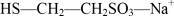Label: MESNA injection, solution
- NDC Code(s): 25021-201-10, 25021-201-11
- Packager: Sagent Pharmaceuticals
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated January 14, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use MESNA INJECTION safely and effectively. See full prescribing information for MESNA INJECTION.
MESNA injection, for intravenous use
Initial U.S. Approval: 1988INDICATIONS AND USAGE
Mesna Injection is a cytoprotective agent indicated as a prophylactic agent in reducing the incidence of ifosfamide-induced hemorrhagic cystitis. (1)
Limitation of Use:
Mesna Injection is not indicated to reduce the risk of hematuria due to other pathological conditions such as thrombocytopenia. (1)
DOSAGE AND ADMINISTRATION
Mesna injection may be given on a fractionated dosing schedule of three bolus intravenous injections or a single bolus injection followed by two oral administrations of mesna tablets as outlined below. The dosing schedule should be repeated on each day that ifosfamide is administered. When the dosage of ifosfamide is adjusted, the ratio of mesna to ifosfamide should be maintained. (2)
Intravenous Dosing Schedule:
0 Hours 4 Hours 8 Hours Ifosfamide 1.2 g/m2 - - Mesna injection 240 mg/m2 240 mg/m2 240 mg/m2 Intravenous and Oral Dosing Schedule:
0 Hours 2 Hours 6 Hours Ifosfamide 1.2 g/m2 - - Mesna injection 240 mg/m2 - - Mesna tablets - 480 mg/m2 480 mg/m2 Maintain sufficient urinary output, as required for ifosfamide treatment, and monitor urine for the presence of hematuria. (2.3)
DOSAGE FORMS AND STRENGTHS
- Mesna Injection: 1 gram (100 mg per mL) Multi-Dose vials (3)
CONTRAINDICATIONS
- Known hypersensitivity to mesna or to any of the excipients in mesna, including benzyl alcohol. (4)
WARNINGS AND PRECAUTIONS
- Hypersensitivity reactions: Anaphylactic reactions have been reported. Less severe hypersensitivity reactions may also occur. Monitor patients. If a reaction occurs, discontinue mesna and provide supportive care. (5.1)
- Dermatologic toxicity: Skin rash with eosinophilia and systemic symptoms, Stevens-Johnson syndrome, and toxic epidermal necrolysis have occurred. Skin rash, urticaria, and angioedema have also been seen. Monitor patients. If a reaction occurs, discontinue mesna and provide supportive care. (5.2)
- Benzyl alcohol toxicity: Serious and fatal adverse reactions can occur in premature neonates and low-birth weight infants treated with benzyl alcohol-preserved drugs, including mesna injection. Avoid use in premature neonates and low-birth weight infants. (5.3)
- Laboratory test alterations: False positive tests for urinary ketones and interference with enzymatic CPK activity tests have been seen. (5.4)
ADVERSE REACTIONS
The most common adverse reactions (> 10%) when mesna is given with ifosfamide are nausea, vomiting, constipation, leukopenia, fatigue, fever, anorexia, thrombocytopenia, anemia, granulocytopenia, diarrhea, asthenia, abdominal pain, headache, alopecia, and somnolence. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Sagent Pharmaceuticals, Inc. at 1-866-625-1618 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
USE IN SPECIFIC POPULATIONS
- Pregnancy: Mesna in combination with ifosfamide can cause fetal harm. Advise patients of potential risk to a fetus. (8.1)
- Lactation: Do not breastfeed. (8.2)
- Females and Males of Reproductive Potential: Advise patients to use effective contraception. Verify pregnancy status prior to initiation of mesna in combination with ifosfamide. (8.3)
- Pediatric use: In premature neonates and low-birth weight infants, avoid use of benzyl alcohol–containing solutions. (8.4)
- Geriatric use: Dose selection should be cautious. (8.5)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 1/2021
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Intravenous Dosing
2.2 Intravenous and Oral Dosing
2.3 Monitoring for Hematuria
2.4 Preparation for Intravenous Administration and Stability
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions
5.2 Dermatologic Toxicity
5.3 Benzyl Alcohol Toxicity
5.4 Laboratory Test Interferences
5.5 Use in Patients with a History of Adverse Reactions to Thiol Compounds
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Use in Patients with Renal Impairment
8.7 Use in Patients with Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Intravenous Mesna
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Intravenous Dosing
Mesna injection may be given on a fractionated dosing schedule of three bolus intravenous injections as outlined below.
