GAMMAGARD S/D- human immunoglobulin g
Baxalta US Inc.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use GAMMAGARD S/D safely and effectively. See full prescribing information for GAMMAGARD S/D
GAMMAGARD S/D, Immune Globulin Intravenous (Human) Solvent Detergent Treated IgA less than or equal to 2.2 µg/mL in a 5% Solution Initial U.S. Approval: 1994 WARNINGSee full prescribing information for complete boxed warning
INDICATIONS AND USAGEGAMMAGARD S/D is an Immune Globulin Intravenous (Human), indicated for:
DOSAGE AND ADMINISTRATIONIntravenous Use Only
DOSAGE FORMS AND STRENGTHSFreeze-dried preparation containing 2.5g, 5 g and 10g IgG. (3) CONTRAINDICATIONSWARNINGS AND PRECAUTIONS
ADVERSE REACTIONS
To report SUSPECTED ADVERSE REACTIONS, contact Baxter Healthcare Corporation at 1-866-888-2472 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch DRUG INTERACTIONSPassive transfer of antibodies may interfere with the immune response to live vaccines, such as measles, mumps, and rubella. (7) USE IN SPECIFIC POPULATIONSSee 17 for PATIENT COUNSELING INFORMATION. Revised: 7/2010 |
|||||||||||||||||||
FULL PRESCRIBING INFORMATION
WARNING: RENAL DYSFUNCTION/ACUTE RENAL FAILURE
- Intravenous use of human immune globulin (IGIV) products, particularly those containing sucrose, has been reported to be associated with renal dysfunction, acute renal failure, osmotic nephropathy, and death. Patients at risk of acute renal failure include those with any degree of pre-existing renal insufficiency, diabetes mellitus, advanced age (above 65 years of age), volume depletion, sepsis, paraproteinemia, or those receiving known nephrotoxic drugs. GAMMAGARD S/D does not contain sucrose.
- For patients at risk of renal dysfunction or failure, administer GAMMAGARD S/D at the minimum concentration available and the minimum rate of infusion practicable.
1 INDICATIONS AND USAGE
1.1 Primary Immunodeficiency (PI)
GAMMAGARD S/D is indicated for the treatment of primary immunodeficiency (PI) associated with defects in humoral immunity, in adults and children two years and older. This includes, but is not limited to, congenital agammaglobulinemia, common variable immunodeficiency, Wiskott-Aldrich syndrome, and severe combined immunodeficiencies.1,2,3
1.2 B-cell Chronic Lymphocytic Leukemia (CLL)
GAMMAGARD S/D is indicated for prevention of bacterial infections in patients with hypogammaglobulinemia and/or recurrent bacterial infections associated with B-cell Chronic Lymphocytic Leukemia (CLL).4
2 DOSAGE AND ADMINISTRATION
For Intravenous Use Only
2.1 Preparation and Handling
Instruction for Reconstitution:
Allow GAMMAGARD S/D and diluent to reach room temperature before reconstitution and administration if refrigerated.
Reconstitution:
1. Remove caps from concentrate and diluent bottles to expose central portion of rubber stoppers, and cleanse stoppers with germicidal solution.
To make a 5% solution: Use the full volume of the diluent bottle
To make a 10% solution: Remove half of the volume of the diluent bottle
Table 1 indicates the volume of diluent that should beremoved from the vial before attaching the transfer device to produce a 10% concentration. Using aseptic technique, withdraw the unnecessary volume of diluent using a sterile hypodermic syringe and needle. Discard the filled syringe into a suitable puncture proof container (Sharps Container).
| Concentration | 2.5 g bottle | 5 g bottle | 10 g bottle |
| 5% | Do not remove any diluent for reconstitution of 5% Solution | ||
| 10% | 25 ml | 48 mL | 96 mL |
2. Remove spike cap from one end of the transfer device. Do not touch spike.
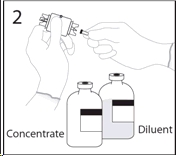
3a. Place the diluent bottle on a flat surface and hold the bottle to prevent slipping. Use exposed end of transfer device to spike diluent bottle perpendicularly through center of the stopper.
CAUTION: Failure to insert spike into center of the stopper may result in dislodging of the stopper.
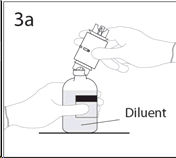
3b. Ensure that the collar collapses fully into the device by pushing down on the transfer device firmly.
While holding onto transfer device, remove remaining spike cover from the other end of the transfer device. Do not touch spike.
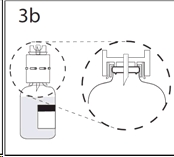
4. Hold diluent bottle with attached transfer device at an angle to the concentrate bottle to prevent spilling the diluent.
Note: Do not hold diluent bottle upside down, for this can lead to diluent spillage.
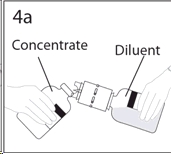
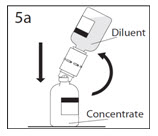
5a. Spike concentrate bottle through center of the stopper while quickly inverting the diluent bottle to minimize spilling out diluent.
