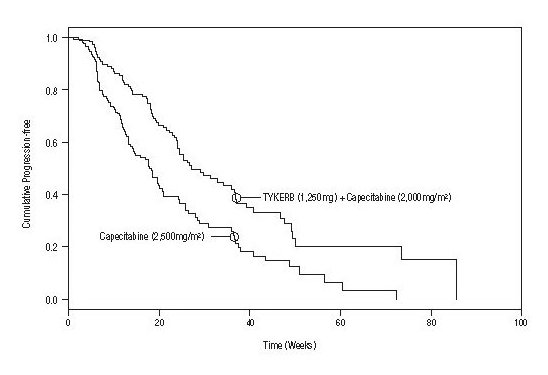
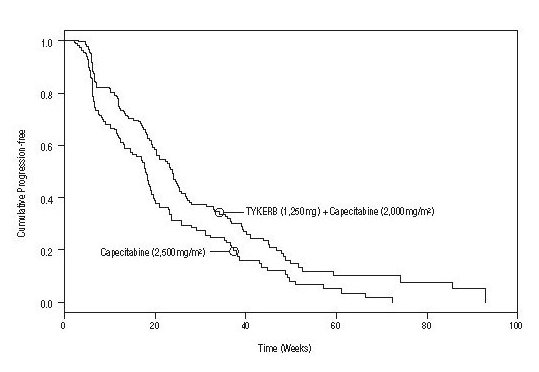
Efficacy analyses 4 months after the interim analysis are presented in Table 5, Figure 1, and Figure 2.
|
Independent Assessmenta |
Investigator Assessment |
|||
|
TYKERB 1,250 mg/day + Capecitabine 2,000 mg/m2/day |
Capecitabine 2,500 mg/m2/day |
TYKERB 1,250 mg/day + Capecitabine 2,000 mg/m2/day |
Capecitabine 2,500 mg/m2/day |
|
|
(N = 198) |
(N = 201) |
(N = 198) |
(N = 201) |
|
|
Number of TTP events |
82 |
102 |
121 |
126 |
|
Median TTP, weeks |
27.1 |
18.6 |
23.9 |
18.3 |
|
(25th, 75th, Percentile), weeks |
(17.4, 49.4) |
(9.1, 36.9) |
(12.0, 44.0) |
(6.9, 35.7) |
|
Hazard Ratio (HR) |
0.57 |
0.72 |
||
|
(95% CI) |
(0.43, 0.77) |
(0.56, 0.92) |
||
|
P value |
0.00013 |
0.00762 |
||
|
Response Rate (%) |
23.7 |
13.9 |
31.8 |
17.4 |
|
(95% CI) |
(18.0, 30.3) |
(9.5, 19.5) |
(25.4, 38.8) |
(12.4, 23.4) |
TTP = Time to progression.
a The time from last tumor assessment to the data cut-off date was >100 days in approximately 30% of patients in the independent assessment. The pre-specified assessment interval was 42 or 84 days.
At the time of above efficacy analysis, the overall survival data were not mature (32% events). However, based on the TTP results, the study was unblinded and patients receiving capecitabine alone were allowed to cross over to treatment with TYKERB plus capecitabine. The survival data were followed for an additional 2 years to be mature and the analysis is summarized in Table 6.
|
TYKERB 1,250 mg/day + Capecitabine 2,000 mg/m2/day (N = 207) |
Capecitabine 2,500 mg/m2/day (N = 201) |
|
|
Overall Survival | ||
|
Died |
76% |
82% |
|
Median Overall Survival (weeks) |
75.0 |
65.9 |
|
Hazard ratio, 95% CI (P value) |
0.89 (0.71, 1.10) 0.276 |
|
CI = confidence interval.
Clinical Studies Describing Limitation of Use: In two randomized trials, TYKERB-based chemotherapy regimens have been shown to be less effective than trastuzumab-based chemotherapy regimens. The first randomized, open-label study compared the safety and efficacy of TYKERB in combination with capecitabine relative to trastuzumab in combination with capecitabine in women with HER2-positive metastatic breast cancer (N = 540). The study was stopped early based on the findings of a pre-planned interim analysis showing a low incidence of CNS events (primary endpoint) and superior efficacy of the trastuzumab plus capecitabine. The median progression-free survival was 6.6 months in the group receiving TYKERB in combination with capecitabine compared with 8.0 months in the group receiving the trastuzumab combination [HR = 1.30 (95% CI: 1.04, 1.64)]. Overall survival was analyzed when 26% of deaths occurred in the group receiving TYKERB in combination with capecitabine and 22% in the group receiving the trastuzumab combination [HR = 1.34 (95% CI: 0.95, 1.92)].
