PANDEL- hydrocortisone probutate cream
PharmaDerm a division of Fougera Pharmaceuticals Inc.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use PANDEL safely and effectively. See full prescribing information for PANDEL.
PANDEL (hydrocortisone probutate) cream, for topical use Initial U.S. Approval: 1997 INDICATIONS AND USAGEPANDEL® (hydrocortisone probutate) Cream, 0.1% is a corticosteroid indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses in patients 18 years of age or older. (1) DOSAGE AND ADMINISTRATIONDOSAGE FORMS AND STRENGTHSCream, 0.1%. (3) CONTRAINDICATIONSNone. (4) WARNINGS AND PRECAUTIONS
ADVERSE REACTIONS• Most frequent adverse reactions include burning, stinging, rash, papulovesicular rash, redness, itching, moderate paresthesia, and contact dermatitis. (6) To report SUSPECTED ADVERSE REACTIONS, contact PharmaDerm®, A division of Fougera Pharmaceuticals Inc. at 1-800-645-9833 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling. Revised: 8/2020 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
PANDEL® (hydrocortisone probutate) Cream, 0.1% is indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses in patients 18 years of age or older.
2 DOSAGE AND ADMINISTRATION
Apply a thin film of PANDEL to the affected area once or twice a day depending on the severity of the condition. Massage gently until the medication disappears.
Occlusive dressings may be used for the management of refractory lesions of psoriasis and other deep-seated dermatoses, such as localized neurodermatitis (lichen simplex chronicus).
Discontinue PANDEL when control is achieved. If no improvement is seen within 2 weeks, reassessment of the diagnosis may be necessary.
Do not use PANDEL with occlusive dressings unless directed by the physician. Do not apply PANDEL in the diaper area, as diapers or plastic pants may constitute occlusive dressings.
3 DOSAGE FORMS AND STRENGTHS
Cream, 0.1%. Each gram of PANDEL contains 1 mg of hydrocortisone probutate in a cream base.
5 WARNINGS AND PRECAUTIONS
5.1 Hypothalamic-Pituitary-Adrenal (HPA) Axis Suppression and Other Unwanted Systemic Glucocorticoid Effects
PANDEL can produce reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for glucocorticosteroid insufficiency. This may occur during or after withdrawal of treatment. Factors that predispose to HPA axis suppression include the use of high-potency corticosteroids, large treatment surface areas, prolonged use, use of occlusive dressings, altered skin barrier, liver failure, and young age.
Use of topical corticosteroids may require periodic evaluation for HPA axis suppression. Evaluation for HPA axis suppression may be done by using the adrenocorticotropic hormone (ACTH) stimulation test.
If HPA axis suppression is documented, gradually withdraw the drug, reduce the frequency of application, or substitute with a less potent corticosteroid. If signs and symptoms of steroid withdrawal occur, supplemental systemic corticosteroids may be required. Recovery of HPA axis function is generally prompt and complete upon discontinuation of the drug.
In a trial including 15 evaluable subjects 18 years of age or older with psoriasis or atopic dermatitis affecting more than 20% of body surface area, 1 subject (6.7%) had ACTH stimulation test results suggestive of adrenal suppression after treatment with PANDEL twice daily for 21 days. Recovery of HPA axis suppression for this subject is unknown [see Clinical Pharmacology (12.2)].
Systemic effects of topical corticosteroids may also manifest as Cushing’s syndrome, hyperglycemia, and unmasking latent diabetes mellitus.
Patients applying a topical steroid to a large surface area or to areas under occlusion should be evaluated periodically for evidence of HPA-axis suppression.
Pediatric patients may be more susceptible to systemic toxicity due to their larger skin surface to body mass ratios [see Use in Specific Populations (8.4)].
5.2 Allergic Contact Dermatitis
Allergic contact dermatitis with corticosteroids is usually diagnosed by observing a failure to heal rather than noting a clinical exacerbation, as observed with most topical products not containing corticosteroids. If irritation develops, discontinue PANDEL and institute appropriate therapy.