CAUTION: Failure to insert the spike into the center of the stopper may result in dislodging of the stopper and loss of vacuum.
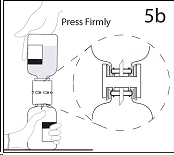
5b. Ensure that the collar collapses fully into the device by pushing down on the diluent bottle firmly.
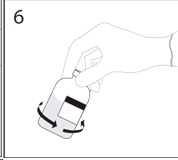
6. After transfer of diluent is complete, remove transfer device and empty diluent bottle. Immediately swirl the concentrate bottle gently to thoroughly mix contents. Repeat gentle rotation as long as undissolved product is observed.
CAUTION: Do not shake. Avoid foaming.
Discard transfer device after single use per local guidelines.
Reconstituted GAMMAGARD S/D is a clear to slightly opalescent and colorless to pale yellow solution. Visually inspect parenteral drug product for particulate matter and discoloration prior to administration. Do not use if particulate matter and/or discoloration is observed.
Follow directions for use which accompany the administration set provided. If another administration set is used, ensure that the set contains a similar filter.
Begin administration as soon as possible within 2 hours if reconstitution is performed aseptically outside of a sterile laminar air flow hood.
Administer within 24 hours if reconstitution is performed aseptically inside of a sterile laminar flow hood and stored in the original glass container or pooled into ViaFlex bags under constant refrigeration (2 °C to 8 °C). Record the date and time of reconstitution/pooling. Discard partially used vials.
2.2 Dose
Primary Immunodeficiency (PI)
The recommended dose of GAMMAGARD S/D for patients with PI is 300-600 mg/kg infused at 3 to 4 week intervals.1,2,6 Adjust dose according to the clinical response; the frequency and dose of immunoglobulin may vary from patient to patient. No randomized controlled clinical trials are available to determine an optimum target trough serum IgG level.
B-cell Chronic Lymphocytic Leukemia (CLL)
The recommended dose of GAMMAGARD S/D for patients with hypogammaglobulinemia and/or recurrent bacterial infections due to B-cell CLL is 400 mg/kg body weight infused at every 3 to 4 week intervals.4
Idiopathic Thrombocytopenic Purpura (ITP)
The recommended dose of GAMMAGARD S/D for patients with chronic ITP is 1 g/kg. The need for additional doses can be determined by clinical response and platelet count. Up to three separate doses may be given on alternate days if required.
Kawasaki Syndrome
The recommended dose of GAMMAGARD S/D for patients with Kawasaki Syndrome is either a single 1 g/kg dose or a dose of 400 mg/kg for four consecutive days beginning within seven days of the onset of fever, administered concomitantly with appropriate aspirin therapy (80-100 mg/kg/day in four divided doses) is recommended.5,7,8
2.3 Administration
Administer GAMMAGARD S/D as soon after reconstitution as possible and administer the reconstituted material at room temperature.
The recommended initial 5% solution infusion rate is 0.5 mL/kg/hour. The infusion rate may be gradually increased to a maximum rate of 4 mL/kg/hour as tolerated for patients with no history of adverse reactions to IGIV and no significant risk factors for renal dysfunction or thrombotic complications. Patients who tolerate the 5% concentration at 4 mL/kg/hour can be infused with the 10% concentration starting at 0.5 mL/kg/hour. The rate can be increased gradually up to a maximum of 8 mL/kg/hour if no adverse effects occur.9
Monitor patient vital signs throughout the infusion. Certain adverse reactions such as headaches, flushing and changes in pulse rate and blood pressure may be related to the rate of infusion. Slow or stop the infusion if adverse reactions occur. If symptoms subside promptly, the infusion may be resumed at a lower rate that does not result in reoccurrence of the symptoms.
It is recommended that, if possible, the antecubital veins are used, especially for 10% solutions, to reduce the likelihood of discomfort at the infusion site (see ADVERSE REACTIONS [6]).
Adverse reactions may occur more frequently in patients who receive human IGIV for the first time, upon switching brands, or if there has been a long hiatus since the previous infusion. In such cases, start at a lower rate and gradually increase as tolerated.
There are no prospective studies demonstrating that any concentration or rate of infusion is completely safe. However, the risk may be decreased at lower rates of infusion.10 For patients who are judged to be at risk of renal dysfunction or thrombotic complications, administer GAMMAGARD S/D at the minimum allowable rate of infusion and gradually titrated up to a more conservative maximal rate of less than 3.3 mg/kg/min (< 2mL/kg/hour of a 10% or < 4mL/kg/hour of a 5% solution) (see WARNINGS AND PRECAUTIONS [5.3]).
3 DOSAGE FORMS AND STRENGTHS
GAMMAGARD S/D is a freeze-dried preparation containing 2.5 g, 5 g or 10 g IgG.
4 CONTRAINDICATIONS
GAMMAGARD S/D is contraindicated in patients who have had a history of anaphylactic or severe systemic hypersensitivity reactions to the administration of human immunoglobulin.
GAMMAGARD S/D is contraindicated in IgA deficient patients with antibodies to IgA and a history of hypersensitivity.
