METHYCLOTHIAZIDE- methyclothiazide tablet
Mylan Pharmaceuticals Inc.
----------
DESCRIPTION
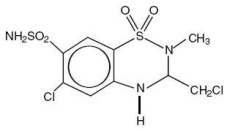
Methyclothiazide, a diuretic-antihypertensive agent, is a member of the benzothiadiazine (thiazide) class of drugs. It is an analogue of hydrochlorothiazide and occurs as a white to practically white crystalline powder which is basically odorless. Methyclothiazide is very slightly soluble in water and chloroform, and slightly soluble in alcohol. Chemically, methyclothiazide is represented as 6-chloro-3-(chloromethyl)-3,4-dihydro-2-methyl-2H-1,2,4-benzothiadiazine-7-sulfonamide 1,1-dioxide. The structural formula is:
Each methyclothiazide tablet, USP for oral administration contains 5 mg methyclothiazide, USP and the following inactive ingredients: colloidal silicon dioxide, FD&C Blue No. 1 Aluminum Lake, magnesium stearate, microcrystalline cellulose, pregelatinized starch, sodium lauryl sulfate and sodium starch glycolate.
CLINICAL PHARMACOLOGY
The diuretic and saluretic effects of methyclothiazide result from a drug-induced inhibition of renal tubular reabsorption of electrolytes. The excretion of sodium and chloride is greatly enhanced. Potassium excretion is also enhanced to a variable degree, as it is with the other thiazides. Although urinary excretion of bicarbonate is increased slightly, there is usually no significant change in urinary pH. Methyclothiazide has a per mg natriuretic activity approximately 100 times that of the prototype thiazide, chlorothiazide. At maximal therapeutic dosages, all thiazides are approximately equal in their diuretic/natriuretic effects.
There is significant natriuresis and diuresis within 2 hours after administration of a single-dose of methyclothiazide. These effects reach a peak in about 6 hours and persist for 24 hours following oral administration of a single-dose.
Like other benzothiadiazines, methyclothiazide also has antihypertensive properties, and may be used for this purpose either alone or to enhance the antihypertensive action of other drugs. The mechanism by which the benzothiadiazines, including methyclothiazide, produce a reduction of elevated blood pressure is not known. However, sodium depletion appears to be involved.
Methyclothiazide is rapidly absorbed and slowly eliminated by the kidneys as intact drug but primarily as an inactive metabolite. Additional information on the pharmacokinetics is not known at this time.
INDICATIONS AND USAGE
Methyclothiazide tablets are indicated in the management of hypertension either as the sole therapeutic agent or to enhance the effect of other antihypertensive drugs in the more severe forms of hypertension.
Methyclothiazide tablets are indicated as adjunctive therapy in edema associated with congestive heart failure, hepatic cirrhosis, and corticosteroid and estrogen therapy.
Methyclothiazide tablets have also been found useful in edema due to various forms of renal dysfunction such as the nephrotic syndrome, acute glomerulonephritis, and chronic renal failure.
Usage in Pregnancy
The routine use of diuretics in an otherwise healthy pregnant woman is inappropriate and exposes mother and fetus to unnecessary hazard. Diuretics do not prevent development of toxemia of pregnancy, and there is no satisfactory evidence that they are useful in the treatment of developed toxemia.
Edema during pregnancy may arise from pathological causes or from the physiological and mechanical consequences of pregnancy. Thiazides are indicated in pregnancy when edema is due to pathological causes, just as they are in the absence of pregnancy (see PRECAUTIONS: Pregnancy). Dependent edema in pregnancy, resulting from restriction of venous return by the expanded uterus, is properly treated through elevation of the lower extremities and use of support hose; use of diuretics to lower intravascular volume in this case is illogical and unnecessary. There is hypervolemia during normal pregnancy that is harmful to neither the fetus nor the mother (in the absence of cardiovascular disease), but that is associated with edema, including generalized edema, in the majority of pregnant women. If this edema produces discomfort, increased recumbency will often provide relief. In rare instances, this edema may cause extreme discomfort that is not relieved by rest. In these cases, a short course of diuretics may provide relief and may be appropriate.
CONTRAINDICATIONS
Methyclothiazide tablets are contraindicated in patients with anuria and in patients with a history of hypersensitivity to this compound or other sulfonamide-derived drugs.
WARNINGS
Methyclothiazide shares with other thiazides the propensity to deplete potassium reserves to an unpredictable degree.
There have been isolated reports that certain non-edematous individuals developed severe fluid and electrolyte derangements after only brief exposure to normal doses of thiazide and non-thiazide diuretics.
Thiazides should be used with caution in patients with renal disease or significant impairment of renal function, since azotemia may be precipitated and cumulative drug effects may occur.