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The most frequent adverse reactions reported for PANDEL during clinical trials were application site reactions, including burning in 4, stinging in 2, and moderate paresthesia in 1 out of 226 subjects.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of PANDEL because these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
These adverse reactions are as follows:
Skin and Subcutaneous Tissue Disorders: rash, papulovesicular rash
Application Site Reactions: dryness, erythema, pruritus, allergic contact dermatitis.
The following local adverse reactions are reported with topical corticosteroids, and they may occur more frequently with the use of occlusive dressings. These reactions are listed in an approximate decreasing order of occurrence: itching, irritation, dryness, folliculitis, hypertrichosis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, secondary infections, skin atrophy, striae, and miliaria.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There is no clinical information on PANDEL use in pregnant women to inform any drug-associated risk for major birth defects and miscarriage. In animal reproduction studies, hydrocortisone probutate given by the subcutaneous route during the period of organogenesis was teratogenic at doses equal to or greater than 1 mg/kg/day in rats or 0.1 mg/kg/day in rabbits (12 times and 2 times the human topical dose, respectively) [see Data].
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
Effects on embryo-fetal development were evaluated in rats and rabbits following subcutaneous administration of hydrocortisone probutate during the period of organogenesis. Hydrocortisone probutate was teratogenic in rats when given during the period of organogenesis at subcutaneous doses equal to or greater than 1 mg/kg/day (12 times the human average topical dose of PANDEL assuming 3% absorption and an application of 30 g/day on a 70 kg individual). Abnormalities included delayed ossification of the caudal vertebrae and other skeletal variations, cleft palate, umbilical hernia, edema, and exencephalia.
In rabbits, hydrocortisone probutate given by the subcutaneous route was teratogenic at doses equal to or greater than 0.1 mg/kg/day (2 times the human average topical dose of PANDEL assuming 3% absorption and an application of 30 g/day on a 70 kg individual). Fetal weight and survival were affected. Delayed ossification and increased incidences of malformations (skeletal abnormalities and cleft palate) were also noted.
No adverse effects were seen in rats following subcutaneous administration of up to 1 mg/kg/day of hydrocortisone probutate during the perinatal and postnatal period (12 times the human average topical dose of PANDEL assuming 3% absorption and an application of 30 g/day on a 70 kg individual).
8.2 Lactation
Risk Summary
There is no information on the presence of hydrocortisone probutate in breast milk, or on its effects on the breastfed infant or on milk production. It is not known whether topical administration of PANDEL could result in sufficient systemic absorption to produce detectable quantities in human milk. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for PANDEL and any potential adverse effects on the breastfed infant from PANDEL or from the underlying maternal condition.
Clinical Considerations
To minimize potential exposure to the breastfed infant via breast milk, use PANDEL on the smallest area of skin and for the shortest duration possible while breastfeeding.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established. Because of a higher ratio of skin surface area to body mass, pediatric patients are at a greater risk than adults of HPA axis suppression and Cushing’s syndrome when they are treated with topical corticosteroids. They are therefore also at a greater risk of adrenal insufficiency during and/or after withdrawal of treatment. Adverse effects including striae have been reported with inappropriate use of topical corticosteroids in infants and children.
Hypothalamic-pituitary-adrenal (HPA) axis suppression, Cushing’s syndrome, linear growth retardation, delayed weight gain, and intracranial hypertension have been reported in children receiving topical corticosteroids. Manifestations of adrenal suppression in children include low plasma cortisol levels and an absence of response to ACTH stimulation. Manifestations of intracranial hypertension include bulging fontanelles, headaches, and bilateral papilledema.
11 DESCRIPTION
PANDEL(hydrocortisone probutate) Cream, 0.1% contains hydrocortisone probutate, a synthetic corticosteroid. The topical corticosteroids constitute a class of primarily synthetic steroids used as anti-inflammatory and anti-pruritic agents.