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity
Severe hypersensitivity reactions and anaphylactic reactions with a fall in blood pressure have occurred in patients receiving GAMMAGARD S/D, including patients who tolerated previous treatments with GAMMAGARD S/D, even though it contains low levels of IgA. If hypersensitivity reaction develops, discontinue GAMMAGARD S/D infusion immediately and institute appropriate treatment.
GAMMAGARD S/D contains trace amounts of IgA (≤ 2.2 µg/mL in a 5% solution). Patients with IgA deficiency and antibodies to IgA have a greater risk of developing potentially severe hypersensitivity and anaphylactic reactions. GAMMAGARD S/D is contraindicated in patients with antibodies against IgA and a history of hypersensitivity reaction (seeCONTRAINDICATIONS [4]).
5.2 Renal Dysfunction Failure
Acute renal failure has been reported in association with GAMMAGARD S/D. Acute renal dysfunction/failure, acute tubular necrosis, proximal tubular nephropathy, osmotic nephrosis and death have been reported in patients receiving IGIV, particularly those products containing sucrose.10 GAMMAGARD S/D does not contain sucrose.
Assure that patients are not volume depleted prior to the initiation of the infusion of GAMMAGARD S/D. In patients who are at risk of developing renal dysfunction, because of pre-existing renal insufficiency or predisposition to acute renal failure (such as diabetes mellitus, age greater than 65, volume depletion, sepsis, paraproteinemia, or patients receiving known nephrotoxic drugs, etc.), administer GAMMAGARD S/D at an infusion rate less than 4 mL/kg/hour (< 3.3 mg IG/kg/min) for a 5% solution or at a rate less than 2 mL/kg/hour (< 3.3 mg IG/kg/min) for a 10 % solution (see DOSAGE AND ADMINISTRATION [2]).
Periodic monitoring of renal function and urine output is particularly important in patients judged to be at increased risk for developing acute renal failure. Assess renal function, including measurement of blood urea nitrogen (BUN) and serum creatinine, before the initial infusion of GAMMAGARD S/D and again at appropriate intervals thereafter. If renal function deteriorates, consider discontinuation of GAMMAGARD S/D.
5.3 Thromboembolic Events
Thromboembolic events, including myocardial infarction, cerebral vascular accident, deep vein thrombosis, and pulmonary embolism, have been reported in association with IGIV therapy, including GAMMAGARD S/D (see ADVERSE REACTIONS [6]).10,11 Patients at risk for thromboembolic events include those with a history of atherosclerosis, multiple cardiovascular risk factors, advanced age, impaired cardiac output, known or suspected hyperviscosity, hypercoagulable disorders, prolonged periods of immobilization, obesity, diabetes mellitus, acquired or inherited thrombophilic disorder, a history of vascular diseases, and a history of a previous thrombotic or thromboembolic event (see WARNINGS AND PRECAUTIONS [5.9]).
Consider baseline assessment of blood viscosity in patients at risk for hyperviscosity, including those with cryoglobulins, fasting chylomicronemia/markedly high triacylglycerols (triglycerides), or monoclonal gammopathies (seeWARNINGS AND PRECAUTIONS [5.9]). For patients judged to be at risk of developing thrombotic events, administer GAMMAGARD S/D at the minimum rate of infusion practicable (see DOSAGE AND ADMINISTRATION [2.3]).
5.4 Aseptic Meningitis Syndrome (AMS)
AMS has been reported to occur in association with IGIV therapy, including GAMMAGARD S/D. Discontinuation of IGIV treatment has resulted in remission of AMS within several days without sequelae. The syndrome of AMS usually begins within several hours to two days following IGIV treatment.
AMS is characterized by the following symptoms and signs: severe headache, nuchal rigidity, drowsiness, fever, photophobia, painful eye movements, nausea and vomiting. Cerebrospinal fluid (CSF) studies are frequently positive with pleocytosis up to several thousand cells per cubic mm, predominantly from the granulocytic series, and with elevated protein levels up to several hundred mg/dL, but negative culture results. Conduct a thorough neurological examination on patients exhibiting such symptoms and signs, including CSF studies, to rule out other causes of meningitis.
AMS may occur more frequently with high dose (2 g/kg) IGIV treatment.12
5.5 Hemolysis
Hemolytic anemia can develop subsequent to IGIV therapy, including GAMMAGARD S/D. (see ADVERSE REACTIONS (6.2)) GAMMAGARD S/D contains blood group antibodies which may act as hemolysins and induce in vivocoating of red blood cells (RBC) with immunoglobulin, causing a positive direct antiglobulin reaction and, rarely, hemolysis. Acute intravascular hemolysis has been reported, and delayed hemolytic anemia can develop subsequent to IGIV therapy due to enhanced RBC sequestration (see ADVERSE REACTIONS (6.2)).
Monitor patients for clinical signs and symptoms of hemolysis. If signs and/or symptoms of hemolysis are present after GAMMAGARD S/D infusion, perform appropriate confirmatory laboratory testing.