Mesna injection is given as intravenous bolus injections in a dosage equal to 20% of the ifosfamide dosage weight by weight (w/w) at the time of ifosfamide administration and 4 and 8 hours after each dose of ifosfamide. The total daily dose of mesna injection is 60% of the ifosfamide dose. The recommended dosing schedule is outlined below in Table 1.
Table 1. Recommended Intravenous Dosing Schedule 1The dosing schedule should be repeated on each day that ifosfamide is administered. When the dosage of ifosfamide is increased or decreased, the ratio of mesna to ifosfamide should be maintained.
0 Hours 4 Hours 8 Hours Ifosfamide 1.2 g/m2 - - Mesna injection1 240 mg/m2 240 mg/m2 240 mg/m2 2.2 Intravenous and Oral Dosing
Mesna injection may be given on a fractionated dosing schedule of a single bolus injection followed by two oral administrations of mesna tablets as outlined below.
Mesna injection is given as intravenous bolus injections in a dosage equal to 20% of the ifosfamide dosage (w/w) at the time of ifosfamide administration. Mesna tablets are given orally in a dosage equal to 40% of the ifosfamide dose 2 and 6 hours after each dose of ifosfamide. The total daily dose of mesna is 100% of the ifosfamide dose. The recommended dosing schedule is outlined in Table 2.
Table 2. Recommended Intravenous and Oral Dosing Schedule 1The dosing schedule should be repeated on each day that ifosfamide is administered. When the dosage of ifosfamide is increased or decreased, the ratio of mesna to ifosfamide should be maintained.
0 Hours 2 Hours 6 Hours Ifosfamide 1.2 g/m2 - - Mesna injection1 240 mg/m2 - - Mesna tablets - 480 mg/m2 480 mg/m2 The efficacy and safety of this ratio of intravenous and oral mesna has not been established as being effective for daily doses of ifosfamide higher than 2 g/m2.
Patients who vomit within two hours of taking oral mesna should repeat the dose or receive intravenous mesna injection.
2.3 Monitoring for Hematuria
Maintain adequate hydration and sufficient urinary output, as required for ifosfamide treatment, and monitor urine for the presence of hematuria. If severe hematuria develops when mesna injection is given according to the recommended dosage schedule, dosage reductions or discontinuation of ifosfamide therapy may be required.
2.4 Preparation for Intravenous Administration and Stability
Preparation
Determine the volume of mesna injection for the intended dose.
Dilute the volume of mesna injection for the dose in any of the following fluids to obtain a final concentration of 20 mg per mL:
- 5% Dextrose Injection, USP
- 5% Dextrose and 0.2% Sodium Chloride Injection, USP
- 5% Dextrose and 0.33% Sodium Chloride Injection, USP
- 5% Dextrose and 0.45% Sodium Chloride Injection, USP
- 0.9% Sodium Chloride Injection, USP
- Lactated Ringer's Injection, USP
Stability
The mesna injection multidose vials may be stored and used for up to 8 days after initial puncture.
Store diluted solutions at 25°C (77°F). Use diluted solutions within 24 hours.
Do not mix mesna injection with epirubicin, cyclophosphamide, cisplatin, carboplatin, and nitrogen mustard.
The benzyl alcohol contained in mesna injection vials can reduce the stability of ifosfamide. Ifosfamide and mesna injection may be mixed in the same bag provided the final concentration of ifosfamide does not exceed 50 mg per mL. Higher concentrations of ifosfamide may not be compatible with mesna injection and may reduce the stability of ifosfamide.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit. Any solutions which are discolored, hazy, or contain visible particulate matter should not be used.