The second randomized, open-label study compared the safety and efficacy of taxane-based chemotherapy plus TYKERB to taxane-based chemotherapy plus trastuzumab as first-line therapy in women with HER2-positive, metastatic breast cancer (N = 652). The study was stopped early based on findings from a pre-planned interim analysis. The median progression-free survival was 11.3 months in the trastuzumab combination treatment arm compared to 9.0 months in patients treated with TYKERB in the combination arm for the intent-to-treat population [HR = 1.37 (95% CI: 1.13, 1.65)].
14.2 Hormone Receptor-Positive, HER2-Positive Metastatic Breast Cancer
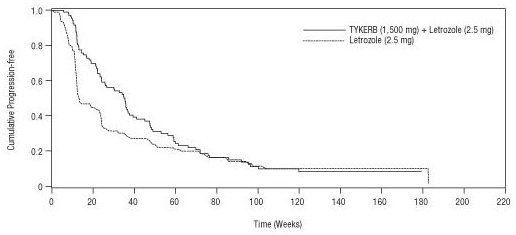
The efficacy and safety of TYKERB in combination with letrozole were evaluated in a double-blind, placebo-controlled, multi-center study. A total of 1,286 postmenopausal women with hormone receptor-positive (ER-positive and/or PgR-positive) metastatic breast cancer, who had not received prior therapy for metastatic disease, were randomly assigned to receive either TYKERB (1,500 mg once daily) plus letrozole (2.5 mg once daily) (n = 642) or letrozole (2.5 mg once daily) alone (n = 644). Of all patients randomized to treatment, 219 (17%) patients had tumors overexpressing the HER2 receptor, defined as fluorescence in situ hybridization (FISH) ≥2 or 3+ immunohistochemistry (IHC). There were 952 (74%) patients who were HER2-negative and 115 (9%) patients did not have their HER2 receptor status confirmed. The primary objective was to evaluate and compare progression-free survival (PFS) in the HER2-positive population. Progression-free survival was defined as the interval of time between date of randomization and the earlier date of first documented sign of disease progression or death due to any cause.
The baseline demographic and disease characteristics were balanced between the two treatment arms. The median age was 63 years and 45% were 65 years of age or older. Eighty-four percent (84%) of the patients were white. Approximately 50% of the HER2-positive population had prior adjuvant/neo-adjuvant chemotherapy and 56% had prior hormonal therapy. Only 2 patients had prior trastuzumab.
In the HER2-positive subgroup (n = 219), the addition of TYKERB to letrozole resulted in an improvement in PFS. In the HER2-negative subgroup, there was no improvement in PFS of the combination of TYKERB plus letrozole compared to the letrozole plus placebo. Overall response rate (ORR) was also improved with the combination of TYKERB plus letrozole. The overall survival (OS) data were not mature. Efficacy analyses for the hormone receptor-positive, HER2-positive and HER2-negative subgroups are presented in Table 7 and Figure 3.
|
HER2-Positive Population |
HER2-Negative Population |
|||
|
TYKERB 1500 mg/day + Letrozole 2.5 mg/day |
Letrozole 2.5 mg/day |
TYKERB 1500 mg/day + Letrozole 2.5 mg/day |
Letrozole 2.5 mg/day |
|
|
(N = 111) |
(N = 108) |
(N = 478) |
(N = 474) |
|
|
Median PFSa, weeks (95% CI) |
35.4 (24.1, 39.4) |
13.0 (12.0, 23.7) |
59.7 (48.6, 69.7) |
58.3 (47.9, 62.0) |
|
Hazard Ratio (95% CI) P value |
0.71 (0.53, 0.96) 0.019 |
0.90 (0.77, 1.05) 0.188 |
||
|
Response Rate (%) (95% CI) |
27.9 (19.8, 37.2) |
14.8 (8.7, 22.9) |
32.6 (28.4, 37.0) |
31.6 (27.5, 36.0) |
PFS = progression-free survival; CI = confidence interval.
a Kaplan-Meier estimate.
16 HOW SUPPLIED/STORAGE AND HANDLING
The 250 mg tablets of TYKERB are oval, biconvex, orange, and film-coated with GS XJG debossed on one side and are available in:
Bottles of 150 tablets: NDC 0173-0752-00
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Patients should be informed of the following:
- •
- TYKERB has been reported to decrease left ventricular ejection fraction which may result in shortness of breath, palpitations, and/or fatigue. Patients should inform their physician if they develop these symptoms while taking TYKERB.