5.6 Transfusion-Related Acute Lung Injury (TRALI)
Non-cardiogenic pulmonary edema (TRALI) has been reported in patients following the administration of gammaglobulin products, including GAMMAGARD S/D therapy (seeADVERSE REACTIONS (6.2)). TRALI is characterized by severe respiratory distress, pulmonary edema, hypoxemia, normal left ventricular function, and fever. Symptoms typically occur within 1 to 6 hours after treatment.
Monitor patients for pulmonary adverse reactions. If TRALI is suspected, perform appropriate tests for the presence of anti-neutrophil and anti-HLA antibodies in both the product and patient serum. TRALI may be managed using oxygen therapy with adequate ventilatory support.
5.7 Transmissible Infectious Agents
Because GAMMAGARD S/D is made from human plasma, it may carry a risk of transmitting infectious agents, e.g., viruses, and theoretically, the Creutzfeldt-Jakob disease (CJD) agent. This also applies to unknown or emerging viruses and other pathogens. No cases of transmission of viral diseases or CJD have ever been identified for GAMMAGARD S/D. All infections thought by a physician possibly to have been transmitted by this product should be reported by the physician or other healthcare provider to Baxter Healthcare Corporation at 1-800-423-2862 (in the U.S.) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. The physician should discuss the risks and benefits of this product with the patient.
5.8 Hyperproteinemia, Increased Serum Viscosity, and Hypernatremia
Hyperproteinemia and increased serum viscosity may occur in patients receiving GAMMAGARD S/D.
The amount of sodium in the product may add materially to the recommended daily allowance of dietary sodium for patients on a low sodium diet. In these patients, calculate the amount of sodium from the product and use it when determining dietary sodium intake. GAMMAGARD S/D contains approximately 0.85% NaCI or approximately 3340 mg sodium/liter at a 5% concentration. A 70 kg patient receiving 1g/kg (1.4 L) of the product would receive 4676 mg of sodium.
5.9 Monitoring: Laboratory Tests
- Periodic monitoring of renal function and urine output should be considered in patients judged to be at increased risk of developing acute renal failure. Assess renal function, including measurement of BUN and serum creatinine, before the initial infusion of GAMMAGARD S/D and at appropriate intervals thereafter.10
- Consider baseline assessment of blood viscosity in patients at risk for hyperviscosity, including those with cryoglobulins, fasting chylomicronemia/markedly high triacylglycerols (triglycerides), or monoclonal gammopathies because of the potentially increased risk of thrombosis.
- If signs and/or symptoms of hemolysis are present after an infusion of GAMMAGARD S/D, perform appropriate laboratory testing for confirmation.
- If TRALI is suspected, perform appropriate tests for the presence of anti-neutrophil antibodies and anti-HLA antibodies in both the product and patient's serum.
5.10 Interference with Laboratory Tests
After infusion of IgG, the transitory rise of the various passively transferred antibodies in the patient's blood may yield false positive serological testing results, with the potential for misleading interpretation. Passive transmission of antibodies to erythrocyte antigens (e.g., A, B, and D) may cause a positive direct or indirect antiglobulin (Coombs') test.
6 ADVERSE REACTIONS
The most common adverse reactions reported in ≥ 5% of clinical trial subjects occurring during or within 48 hours of an infusion were headache, nausea, chills, asthenia (fatigue), pyrexia, upper abdominal pain, diarrhea, back pain, hyperhidrosis, and flushing.
There were no serious adverse events that were attributed to GAMMAGARD S/D in the clinical trials.
In postmarketing surveillance, serious adverse reactions reported with GAMMAGARD S/D were anaphylaxis, acute renal failure, myocardial infarction, cerebral vascular accident, transient ischemic attack, deep vein thrombosis, pulmonary embolism; aseptic meningitis, acute hemolysis, and TRALI.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Primary Immunodeficiency (PI)
In 17 patients receiving GAMMAGARD (5% solution) for 56 to 77 months, 12 (71%) were adults, and 5 (29%) were children (16 years or younger).3 Adverse reactions are those adverse events (AEs) that were deemed by the investigators as causally related to the infusion of GAMMAGARD. Twenty-one adverse reactions occurred in 6 of the 17 subjects of the total 341 infusions (6%). There was one death in a woman from a cerebral vascular hemorrhage secondary to thrombocytopenia and was considered unrelated to study product. Of the 5 subjects who received an infusion with 600 mg/kg at a rate of 0.3 g/kg/hour, two subjects experienced adverse reactions, with an adverse reaction rate of 40%.