Thiazides should be used with caution in patients with impaired hepatic function or progressive liver disease, since minor alterations of fluid and electrolyte balance may precipitate hepatic coma.
Sensitivity reactions may occur in patients with a history of allergy or bronchial asthma.
The possibility of exacerbation or activation of systemic lupus erythematosus has been reported.
Hyperuricemia may occur or frank gout may be precipitated in certain patients receiving thiazide therapy.
PRECAUTIONS
General
All patients should be observed for clinical signs of electrolyte imbalances such as dryness of mouth, thirst, weakness, lethargy, drowsiness, restlessness, muscle pains or cramps, muscular fatigue, hypotension, oliguria, tachycardia, and gastrointestinal disturbances such as nausea and vomiting.
Hypokalemia may develop, especially with brisk diuresis, when severe cirrhosis is present, during concomitant use of corticosteroids or ACTH, or after prolonged therapy.
Interference with adequate oral electrolyte intake will also contribute to hypokalemia. Hypokalemia may be avoided or treated by use of potassium supplements or foods with a high potassium content.
Any chloride deficit is generally mild and usually does not require specific treatment except under extraordinary circumstances (as in liver disease or renal disease). Dilutional hyponatremia may occur in edematous patients in hot weather; appropriate therapy is water restriction rather than administration of salt, except in rare instances when the hyponatremia is life threatening. In actual salt depletion, appropriate replacement is the therapy of choice.
Latent diabetes mellitus may become manifest during thiazide administration.
The antihypertensive effects of the drug may be enhanced in the postsympathectomy patient.
If progressive renal impairment becomes evident as indicated by a rising nonprotein nitrogen or blood urea nitrogen, a careful reappraisal of therapy is necessary with consideration given to withholding or discontinuing diuretic therapy.
Thiazides may decrease urinary calcium excretion. Thiazides may cause intermittent and slight elevation of serum calcium in the absence of known disorders of calcium metabolism. Marked hypercalcemia may be evidence of hidden hyperparathyroidism. Thiazides should be discontinued before carrying out tests for parathyroid function.
Thiazides may cause increased concentrations of total serum cholesterol, total triglycerides, and low-density lipoproteins in some patients. Use thiazides with caution in patients with moderate or high cholesterol concentrations and in patients with elevated triglyceride levels.
Information for Patients
Patients should inform their doctor if they have:
1) had an allergic reaction to methyclothiazide or other diuretics 2) asthma 3) kidney disease 4) liver disease 5) gout 6) systemic lupus erythematosus, or 7) been taking other drugs such as cortisone, digitalis, lithium carbonate, or drugs for diabetes.
The physician should inform patients of possible side effects and caution the patient to report any of the following symptoms of electrolyte imbalance; dryness of mouth, thirst, weakness, tiredness, drowsiness, restlessness, muscle pains or cramps, nausea, vomiting or increased heart rate.
The physician should advise the patient to take this medication every day as directed. Physicians should also caution patients that drinking alcohol can increase the chance of dizziness.
Laboratory Tests
Initial and periodic determinations of serum electrolytes should be performed at appropriate intervals for the purpose of detecting possible electrolyte imbalances such as hyponatremia, hypochloremic alkalosis, and hypokalemia. Serum and urine electrolyte determinations are particularly important when a patient is vomiting excessively or receiving parenteral fluids.
Drug Interactions
Hypokalemia can sensitize or exaggerate the response of the heart to the toxic effects of digitalis (e.g., increased ventricular irritability).
Hypokalemia may develop during concomitant use of steroids or ACTH.
Insulin requirements in diabetic patients may be increased, decreased, or unchanged.
Thiazides may decrease arterial responsiveness to norepinephrine. This diminution is not sufficient to preclude effectiveness of the pressor agent for therapeutic use.
Thiazide drugs may increase the responsiveness of tubocurarine.
Lithium renal clearance is reduced by thiazides, increasing the risk of lithium toxicity.
Thiazides may add to or potentiate the action of other antihypertensive drugs. Potentiation occurs with ganglionic or peripheral adrenergic blocking drugs.
Drug/Laboratory Test Interactions
Thiazides may decrease serum PBI levels without signs of thyroid disturbance.
Thiazides should be discontinued before carrying out tests for parathyroid function.
Carcinogenesis, Mutagenesis, Impairment of Fertility
No data is available concerning the potential for carcinogenicity or mutagenicity in animals or humans. Methyclothiazide did not impair fertility in rats receiving up to 4 mg/kg/day (at least 20 times the maximum recommended human dose of 10 mg, assuming patient weight equal to or greater than 50 kg).