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
Mesna is contraindicated in patients known to be hypersensitive to mesna or to any of the excipients [see Warnings and Precautions (5.1)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions
Mesna may cause systemic hypersensitivity reactions, including anaphylaxis. These reactions may include fever, cardiovascular symptoms (hypotension, tachycardia), acute renal impairment, hypoxia, respiratory distress, urticaria, angioedema, laboratory signs of disseminated intravascular coagulation, hematological abnormalities, increased liver enzymes, nausea, vomiting, arthralgia, and myalgia. These reactions may occur with the first exposure or after several months of exposure. Monitor for signs or symptoms. Discontinue mesna and provide supportive care.
5.2 Dermatologic Toxicity
Drug rash with eosinophilia and systemic symptoms and bullous and ulcerative skin and mucosal reactions, consistent with Stevens-Johnson syndrome or toxic epidermal necrolysis have occurred. Mesna may cause skin and mucosal reactions characterized by urticaria, rash, erythema, pruritus, burning sensation, angioedema, periorbital edema, flushing and stomatitis. These reactions may occur with the first exposure or after several months of exposure. Discontinue mesna and provide supportive care.
5.3 Benzyl Alcohol Toxicity
Serious adverse reactions including fatal reactions and the “gasping syndrome” occurred in premature neonates and low-birth weight infants who received benzyl alcohol dosages of 99 to 234 mg/kg/day (blood levels of benzyl alcohol were 0.61 to 1.378 mmol/L). Symptoms associated with “gasping syndrome” and other potential adverse reactions include gradual neurological deterioration, seizures, intracranial hemorrhage, hematological abnormalities, skin breakdown, hepatic and renal failure, hypotension, bradycardia, and cardiovascular collapse. Premature neonates and low-birth weight infants may be more likely to develop these reactions because they may be less able to metabolize benzyl alcohol. The minimum amount of benzyl alcohol at which toxicity may occur is not known. Mesna injection contains 10.4 mg/mL of the preservative benzyl alcohol. Avoid use of mesna injection in premature neonates and low-birth weight infants. Mesna tablets do not contain benzyl alcohol [see Use in Specific Populations (8.4)].
5.4 Laboratory Test Interferences
False-Positive Urine Tests for Ketone Bodies
A false positive test for urinary ketones may arise in patients treated with mesna when using nitroprusside sodium-based urine tests (including dipstick tests). The addition of glacial acetic acid can be used to differentiate between a false positive result (cherry-red color that fades) and a true positive result (red-violet color that intensifies).
5.5 Use in Patients with a History of Adverse Reactions to Thiol Compounds
Mesna is a thiol compound, i.e., a sulfhydryl (SH) group-containing organic compound. Hypersensitivity reactions to mesna and to amifostine, another thiol compound, have been reported. It is not clear whether patients who experienced an adverse reaction to a thiol compound are at increased risk for a hypersensitivity reaction to mesna.
-
6 ADVERSE REACTIONS
The following are discussed in more detail in other sections of the labeling.
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Dermatological Toxicity [see Warnings and Precautions (5.2)]
- Benzyl Alcohol Toxicity [see Warnings and Precautions (5.3)]
- Laboratory Test Interferences [see Warnings and Precautions (5.4)]
- Use in Patients with a History of Adverse Reactions to Thiol Compounds [see Warnings and Precautions (5.5)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Mesna adverse reaction data are available from four Phase 1 studies in which single intravenous doses of 600 to 1200 mg mesna injection without concurrent chemotherapy were administered to a total of 53 healthy volunteers. The most frequently reported side effects (observed in two or more healthy volunteers) for healthy volunteers receiving single doses of mesna injection alone were headache, injection site reactions, flushing, dizziness, nausea, vomiting, somnolence, diarrhea, anorexia, fever, pharyngitis, hyperesthesia, influenza-like symptoms, and coughing. In two Phase 1 multiple-dose studies where healthy volunteers received mesna tablets alone or intravenous mesna followed by repeated doses of mesna tablets, flatulence and rhinitis were reported. In addition, constipation was reported by healthy volunteers who had received repeated doses of intravenous mesna.