The adverse reactions occurred in ≥ 5% of subjects during or within 48 hours of infusion are listed in Table 2.
| Adverse Reaction |
By Subjects (%) Total number of subjects: 17 |
By Infusions (%) Total number of infusions: 341 |
| Headache | 3 (17.6) | 3 (0.9) |
| Chills | 2 (11.8) | 6 (1.8) |
| Backache | 2 (11.8) | 2 (0.6) |
| Emesis | 1 (5.9) | 1 (0.3) |
| Flushing | 1 (5.9) | 1 (0.3) |
| Fatigue | 1 (5.9) | 4 (1.2) |
| Dizziness | 1 (5.9) | 1 (0.3) |
In a double blind, crossover study, 36 subjects with PI were treated for 6 months with GAMMAGARD S/D and 6 months with Gamimune N. One hundred AEs were considered to be possibly or probably related to treatment with GAMMAGARD S/D. Of these, 72 were mild, 24 were moderate, and 4 were severe. The numbers and percentages of AEs were similar for GAMMAGARD S/D and Gamimune-N. There were no deaths during the study. The adverse reactions that occurred during GAMMAGARD S/D treatment in ≥ 5% of subjects in the study are shown in Table 3.
| Adverse Reactions |
By Subject (%) Total number of subjects: 36 |
By Infusion (%) Total number of infusions: 211 |
| Headache | 11 (30.56) | 23 (10.9) |
| Nausea | 8 (22.22) | 14 (6.64) |
| Chills | 7 (19.44) | 14 (6.64) |
| Fatigue | 4 (11.11) | 11 (5.21) |
| Pyrexia | 4 (11.11) | 6 (2.84) |
| Upper Abdominal Pain | 3 (8.33) | 3 (1.42) |
| Diarrhea | 3 (8.33) | 3 (1.42) |
| Back Pain | 3 (8.33) | 4 (1.90) |
| Infusion Site Pain | 2 (5.56) | 3 (1.42) |
| Hyperhidrosis | 2 (5.56) | 4 (1.90) |
| Flushing | 2 (5.56) | 2 (0.95) |
In 10 subjects who participated in a PK crossover study of GAMMAGARD and GAMMAGARD S/D 5 adverse reactions were reported to be associated with the total 28 infusions (17.5%). Three of the adverse reactions were associated with 10 GAMMAGARD infusions and 2 were associated with 18 GAMMAGARD S/D infusions. Two subjects withdrew from the study. One subject developed a recurrence of chronic cellulitis and was hospitalized and the event was not considered to be related to study drug. The other subject withdrew due to the experience of moderate severe adverse reactions such as chills, anxiety and increased temperature after infusion of GAMMAGARD.
Adverse reactions occurred in the PK study and in the safety study are shown in Table 4.
| Event |
By Infusion (%) Total number: 394 |
| Headache | 20 (5.1) |
| Chills | 11 (2.7) |
| Elevated Temperature | 7 (1.8) |
| Nausea | 6 (1.5) |
| Emesis | 5 (1.3) |
| Hypertension | 4 (1.0) |
| Fatigue | 4 (1.0) |
| Flushing | 4 (1.0) |
| Leg Cramps | 3 (0.8) |
| Flu-Like Symptoms | 2 (0.5) |
| Exanthema | 2 (0.5) |
| Loss of Appetite | 2 (0.5) |
| Anxiety | 1 (0.25) |
| Backache | 1 (0.25) |
| Urticaria | 1 (0.25) |
The tolerability and viral safety of GAMMAGARD S/D were evaluated in a study of 38 subjects, who were treated with GAMMAGARD S/D for an average of 7.7 months. Adverse reactions were reported from 20 of the 38 subjects (52.6%) in 50 of the total 394 infusions (12.7%) during or within 48 hours of an infusion. Twenty-four (48%) of the adverse reactions occurred in 3 subjects and 26 occurred in the other 35 subjects in 350 infusions. No subject withdrew during the study. Five subjects had a transient borderline elevation in liver enzyme (AST). No subject developed a positive serologic response to Hepatitis C or HIV. There were no other significant laboratory abnormalities.
The adverse experiences of GAMMAGARD S/D reconstituted as a 10% solution and the maximal tolerated infusion rate were examined in a post-marketing study of 27 subjects. Local pain and/or irritation occurred in 42 of the total 276 infusions (15.2%). Ninety percent of the reactions occurred when the patients received the 10% solution compared to the 5% control. These local reactions tended to be more common following hand vein infusions and their incidence may be reduced by infusions via the antecubital vein. Application of a warm compress to the infusion site alleviated local symptoms. Twenty-six subjects were able to achieve the maximal infusion rate of 8 mL/kg/hour with the GAMMAGARD S/D reconstituted to a 10% solution.
B-cell Chronic Lymphocytic Leukemia (CLL)
In the study of 81 patients with B-cell CLL, the incidence of adverse reactions following GAMMAGARD infusions was approximately 1.3% compare to the rate of the placebo (normal saline) group which was 0.6%.3 There were 23 adverse reactions associated with the 1235 infusions in the study. Sixteen of the adverse reactions occurred in the GAMMAGARD group (1.6%) and 7 in the control group (0.6%). The most common reactions were fever and chills. Sleepiness was noted during 4 infusions. One subject had a myocardial infarction which was considered to be unrelated to the GAMMAGARD. Twenty-four of the subjects did not complete all 17 infusions. Three subjects in each group died during the study, five of whom were due to infection. The other 18 subjects withdrew for reasons unrelated to the treatment.