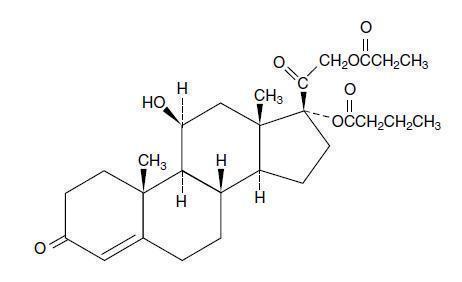
Hydrocortisone probutate is a tasteless and odorless white crystalline powder practically insoluble in hexane or water, slightly soluble in ether, and very soluble in dichloromethane, methanol and acetone. Chemically, it is 11β,17,21-trihydroxypregn-4-ene-3,20-dione 17-butyrate 21-propionate. The structural formula is:
Molecular Formula: C28H40O7
Molecular Weight: 488.62
Each gram of PANDEL (hydrocortisone probutate) Cream, 0.1% contains: 1 mg of hydrocortisone probutate in a cream base of propylene glycol, white petrolatum, light mineral oil, stearyl alcohol, polysorbate 60, sorbitan monostearate, glyceryl monostearate, PEG-20 stearate, glyceryl stearate SE, methylparaben, butylparaben, citric acid, sodium citrate anhydrous, and purified water.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Corticosteroids play a role in cellular signaling, immune function, inflammation, and protein regulation; however, the precise mechanism of action in corticosteroid responsive dermatoses is unknown.
12.2 Pharmacodynamics
Vasoconstrictor Assay
Studies performed with PANDEL indicate that it is in the medium range of potency as demonstrated in vasoconstrictor trials in healthy subjects when compared with other topical corticosteroids. However, similar blanching scores do not necessarily imply therapeutic equivalence.
Hypothalamic-Pituitary-Adrenal (HPA) Axis Suppression
In an open label HPA axis suppression trial, 19 adult subjects (ages 23 to 82 years) with atopic dermatitis or plaque psoriasis covering greater than 20% Body Surface Area (BSA) were treated with PANDEL twice daily for 21 days and were assessed for HPA axis suppression. At baseline, the mean disease BSA involvement was 36%. The criterion for HPA axis suppression was a serum cortisol level of less than or equal to 18 micrograms per deciliter at 30-minutes after cosyntropin stimulation. Of these subjects, 15 were considered evaluable with respect to their adrenal axis function post-treatment. One of the evaluable subjects (6.7%) showed laboratory evidence of suppression on Day 22. This subject had psoriasis covering 48% of BSA at baseline and was reported to have received 98% of the twice-daily applications of PANDEL over the 21 day treatment period. It is not known if this subject had recovery of adrenal function because follow-up testing was not performed.
12.3 Pharmacokinetics
The extent of percutaneous absorption of topical corticosteroids is determined by many factors, including the vehicle and the integrity of the epidermal barrier. Use of occlusive dressings with hydrocortisone for up to 24 hours has not been shown to increase penetration; however, occlusion of hydrocortisone for 96 hours does markedly enhance penetration. Topical corticosteroids can be absorbed from normal intact skin. Inflammation and/or other disease processes in the skin increase percutaneous absorption.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term studies in animals have been performed to evaluate the carcinogenic potential of hydrocortisone probutate.
Hydrocortisone probutate revealed no evidence of mutagenic or clastogenic potential based on the results of an in vitro genotoxicity test (Ames assay) and an in vivo genotoxicity test (mouse micronucleus assay).
Effects on fertility and early embryonic development were evaluated in rats following subcutaneous administration of up to 0.4 mg/kg/day hydrocortisone probutate (5 times the human average topical dose of PANDEL assuming 3% absorption and an application of 30 g/day on a 70 kg individual) prior to and during mating and through early pregnancy. No treatment related effects on fertility or mating parameters were noted in this study.
16 HOW SUPPLIED/STORAGE AND HANDLING
PANDEL, a white to off-white opaque cream is supplied as follows:
80 g tubes NDC 10337-153-80
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
17 PATIENT COUNSELING INFORMATION
Advise the patient and/or caregiver to read the FDA-approved patient labeling (Patient Information).
Inform patients and/or caregivers of the following:
- 1.
- Discontinue therapy when control is achieved unless directed otherwise by the physician.
- 2.
- If no improvement is seen within two weeks, contact the physician.