Additional adverse reactions in healthy volunteers receiving mesna alone included injection site reactions, abdominal pain/colic, epigastric pain/burning, mucosal irritation, lightheadedness, back pain, arthralgia, myalgia, conjunctivitis, nasal congestion, rigors, paresthesia, photophobia, fatigue, lymphadenopathy, extremity pain, malaise, chest pain, dysuria, pleuritic pain, dry mouth, dyspnea, and hyperhidrosis. In healthy volunteers, mesna was commonly associated with a rapid (within 24 hours) decrease in lymphocyte count, which was generally reversible within one week of administration.
Because mesna is used in combination with ifosfamide or ifosfamide-containing chemotherapy regimens, it is difficult to distinguish the adverse reactions which may be due to mesna from those caused by the concomitantly administered cytotoxic agents.
Adverse reactions reasonably associated with mesna administered intravenously and orally in four controlled studies in which patients received ifosfamide or ifosfamide-containing regimens are presented in Table 3.
Table 3: Adverse Reactions in ≥5% of Patients Receiving Mesna in combination with Ifosfamide-containing Regimens 1Intravenous dosing of ifosfamide and mesna followed by either intravenous or oral doses of mesna according to the applicable dosage schedule [see Dosage and Administration (2)].
Mesna Regimen Intravenous-Intravenous-Intravenous1 Intravenous-Oral-Oral1 N exposed 119 (100.0%) 119 (100%) Incidence of AEs 101 (84.9%) 106 (89.1%) Nausea 65 (54.6) 64 (53.8) Vomiting 35 (29.4) 45 (37.8) Constipation 28 (23.5) 21 (17.6) Leukopenia 25 (21.0) 21 (17.6) Fatigue 24 (20.2) 24 (20.2) Fever 24 (20.2) 18 (15.1) Anorexia 21 (17.6) 19 (16.0) Thrombocytopenia 21 (17.6) 16 (13.4) Anemia 20 (16.8) 21 (17.6) Granulocytopenia 16 (13.4) 15 (12.6) Asthenia 15 (12.6) 21 (17.6) Abdominal Pain 14 (11.8) 18 (15.1) Alopecia 12 (10.1) 13 (10.9) Dyspnea 11 (9.2) 11 (9.2) Chest Pain 10 (8.4) 11 (9.2) Hypokalemia 10 (8.4) 11 (9.2) Diarrhea 9 (7.6) 17 (14.3) Dizziness 9 (7.6) 5 (4.2) Headache 9 (7.6) 13 (10.9) Pain 9 (7.6) 10 (8.4) Sweating Increased 9 (7.6) 2 (1.7) Back Pain 8 (6.7) 6 (5.0) Hematuria 8 (6.7) 7 (5.9) Injection Site Reaction 8 (6.7) 10 (8.4) Edema 8 (6.7) 9 (7.6) Edema Peripheral 8 (6.7) 8 (6.7) Somnolence 8 (6.7) 12 (10.1) Anxiety 7 (5.9) 4 (3.4) Confusion 7 (5.9) 6 (5.0) Face Edema 6 (5.0) 5 (4.2) Insomnia 6 (5.0) 11 (9.2) Coughing 5 (4.2) 10 (8.4) Dyspepsia 4 (3.4) 6 (5.0) Hypotension 4 (3.4) 6 (5.0) Pallor 4 (3.4) 6 (5.0) Dehydration 3 (2.5) 7 (5.9) Pneumonia 2 (1.7) 8 (6.7) Tachycardia 1 (0.8) 7 (5.9) Flushing 1 (0.8) 6 (5.0) 6.2 Postmarketing Experience
The following adverse reactions have been reported in the postmarketing experience of patients receiving mesna in combination with ifosfamide or similar drugs, making it difficult to distinguish the adverse reactions which may be due to mesna from those caused by the concomitantly administered cytotoxic agents. Because these reactions are reported from a population of unknown size, precise estimates of frequency cannot be made.