Idiopathic Thrombocytopenic Purpura (ITP)
During the clinical study of GAMMAGARD for the treatment of ITP, headache occurred in 12 of 16 subjects (75%) and was the only adverse reaction reported. Of these 12 subjects, 11 had chronic ITP (9 adults, 2 children), and one child had acute ITP. Oral antihistamines and analgesics alleviated the symptoms and were used as pretreatment for those subjects requiring additional IGIV therapy.
Kawasaki Syndrome
In a study of 51 subjects with Kawasaki syndrome, no hypersensitivity-type reactions (urticaria, bronchospasm or generalized anaphylaxis) were reported in subjects receiving either a single 1g/kg dose or 400 mg/kg of GAMMAGARD for four consecutive days. Adverse reactions, including chills, flushing, cramping, headache, hypotension, nausea, rash and wheezing, were reported with both dose regimens. These adverse reactions occurred in 7 of the 51 (13.7%) subjects associated with 7 of the 129 (5.4%) infusions. Of the 25 subjects who received a single 1 g/kg dose, 4 subjects (16%) experienced adverse reactions. Of the 26 subjects who received 400 mg/kg/day over 4 days, 3 (11.5%) experienced adverse reactions.
6.2 Postmarketing Experience
Because adverse reactions are reported voluntarily post-approval from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to product exposure.
The following adverse reactions have been reported during postmarketing use of GAMMAGARD S/D (Table 5).
|
|
| Infections and Infestations | Aseptic Meningitis Syndrome |
| Blood and Lymphatic System Disorders | Anemia, Hemolysis, Lymphadenopathy, Thrombocytopenia |
| Immune System Disorders |
Anaphylactic Shock, Anaphylactic/Anaphylactoid Reaction, Hypersensitivity |
| Psychiatric Disorders | Restlessness |
| Nervous System Disorders | Cerebrovascular Accident, Transient Ischemic Attack, Convulsion, Dizziness, Migraine, Paresthesia, Syncope, Tremor |
| Eye Disorders | Retinal Vein Thrombosis, Eye Pain, Photophobia, Visual Disturbance |
| Cardiac Disorders | Myocardial Infarction, Cyanosis, Tachycardia, Bradycardia |
| Vascular Disorders | Vena Cava Thrombosis, Arterial Thrombosis, Deep Vein Thrombosis, Hypotension, Hypertension, Pallor, Thrombophlebitis |
| Respiratory, Thoracic And Mediastinal Disorders | Pulmonary Embolism, Pulmonary Edema, Bronchospasm, Wheezing, Cough, Hyperventilation, Hypoxia, Throat Tightness |
| Gastrointestinal Disorders | Abdominal Pain, Dyspepsia |
| Hepatobiliary Disorders | Hepatitis* |
| Skin and Subcutaneous Tissue Disorders | Angioedema, Dermatitis, Erythema, Rash |
| Musculoskeletal And Connective Tissue Disorders | Arthralgia, Myalgia |
| Renal and Urinary Disorders | Renal Failure |
| General Disorders and Administration-Site Conditions | Infusion Site Reaction, Asthenia, Edema, Rigors |
| Investigations | Positive Direct Coombs Test |
In addition to the events listed above which were observed for GAMMAGARD S/D, the following events have been identified for IGIV products in general:
| Renal | Osmotic nephropathy |
| Respiratory | Cyanosis, Apnea, Acute Respiratory Distress Syndrome (ARDS) |
| Integumentary | Bullous dermatitis, Epidermolysis, Erythema multiforme, Stevens-Johnson Syndrome |
| Cardiovascular | Cardiac arrest, Vascular collapse |
| Neurological | Coma, Loss of consciousness |
|
Hematologic Gastrointestinal |
Pancytopenia Hepatic dysfunction |
7 DRUG INTERACTIONS
Admixtures of GAMMAGARD S/D with other drugs and intravenous solutions have not been evaluated. It is recommended that GAMMAGARD S/D be administered separately from other drugs or medications which the patient may be receiving. Do not mix the product with human IGIV products from other manufacturers.
Passive transfer of antibodies may transiently impair the immune responses to live attenuated vaccines, such as measles, mumps, rubella, and varicella. Inform the immunizing physician of recent therapy with GAMMAGARD S/D so that appropriate precautions can be taken.
.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category C. Animal reproduction studies have not been conducted with GAMMAGARD S/D. It is also not known whether GAMMAGARD S/D can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Immunoglobulins cross the placenta from maternal circulation increasingly after 30 weeks of gestation. GAMMAGARD S/D should be given to a pregnant woman only if clearly needed.
8.3 Nursing Mothers
GAMMAGARD S/D has not been evaluated in nursing mothers. GAMMAGARD S/D should be given to nursing women only if clearly indicated.
8.4 Pediatric Use
Clinical studies of GAMMAGARD S/D for the treatment of PI did not include sufficient numbers of subjects aged 16 and younger to determine whether they respond differently from adults. Five children under the age of 16 were treated in the initial trial of GAMMAGARD. The mean age of subjects in the phase 4 study was 17.8 years (range 1.7 to 55.3). Efficacy and safety of GAMMAGARD S/D in pediatric patients with chronic ITP has not been established.
