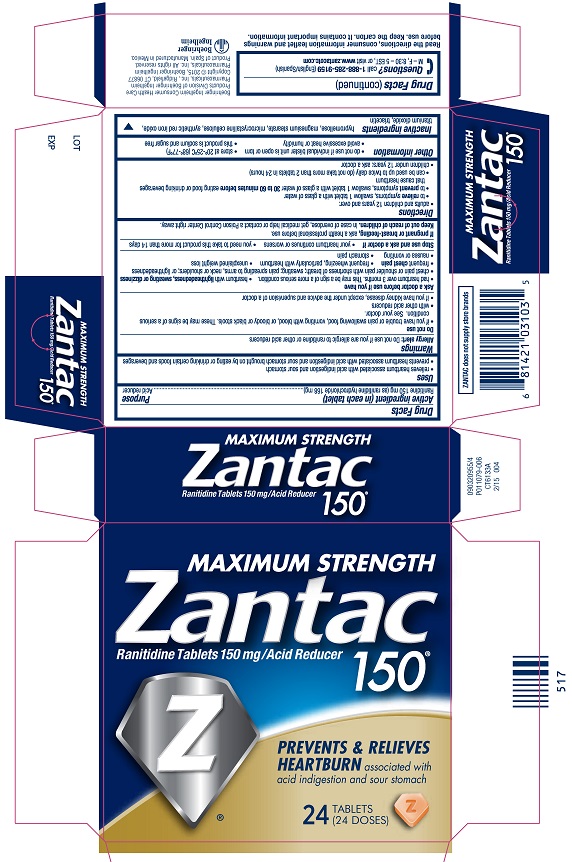
Drug Facts
| Active ingredient (in each tablet) | Purpose |
| Ranitidine 150 mg (as ranitidine hydrochloride 168 mg)...................................................................................................... | Acid reducer |
Uses
- relieves heartburn associated with acid indigestion and
sour stomach
- prevents heartburn associated with acid indigestion and
sour stomach brought on by eating or drinking certain foods and beverages
Warnings
Allergy
alert: Do not use if you are allergic to ranitidine or other
acid reducers
Do not use
- if you have trouble or pain swallowing food, vomiting with
blood, or bloody or black stools. These may be signs of a serious
condition. See your doctor.
- with other acid reducers
- if you have kidney disease, except under the advice and
supervision of a doctor
Ask a doctor before use if you have
- had heartburn over 3 months. This may be a sign of a more
serious condition.
- heartburn with lightheadedness, sweating or dizziness
- chest pain or shoulder pain with shortness of breath; sweating;
pain spreading to arms, neck or shoulders; or lightheadedness
- frequent chest pain
- frequent wheezing, particularly with heartburn
- unexplained weight loss
- nausea or vomiting
- stomach pain
Stop use and ask a doctor if
- your heartburn continues or worsens
- you need to take this product for more than 14 days
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children. In case
of overdose, get medical help or contact a Poison Control Center right
away.
Directions
- adults and children 12 years and over:
- to relieve symptoms, swallow 1 tablet with
a glass of water
- to prevent symptoms, swallow 1 tablet with
a glass of water 30 to 60 minutes before eating food
or drinking beverages that cause heartburn
- can be used up to twice daily (do not take more than 2 tablets
in 24 hours)
- children under 12 years: ask a doctor
Other information
- do not use if printed foil under bottle cap is open or torn
(bottles)
- do not use if individual blister unit is open or torn (blisters)
- do not use if individual foil packet is open or torn (pouch)
- store at 20º-25ºC (68º-77ºF)
- avoid excessive heat or humidity
- this product is sodium and sugar free
Inactive ingredients
hypromellose, magnesium stearate, microcrystalline
cellulose, synthetic red iron oxide, titanium dioxide, triacetin
Questions?
Call 1-888-285-9159 (English/Spanish)
M – F, 8:30 – 5 EST, or visit www.zantacotc.com
Read the directions,
consumer information leaflet and warnings before use. Keep the carton.
It contains important information.
Boehringer Ingelheim Consumer Health Care
Products
Division of Boehringer Ingelheim Pharmaceuticals,
Inc., Ridgefield, CT 06877
Copyright © 2015, Boehringer
Ingelheim Pharmaceuticals, Inc. All rights reserved.
Product
of Spain. Manufactured in Mexico.
Zantac 150 mg - 24
Count Blister Carton
Zantac 150 mg
- 65 Count Bottle Carton
Boehringer Ingelheim Pharmaceuticals Inc.