Cardiovascular: Hypertension
Gastrointestinal: Dysgeusia
Hepatobiliary: Hepatitis
Nervous System: Convulsion
Respiratory: Hemoptysis - 7 DRUG INTERACTIONS
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Mesna is used in combination with ifosfamide or other cytotoxic agents. Ifosfamide can cause fetal harm when administered to a pregnant woman. Refer to the ifosfamide prescribing information for more information on use during pregnancy.
Mesna injection contains the preservative benzyl alcohol. Because benzyl alcohol is rapidly metabolized by a pregnant woman, benzyl alcohol exposure in the fetus is unlikely [see Warnings and Precautions (5.3) and Use in Specific Populations (8.4)].
The estimated background risk of major birth defects and miscarriage for the indicated populations are unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Data
Animal Data
Mesna is used in combination with ifosfamide or other cytotoxic agents. Ifosfamide can cause fetal harm including embryo-fetal lethality. Refer to the ifosfamide prescribing information for more information on use during pregnancy.
In embryo-fetal development studies, oral administration of mesna to pregnant rats (500, 1000, 1500, and 2000 mg/kg) and rabbits (500 and 1000 mg/kg) during the period of organogenesis revealed no adverse developmental outcomes at doses approximately 10 times the maximum recommended total daily human equivalent dose based on body surface area.
8.2 Lactation
Risk Summary
Mesna is used in combination with ifosfamide or other cytotoxic agents. Ifosfamide is excreted in breast milk. Refer to the ifosfamide prescribing information for more information on use during lactation. There are no data on the presence of mesna in human or animal milk, the effect on the breastfed child, or the effect on milk production.
Mesna injection contains the preservative benzyl alcohol. Because benzyl alcohol is rapidly metabolized by a lactating woman, benzyl alcohol exposure in the breastfed infant is unlikely. However, adverse reactions have occurred in premature neonates and low birth weight infants who received intravenously administered benzyl alcohol-containing drugs [see Warnings and Precautions (5.3) and Use in Specific Populations (8.4)].
Because of the potential for serious adverse reactions in a breastfed child, advise lactating women not to breastfeed during treatment and for 1 week after the last dose of mesna or ifosfamide.
8.3 Females and Males of Reproductive Potential
Mesna is used in combination with ifosfamide or other cytotoxic agents. Ifosfamide can cause fetal harm when administered to a pregnant woman. Refer to the ifosfamide prescribing information for more information on contraception and effects on fertility.
Pregnancy Testing
Verify the pregnancy status of females of reproductive potential prior to initiation of mesna in combination with ifosfamide.
8.4 Pediatric Use
Mesna injection contains the preservative benzyl alcohol which has been associated with serious adverse reactions and death when administered intravenously to premature neonates and low-birth weight infants. Avoid use of mesna injection in premature neonates and low-birth weight infants [see Warnings and Precautions (5.3)].
8.5 Geriatric Use
Clinical studies of mesna did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy. The ratio of ifosfamide to mesna should remain unchanged.
-
10 OVERDOSAGE
There is no known antidote for mesna.
In a clinical trial, 11 patients received intravenous mesna 10 mg/kg to 66 mg/kg per day for 3 to 5 days. Patients also received ifosfamide or cyclophosphamide. Adverse reactions included nausea, vomiting, diarrhea and fever. An increased rate of these adverse reactions has also been found in oxazaphosphorine-treated patients receiving ≥80 mg mesna per kg per day intravenously compared with patients receiving lower doses or hydration treatment only.
Postmarketing, administration of 4.5 g to 6.9 g of mesna resulted in hypersensitivity reactions including mild hypotension, shortness of breath, asthma exacerbation, rash, and flushing.