- 3.
- Avoid contact with the eyes.
- 4.
- Do not use with occlusive dressing unless directed by the physician.
- 5.
- Report any signs or symptoms of local or systemic adverse reactions to the physician.
- 6.
- Do not treat diaper dermatitis. Do not apply PANDEL in the diaper area as diapers or plastic pants may constitute occlusive dressings.
- 7.
- Do not use on the face, underarms, or groin areas unless directed by the physician.
- 8.
- Advise a woman to use PANDEL on the smallest area of skin and for the shortest duration possible while breastfeeding.
Manufactured by:
PharmaDerm®
A division of Fougera
PHARMACEUTICALS INC.
Melville, New York 11747 www.pharmaderm.com
46275355A /46275356A
R08/2020
|
PATIENT INFORMATION PANDEL® (pan-del) (hydrocortisone probutate cream)
|
|
Important: PANDEL is for use on skin only (topical). Avoid using PANDEL near or around your eyes. |
|
What is PANDEL? PANDEL is a prescription corticosteroid medicine used on the skin (topical) for the relief of inflammation and itching caused by certain skin conditions in people 18 years of age or older. It is not known if PANDEL is safe and effective in children. |
|
Before using PANDEL tell your healthcare provider about all of your medical conditions, including if you: • have adrenal gland problems • have liver problems • have diabetes • have thinning skin (atrophy) at the site to be treated. • are pregnant or plan to become pregnant. It is not known if PANDEL will harm your unborn baby. • are breastfeeding or plan to breastfeed. It is not known if PANDEL can pass into your breast milk and harm your baby.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. |
|
How should I use PANDEL? • Use PANDEL exactly as your healthcare provider tells you to use it. • Apply a thin film to the affected skin area. Gently rub PANDEL into your skin until it disappears. • Tell your healthcare provider if your symptoms do not improve after 2 weeks of treatment. • Do not bandage, cover, or wrap the treated area unless your healthcare provider tells you to. • Do not apply PANDEL in the diaper area or use with plastic pants. • Do not use PANDEL on your face, underarms (armpits) or groin areas unless your healthcare provider tells you to. • Wash your hands after applying PANDEL, unless your hands are being treated. |
|
What are possible side effects with PANDEL? PANDEL may cause serious side effects, including: • PANDEL can pass through your skin and may cause adrenal gland problems. This is more likely to happen if you use PANDEL for too long, use it over a large treatment area, use it with other topical medicines that contain corticosteroids, cover the treated area, or have liver failure. Your healthcare provider may do blood tests to check your adrenal gland function during and after treatment with PANDEL. • Skin problems, including skin reactions or thinning of your skin (atrophy), skin infections, and allergic reactions (allergic contact dermatitis) at the treatment site. Tell your healthcare provider if you get any skinreactions such as pain, tenderness, swelling, or healing problems. The most common side effects of PANDEL include burning and stinging and moderate tingling or prickling feeling. These are not all the possible side effects with PANDEL. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store PANDEL? Store PANDEL between 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Keep PANDEL and all medicines out of the reach of children. |
|
General information about the safe and effective use of PANDEL. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use PANDEL for a condition for which it was not prescribed. Do not give PANDEL to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about PANDEL that is written for health professionals. |
|
What are the ingredients in PANDEL? Active ingredient: hydrocortisone probutate Inactive ingredients: propylene glycol, white petrolatum, light mineral oil, stearyl alcohol, polysorbate 60, sorbitan monostearate, glyceryl monostearate, PEG-20 stearate, glyceryl stearate SE, methylparaben, butylparaben, citric acid, sodium citrate anhydrous, and purified water. Manufactured by: PharmaDerm® A division of Fougera PHARMACEUTICALS INC. Melville, New York 11747 For more information, go to www.pharmaderm.com or call 1-800-645-9833. |
- This Patient Information has been approved by the U.S. Food and Drug Administration. Issued: 08/2020
| PANDEL
hydrocortisone probutate cream |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| Labeler - PharmaDerm a division of Fougera Pharmaceuticals Inc. (043838424) |