Efficacy and safety of GAMMAGARD S/D in pediatric patients with Kawasaki disease has been established. Virtually all patients treated for Kawasaki's disease were less than 5 years of age, with approximately 20% under the age of 1 year.
8.5 Geriatric Use
Limited information is available for the geriatric use of GAMMAGARD S/D. Clinical studies of GAMMAGARD S/D for the treatment of PI did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Use caution when administering GAMMAGARD S/D to patients age 65 and over who are judged to be at increased risk for developing thromboembolic events or renal insufficiency. Do not exceed recommended dose, and administer GAMMAGARD S/D at the minimum infusion rate practicable (seeBOXED WARNING, WARNINGS AND PRECAUTIONS [5.2, 5.4] andDOSAGE AND ADMINISTRATION[2.2])
10 OVERDOSAGE
Overdose may lead to fluid overload and hyperviscosity. Patients at particular risk of complications of fluid overload and hyperviscosity include elderly patients and patients with cardiac or renal impairment.
11 DESCRIPTION
GAMMAGARD S/D, Immune Globulin Intravenous (Human) [IGIV] is a solvent/ detergent treated, sterile, freeze-dried preparation of purified immunoglobulin G (IgG) derived from large pools of human plasma. IgG preparations are purified from plasma pools using a modified Cohn-Oncley cold ethanol fractionation process, as well as cation and anion exchange chromatography. The distribution of IgG subclasses present in this product is similar to that in normal plasma. The Fc portion is maintained intact. When reconstituted with the total volume of diluent (Sterile Water for Injection, USP) supplied, reconstituted to 5% solution, the product contains approximately 50 mg/mL of protein, of which at least 90% is gamma globulin. GAMMAGARD S/D contains trace amounts of IgA (≤ 2.2µg/mL in a 5% solution) and IgM is present in trace amounts. GAMMAGARD S/D contains all of the IgG antibody activities which are present in the donor population.The product, after reconstituted to 5% solution, contains a physiological concentration of sodium chloride (approximately 8.5 mg/mL) and has a pH of 6.8 ± 0.4. Stabilizing agents and additional components are present in the following maximum amounts for a 5% solution: 3 mg/mL Albumin (Human), 22.5 mg/mL glycine, 20 mg/mL glucose, 2 mg/mL polyethylene glycol (PEG), 1 µg/mL tri-n-butyl phosphate, 1 µg/mL octoxynol 9, and 100 µg/mL polysorbate 80. GAMMAGARD S/D contains no preservative.
To prepare a 10% (100 mg/mL) solution for infusion, add half the volume of diluent, as described in DOSAGE AND ADMINISTRATION.The content of the stabilizing agents and other components, including IgA, for the 10% solution will be doubled compared to the 5% solution.
Screening against potentially infectious agents in the product begins with the donor selection process and continues throughout plasma collection and plasma preparation. Each individual plasma donation used in the manufacture of GAMMAGARD S/D is collected only at FDA approved blood establishments and is tested by FDA licensed serological tests for Hepatitis B Surface Antigen (HBsAg), and for antibodies to Human Immunodeficiency Virus (HIV-1/HIV-2) and Hepatitis C Virus (HCV) in accordance with U.S. regulatory requirements. As an additional safety measure, mini-pools of the plasma are tested for the presence of HIV-1 and HCV by FDA licensed Nucleic Acid Testing (NAT) and found negative.
The manufacturing process includes treatment with an organic solvent/detergent mixture, composed of tri-n-butyl phosphate, octoxynol 9 and polysorbate 80. The GAMMAGARD S/D manufacturing process provides a significant viral reduction inin vitro viral reduction studies. These studies, summarized in Table 6 demonstrate virus clearance during GAMMAGARD S/D manufacturing using infectious human immunodeficiency virus, Type 1 (HIV-1), as the relevant virus for HIV-1 and HIV-2, bovine viral diarrhea virus (BVD); a model virus for enveloped RNA viruses such as hepatitis C virus (HCV), pseudorabies virus (PRV); a generic model virus for enveloped DNA viruses such as hepatitis B virus (HBV); hepatitis A virus (HAV), and mice minute virus (MMV), a model for small non-enveloped DNA viruses such as human parvovirus B19 (B19V)3. These reductions are achieved through a combination of process chemistry, partitioning and/or inactivation during cold ethanol fractionation and the solvent/detergent treatment.
| Process Step Evaluated | Virus Clearance (log10) | ||||
| Enveloped Viruses | Non- Enveloped Viruses | ||||
| BVD | HIV-1 | PRV | HAV | MMV | |
| Step 1: Processing of Cryo-Poor Plasma to Fraction I+II+III Precipitate | 0.6* | 5.6 | 1.0* | 0.5* | NT† |
| Step 2 - 3 | 2.6 | > 5.7 | > 5.2 | > 5.2 | > 5.3 |
| Step 4: Solvent/Detergent Treatment | >4.9 | >3.7 | >4.1 | NA‡ | NA |
| Cumulative Reduction of Virus (log10) | >7.5 | >15.0 | >9.3 | > 5.2 | > 5.3 |
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
GAMMAGARD S/D, Immune Globulin Intravenous (Human), supplies a broad spectrum of opsonizing and neutralizing IgG antibodies against a wide variety of bacterial and viral agents. GAMMAGARD S/D also contains a spectrum of antibodies capable of reacting with cells such as erythrocytes. The role of these antibodies and the mechanisms of action of IgG in GAMMAGARD S/D have not been fully elucidated.