- •
- TYKERB often causes diarrhea which may be severe in some cases. Patients should be told how to manage and/or prevent diarrhea and to inform their physician immediately if there is any change in bowel patterns or severe diarrhea occurs during treatment with TYKERB.
- •
- TYKERB may interact with many drugs; therefore, patients should be advised to report to their healthcare provider the use of any other prescription or nonprescription medication or herbal products.
- •
- TYKERB may interact with grapefruit. Patients should not take TYKERB with grapefruit products.
- •
- TYKERB should be taken at least one hour before or one hour after a meal, in contrast to capecitabine which should be taken with food or within 30 minutes after food.
- •
- The dose of TYKERB should be taken once daily. Dividing the daily dose is not recommended.
TYKERB is a registered trademark of the GlaxoSmithKline group of companies.
GlaxoSmithKline
Research Triangle Park, NC 27709
©2015, GlaxoSmithKline group of companies. All rights reserved.
TKB:15PI
PHARMACIST - DETACH HERE AND GIVE INSTRUCTIONS TO PATIENT
-------------------------------------------------------------------------------------------------------------
PATIENT INFORMATION
TYKERB® (TIE-curb)
(lapatinib)
tablets
Read this leaflet before you start taking TYKERB and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or treatment.
What is TYKERB?
TYKERB is used with the medicine capecitabine for the treatment of people with advanced or metastatic breast cancer that is HER2-positive (tumors that produce large amounts of a protein called human epidermal growth factor receptor-2), and who have already had certain other breast cancer treatments.
TYKERB is also used with a type of medicine called letrozole for the treatment of postmenopausal women with hormone receptor-positive, HER2-positive metastatic breast cancer for whom hormonal therapy is indicated.
It is not known if TYKERB is safe and effective in children.
Who should not take TYKERB?
Do not take TYKERB if you are allergic to any of the ingredients in TYKERB. See the end of this leaflet for a complete list of ingredients in TYKERB.
What should I tell my doctor before taking TYKERB?
Before you take TYKERB, tell your doctor if you:
- •
- have heart problems.
- •
- have liver problems. You may need a lower dose of TYKERB.
- •
- have any other medical conditions.
- •
- are pregnant or plan to become pregnant. TYKERB can harm your unborn baby. You should not become pregnant while taking TYKERB. Tell your doctor right away if you become pregnant during treatment with TYKERB.
- •
- are breastfeeding or plan to breastfeed. It is not known if TYKERB passes into your breast milk. You and your doctor should decide if you will take TYKERB or breastfeed. You should not do both.
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. TYKERB may affect the way other medicines work, and other medicines may affect the way TYKERB works.
Especially tell your doctor if you take:
- •
- antibiotics and anti-fungal medicines (used to treat infections)
- •
- HIV medicines
- •
- medicines used to treat seizures
- •
- medicines used to treat heart problems or high blood pressure
- •
- antidepressants
- •
- medicines that reduce stomach acid (antacids)
- •
- St. John’s wort
Know the medicines you take. Keep a list of your medicines with you to show your doctor and pharmacist when you get a new medicine. Do not take other medicines during treatment with TYKERB without first talking with your doctor.
How should I take TYKERB?
- •
- Take TYKERB exactly as your doctor tells you to take it. Your doctor may change your dose of TYKERB if needed.
- •
- For people with advanced or metastatic breast cancer, TYKERB and capecitabine are taken in 21-day cycles. The usual dose of TYKERB is 1,250 mg (5 tablets) taken by mouth all at once, one time a day on days 1 to 21.
- •
- Your doctor will tell you the dose of capecitabine you should take and when you should take it.
- •
- Take capecitabine with food or within 30 minutes after food.
- •
- For people with hormone receptor-positive, HER2-positive breast cancer, TYKERB and letrozole are taken every day. The usual dose of TYKERB is 1,500 mg (6 tablets) taken by mouth all at once, one time a day. Your doctor will tell you the dose of letrozole you should take and when you should take it.
- •
- TYKERB should be taken at least 1 hour before, or at least 1 hour after a meal.
- •
- Do not eat or drink grapefruit products during treatment with TYKERB.
- •
- If you miss a dose of TYKERB, take your next dose at your regular time the next day.