Pregnancy
Teratogenic Effects. Pregnancy Category B
Reproduction studies performed in rats and rabbits at doses up to 4 mg/kg/day have revealed no evidence of harm to the fetus due to methyclothiazide. There are, however, no adequate and well controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Nonteratogenic Effects
Thiazides cross the placental barrier and appear in cord blood. The use of thiazides in pregnant women requires that the anticipated benefit be weighed against possible hazards to the fetus. These hazards include fetal or neonatal jaundice, thrombocytopenia and possible other adverse reactions that have occurred in the adult.
ADVERSE REACTIONS
Adverse reactions are usually reversible upon reduction of dosage or discontinuation of methyclothiazide tablets. Whenever adverse reactions are moderate or severe, it may be necessary to discontinue the drug.
The following adverse reactions have been observed, but there has not been enough systematic collection of data to support an estimate of their frequency. Consequently the reactions are categorized by organ system and are listed in decreasing order of severity and not frequency.
Body as a Whole: Headache, cramping, weakness.
Cardiovascular System: Orthostatic hypotension (may be potentiated by alcohol, barbiturates, or narcotics).
Digestive System: Pancreatitis, jaundice (intrahepatic cholestatic), sialadenitis, vomiting, diarrhea, nausea, gastric irritation, constipation, anorexia.
Hemic and Lymphatic System: Aplastic anemia, hemolytic anemia, agranulocytosis, leukopenia, thrombocytopenia.
Hypersensitivity Reactions: Anaphylactic reactions, necrotizing angiitis (vasculitis, cutaneous vasculitis), Stevens-Johnson Syndrome, respiratory distress including pneumonitis and pulmonary edema, fever, purpura, urticaria, rash, photosensitivity.
Metabolic and Nutritional Disorders: Hyperglycemia, hyperuricemia, electrolyte imbalance (see PRECAUTIONS section), hypercalcemia.
Nervous System: Vertigo, dizziness, paresthesias, muscle spasm, restlessness.
Special Senses: Transient blurred vision, xanthopsia.
Urogenital System: Glycosuria.
OVERDOSAGE
Symptoms of overdosage include electrolyte imbalance and signs of potassium deficiency such as confusion, dizziness, muscular weakness, and gastrointestinal disturbances. General supportive measures including replacement of fluids and electrolytes may be indicated in treatment of overdosage.
DOSAGE AND ADMINISTRATION
Methyclothiazide tablets are administered orally. Therapy should be individualized according to patient response. This therapy should be titrated to gain maximal therapeutic response as well as the minimal dose possible to maintain that therapeutic response.
For edematous conditions
The usual adult dose ranges from 2.5 mg to 10 mg once daily. Maximum effective single-dose is 10 mg; larger single doses do not accomplish greater diuresis, and are not recommended.
For the treatment of hypertension
The usual adult dose ranges from 2.5 mg to 5 mg once daily.
If control of blood pressure is not satisfactory after 8 to 12 weeks of therapy with 5 mg once daily, another antihypertensive drug should be added. Increasing the dosage of methyclothiazide tablets will usually not result in further lowering of blood pressure.
Methyclothiazide tablets may be either employed alone for mild to moderate hypertension or concurrently with other antihypertensive drugs in the management of more severe forms of hypertension. Combined therapy may provide adequate control of hypertension with lower dosage of the component drugs and fewer or less severe side effects.
When other antihypertensive agents are to be added to the regimen, this should be accomplished gradually. Ganglionic blocking agents should be given at only half the usual dose since their effect is potentiated by pretreatment with methyclothiazide tablets.
HOW SUPPLIED
Methyclothiazide Tablets, USP are available containing 5 mg of methyclothiazide, USP.
The 5 mg tablets are blue tablets debossed with M above the score and 29 below the score on one side of the tablet and blank on the other side. They are available as follows:
NDC 0378-0160-01
bottles of 100 tablets
Store at 20° to 25°C (68° to 77° F). [See USP Controlled Room Temperature.]
Protect from light and moisture.
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure.
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.
REVISED APRIL 2012
METHY:R9
PRINCIPAL DISPLAY PANEL - 5 mg
NDC 0378-0160-01
Methyclothiazide
Tablets, USP
5 mg
Rx only 100 Tablets
Each tablet contains:
Methyclothiazide, USP 5 mg
Dispense in a tight, light-resistant
container as defined in the USP
using a child-resistant closure.
Keep container tightly closed.
Keep this and all medication
out of the reach of children.
Store at 20° to 25°C (68° to 77°F).
[See USP Controlled Room
Temperature.]
Protect from light and moisture.
Usual Adult Dosage: See accom-
panying prescribing information.
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.
Mylan.com
RM0160A7
| METHYCLOTHIAZIDE
methyclothiazide tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Mylan Pharmaceuticals Inc. (059295980) |