-
11 DESCRIPTION
Mesna is a sterile, nonpyrogenic, aqueous detoxifying agent to inhibit the hemorrhagic cystitis induced by ifosfamide. The active ingredient, mesna, is a synthetic sulfhydryl compound designated as sodium-2-mercaptoethane sulfonate with a molecular formula of C2H5NaO3S2 and a molecular weight of 164.18. Its structural formula is as follows:
Mesna Injection is a sterile, nonpyrogenic, aqueous solution of colorless to light pink appearance in clear glass multidose vials for intravenous administration. Mesna Injection contains 100 mg per mL mesna, 0.25 mg per mL edetate disodium, sodium hydroxide for pH adjustment and qs with Water for Injection. Mesna Injection multidose vials also contain 10.4 mg per mL of benzyl alcohol as a preservative. The solution has a pH range of 6.5 to 7.4.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Mesna reacts chemically with the urotoxic ifosfamide metabolites, acrolein and 4-hydroxy-ifosfamide, resulting in their detoxification. The first step in the detoxification process is the binding of mesna to 4-hydroxy-ifosfamide forming a non-urotoxic 4-sulfoethylthioifosfamide. Mesna also binds to the double bonds of acrolein and to other urotoxic metabolites and inhibits their effects on the bladder.
12.3 Pharmacokinetics
Absorption
Following oral administration, peak plasma concentrations were reached within 1.5 to 4 hours and 3 to 7 hours for free mesna and total mesna (mesna plus dimesna and mixed disulfides), respectively. Oral bioavailability averaged 58% (range 45 to 71%) for free mesna and 89% (range 74 to 104%) for total mesna based on plasma AUC data from 8 healthy volunteers who received 1200 mg oral or intravenous doses.
Food does not affect the urinary availability of orally administered mesna.
Distribution
Mean apparent volume of distribution (Vd) for mesna is 0.652 ± 0.242 L/kg after intravenous administration which suggests distribution to total body water (plasma, extracellular fluid, and intracellular water).
Metabolism
Analogous to the physiological cysteine-cystine system, mesna is rapidly oxidized to its major metabolite, mesna disulfide (dimesna). Plasma concentrations of mesna exceed those of dimesna after oral or intravenous administration.
Excretion
Following intravenous administration of a single 800 mg dose, approximately 32% and 33% of the administered dose was eliminated in the urine in 24 hours as mesna and dimesna, respectively. Mean plasma elimination half-lives of mesna and dimesna are 0.36 hours and 1.17 hours, respectively. Mesna has a plasma clearance of 1.23 L/h/kg.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term studies in animals have been performed to evaluate the carcinogenic potential of mesna.
Mesna was not genotoxic in the in vitro Ames bacterial mutagenicity assay, the in vitro mammalian lymphocyte chromosomal aberration assay or the in vivo mouse micronucleus assay.
No studies on male or female fertility were conducted. No signs of male or female reproductive organ toxicity were seen in 6-month oral rat studies (≤2000 mg/kg/day) or 29-week oral dog studies (520 mg/kg/day) at doses approximately 10-fold higher than the maximum recommended human dose on a body surface area basis.
-
14 CLINICAL STUDIES
14.1 Intravenous Mesna
Hemorrhagic cystitis produced by ifosfamide is dose dependent (Table 4). At a dose of 1.2 g/m2 ifosfamide administered daily for 5 days, 16 to 26% of the patients who received conventional uroprophylaxis (high fluid intake, alkalinization of the urine, and the administration of diuretics) developed hematuria (>50 RBC per hpf or macrohematuria) (Studies 1, 2, and 3). In contrast, none of the patients who received mesna injection together with this dose of ifosfamide developed hematuria (Studies 3 and 4). In two randomized studies, (Studies 5 and 6), higher doses of ifosfamide, from 2 g/m2 to 4 g/m2 administered for 3 to 5 days, produced hematuria in 31 to 100% of the patients. When mesna was administered together with these doses of ifosfamide, the incidence of hematuria was less than 7%.
