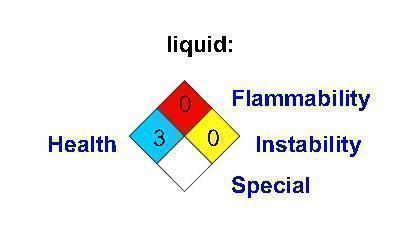
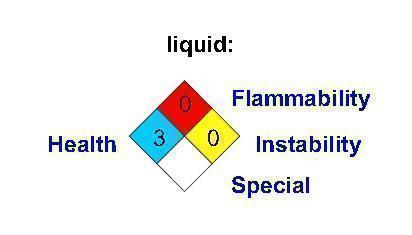
Label: CARBON DIOXIDE gas
-
Contains inactivated NDC Code(s)
NDC Code(s): 63917-001-01 - Packager: Airgas Carbonic, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated February 17, 2014
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CARBON DIOXIDE
carbon dioxide gasProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:63917-001 Route of Administration RESPIRATORY (INHALATION) Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Carbon Dioxide (UNII: 142M471B3J) (Carbon Dioxide - UNII:142M471B3J) Carbon Dioxide 1000 mL in 1 L Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63917-001-01 1 L in 1 TANK Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA205846 12/29/2009 Labeler - Airgas Carbonic, Inc. (041138371) Establishment Name Address ID/FEI Business Operations Airgas Carbonic, Inc. 867448102 manufacture(63917-001) Establishment Name Address ID/FEI Business Operations Airgas Carbonic, Inc. 123380631 manufacture(63917-001) Establishment Name Address ID/FEI Business Operations Airgas Carbonic Inc 024426590 manufacture(63917-001) Establishment Name Address ID/FEI Business Operations Airgas Carbonic, Inc. 019554926 manufacture(63917-001) Establishment Name Address ID/FEI Business Operations Airgas Carbonic 032001558 manufacture(63917-001)