- •
- If you take too much TYKERB, call your doctor or go to the nearest hospital emergency room right away.
What are the possible side effects of TYKERB?
TYKERB may cause serious side effects, including:
- •
-
heart problems, including decreased pumping of blood from the heart and an abnormal heartbeat. Signs and symptoms of an abnormal heartbeat include:
- •
- feeling like your heart is pounding or racing
- •
- dizziness
- •
- tiredness
- •
- feeling lightheaded
- •
- shortness of breath
- Your doctor should check your heart function before you start taking TYKERB and during treatment.
- •
-
liver problems. Liver problems can be severe and deaths have happened. Signs and symptoms of liver problems include:
- •
- itching
- •
- yellowing of your skin or the white part of your eyes
- •
- dark urine
- •
- pain or discomfort in the right upper stomach area
- Your doctor should do blood tests to check your liver before you start taking TYKERB and during treatment.
- •
- diarrhea. Diarrhea is common with TYKERB and may sometimes be severe. Severe diarrhea can cause loss of body fluid (dehydration) and some deaths have happened. Call your doctor right away if you have a change in bowel pattern or if you have severe diarrhea. Follow your doctor’s instructions for what to do to help prevent or treat diarrhea.
- •
- lung problems. Symptoms of a lung problem with TYKERB include a cough that will not go away or shortness of breath.
- •
- severe skin reactions. TYKERB may cause severe skin reactions. Tell your doctor right away if you develop a skin rash, blisters, or peeling of the skin. As severe skin reactions can be life-threatening, your doctor may tell you to stop taking TYKERB.
Call your doctor right away if you have any of the signs or symptoms of the serious side effects listed above.
Common side effects of TYKERB in combination with capecitabine or letrozole include:
- •
- diarrhea
- •
- red, painful hands and feet
- •
- nausea
- •
- rash
- •
- vomiting
- •
- tiredness or weakness
- •
- mouth sores
- •
- loss of appetite
- •
- indigestion
- •
- unusual hair loss or thinning
- •
- nose bleeds
- •
- headache
- •
- dry skin
- •
- itching
- •
- nail disorders such as nail bed changes, nail pain, infection and swelling of the cuticles.
Tell your doctor if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of TYKERB. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
You may also get side effects from the other medicines taken with TYKERB. Talk to your doctor about possible side effects you may get during treatment.
How should I store TYKERB Tablets?
- •
- Store TYKERB Tablets at room temperature between 68° and 77°F (20° and 25°C).
- •
- Keep the container closed tightly.
- •
- Do not keep medicine that is out of date or that you no longer need.
Keep TYKERB and all medicines out of the reach of children.
General information about TYKERB
Medicines are sometimes prescribed for purposes other than those listed in patient information leaflets. Do not use TYKERB for a condition for which it was not prescribed. Do not give TYKERB to other people, even if they have the same symptoms that you have. It may harm them.
This leaflet summarizes the most important information about TYKERB. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about TYKERB that is written for health professionals.
For more information, call 1-888-825-5249 or go to www.tykerb.com.
What are the ingredients in TYKERB?
Active ingredient: Lapatinib.
Inactive ingredients: Tablet Core: Magnesium stearate, microcrystalline cellulose, povidone, sodium starch glycolate. Coating: Orange film-coat: FD&C yellow No. 6/sunset yellow FCF aluminum lake, hypromellose, macrogol/PEG 400, polysorbate 80, titanium dioxide.
This Patient Information has been approved by the U.S. Food and Drug Administration.
TYKERB is a registered trademark of the GlaxoSmithKline group of companies.
GlaxoSmithKline
Research Triangle Park, NC 27709
©2014, GlaxoSmithKline group of companies. All rights reserved.
Revised: December 2014
TKB:14PIL
PRINCIPAL DISPLAY PANEL
NDC 0173-0752-00
Tykerb®
(lapatinib) Tablets
250 mg
150 Tablets
Rx only
Store at 25oC (77oF); excursions permitted to 15o to 30oC (59o to 86oF).
Each tablet contains 398 mg lapatinib ditosylate equivalent to 250 mg lapatinib.
Dosage: See accompanying prescribing information.
Important: Use safety closures when dispensing this product unless otherwise directed by physicians or requested by purchaser.
GlaxoSmithKline
Research Triangle Park, NC 27709
Made in Ireland
10000000100652
Rev. 11/11
| TYKERB
lapatinib tablet |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| Labeler - GlaxoSmithKline LLC (167380711) |