Table 4. Percent of Mesna Patients Developing Hematuria (≥50 RBC/hpf or macrohematuria) *Ifosfamide dose 1.2 g/m2 d x 5
†Ifosfamide dose 2 g/m2 to 4 g/m2 d x 3 to 5
Study Conventional Uroprophylaxis
(number of patients)Standard Mesna Intravenous
Regimen (number of patients)Uncontrolled Studies* Study 1 16% (7/44) - Study 2 26% (11/43) - Study 3 18% (7/38) 0% (0/21) Study 4 - 0% (0/32) Controlled Studies† Study 5 31% (14/46) 6% (3/46) Study 6 100% (7/7) 0% (0/8) -
16 HOW SUPPLIED/STORAGE AND HANDLING
Mesna Injection is supplied as follows:
NDC Mesna Injection (100 mg per mL) Package Factor 25021-201-10 1 gram per 10 mL Multi-Dose Vial 1 vial per carton 25021-201-11 1 gram per 10 mL Multi-Dose Vial 10 vials per carton Storage Conditions
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° and 30°C (59° and 86°F). [See USP Controlled Room Temperature.]
Sterile, Nonpyrogenic.
The container closure is not made with natural rubber latex.If Mesna Injection is co-administered with ifosfamide, refer to the ifosfamide prescribing information for safe handling instructions.
-
17 PATIENT COUNSELING INFORMATION
Hypersensitivity
- Advise the patient to discontinue mesna and seek immediate medical attention if any signs or symptoms of a hypersensitivity reaction, including systemic anaphylactic reactions occur [see Warnings and Precautions (5.1)].
Hemorrhagic Cystitis
- Mesna does not prevent hemorrhagic cystitis in all patients nor does it prevent or alleviate any of the other adverse reactions or toxicities associated with ifosfamide. Advise the patient to report to their healthcare provider if his/her urine has turned a pink or red color [see Dosage and Administration (2.3)].
- Advise the patient to drink 1 to 2 liters of fluid each day during mesna therapy [see Dosage and Administration (2.3)].
Dermatologic Toxicity
- Advise the patient that Stevens-Johnson syndrome, toxic epidermal necrolysis, and drug rash with eosinophilia and systemic symptoms and bullous and ulcerative skin and mucosal reactions have occurred with mesna. Advise the patient to report to their healthcare provider if signs and symptoms of these syndromes occur [see Warnings and Precautions (5.2)].
Embryo-Fetal Toxicity
- Mesna is used in combination with ifosfamide. Ifosfamide or other cytotoxic agents can cause fetal harm when administered to a pregnant woman. Inform female patients of the risk to a fetus and potential loss of the pregnancy. Advise females to inform their healthcare provider if they are pregnant or become pregnant [see Use in Specific Populations (8.1)].
Contraception
- Advise females of reproductive potential to use effective contraception during treatment with mesna in combination with ifosamide and for 6 months after the last dose [see Use in Specific Populations (8.3)].
- Advise male patients with female partners of reproductive potential to use effective contraception during treatment with mesna in combination with ifosamide and for 3 months after the last dose [see Use in Specific Populations (8.3)].
Lactation
- Advise lactating women not to breastfeed during treatment with mesna or ifosfamide and for 1 week after the last dose [see Use in Specific Populations (8.2)].
SAGENT®
Mfd. for SAGENT Pharmaceuticals
Schaumburg, IL 60195 (USA)
Made in India
©2021 Sagent Pharmaceuticals, Inc.Revised: January 2021
SAGENT Pharmaceuticals®
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MESNA
mesna injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:25021-201 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength mesna (UNII: NR7O1405Q9) (2-Mercaptoethanesulfonic Acid - UNII:VHD28S0H7F) mesna 100 mg in 1 mL Inactive Ingredients Ingredient Name Strength edetate disodium (UNII: 7FLD91C86K) benzyl alcohol (UNII: LKG8494WBH) sodium hydroxide (UNII: 55X04QC32I) water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:25021-201-10 1 in 1 CARTON 12/01/2010 1 10 mL in 1 VIAL; Type 0: Not a Combination Product 2 NDC:25021-201-11 10 in 1 CARTON 12/01/2010 2 10 mL in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA090913 12/01/2010 Labeler - Sagent Pharmaceuticals (796852890)