12.3 Pharmacokinetics
Following infusion, IGIV products show a biphasic decay curve. The initial (α) phase is characterized by an immediate post-infusion peak in serum IgG and is followed by rapid decay due to equilibration between the plasma and extravascular fluid compartments. The second (β) phase is characterized by a slower and constant rate of decay. As a class, IgG survives longerin vivo than other serum proteins. Peak levels of IgG reached within 30 minutes after an intravenous infusion of GAMMAGARD S/D. In previous studies, where radio-labeled IgG was injected to subjects, the IgG half-life was 21 to 25 days in healthy individuals or 17.7 to 37.6 days in immunodeficient patients. The half-life of IgG can vary considerably from person to person, however. In particular, high serum concentrations of IgG and hypermetabolism associated with fever and infection have been seen to coincide with a shortened half-life of IgG.
The pharmacokinetics of GAMMAGARD S/D was evaluated in 15 subjects with PI, 10 of them were previously treated. In the previously treated subjects, the half-life of GAMMAGARD S/D is approximately 37.7 ± 15 days compared to 34.1 ± 15.7 days for GAMMAGARD. The half lives of the IgG subclasses were similar, ranging from 28.1 ± 11.2 days for IgG4 to 42.3 ± 26.6 days for IgG1. The half life of pneumococcal antibody in these subjects was 41.4 ± 28.5 days. Pharmacokinetics did not differ between the previously licensed IGIV and GAMMAGARD S/D formulations administered to the previously treated patients. The pharmacokinetics of the GAMMAGARD S/D formulation in previously untreated patients was not significantly different from the results obtained in previously treated patients. The mean trough IgG concentration in the previously untreated patients was 1186 ± 614 mg/dL and the peak post infusion concentration was 1859 ± 872 mg/dL. The mean dose was 460 ± 194 mg/kg.
14 CLINICAL STUDIES
14.1 Primary Immunodeficiency (PI)
Intravenous use of GAMMAGARD was initially evaluated in a study of 17 subjects with PI. Twelve (71%) were adults and 5 (29%) were children 16 years or younger. Six subjects received a series of 5 infusions at 4-week intervals, with the starting infusion does of 100 mg/kg and then increased to 200, 300, and 400 mg/kg at rates of 0.1 to 0.2 mg/kg/hour. Five of the 6 subjects completed the 5 infusions and received another 6 monthly infusions with the following doses each administered twice: 200-400 mg/kg at 0.1 to 0.2 g/kg/hour, 400 mg/kg at 0.1 to 0.4 g/kg/hour and 400-800 mg/kg at 0.1 to 0.4 g/ kg/hour. Then all of the 17 subjects received GAMMAGARD at 400 mg/kg every 4 weeks at a rate of 0.1 to 0.4 g/kg/hour. Fifteen of the subjects were treated for 56 to 77 weeks in this study. There were no instances of pneumonia or other infections that would qualify as an acute bacterial infection. The overall rate of non-serious bacterial infections was 4.4 per subject per year.
In a study of 15 subjects with PI to compare the pharmacokinetics of GAMMAGARD S/D with GAMMAGARD, the subjects received a total of 28 infusions, half with GAMMAGARD S/D and half with GAMMAGARD. Five systemic AEs were reported during the study and 2 occurred with GAMMAGARD S/D treatment. The study then enrolled an additional 38 patients with the diagnosis of PI (8), ITP (13), CVID (5), CLL (2) and other miscellaneous diseases (3) to evaluate acute tolerability and the viral safety of GAMMAGARD S/D.
The mean age of the subjects was 12 years old (range 0.7 to 57.2 years); 17 were males and 21 were females. The subjects received an average of 10 (range 1-22) infusions over an average of 7.7 months (range 0.3-11 months). A total of 394 infusions were administered and all were completed. The average dose was 460 mg/kg (range: 188-1110 mg/kg). Incidence of infections was not recorded, though one subject had a recurrence of chronic cellulitis. Adverse events and viral safety data were analyzed (seeADVERSE REACTIONS [6]).
GAMMAGARD S/D was compared to Gamimune N in a double-blind, crossover study of 36 PI subjects. The mean age of subjects was 17.8 years (range 1.7 to 55.3 years); 22 subjects were male and 14 were female. Eighteen were naïve to IGIV therapy. Each subject received 6 infusions of both products. There were a total of 211 GAMMAGARD S/D infusions and 210 Gamimune N infusions. The dose of GAMMAGARD S/D administered was 300-600 mg/kg every 14 to 28 days for previously untreated subjects and the same as their pre-study dose and frequency for previously treated subjects. The infusions were started at 1.0 mL/kg/hour and increased every 30 minutes to a maximum of 4.8 mL/kg/hour as tolerated. The mean dose administered for both products was 440 mg/kg. The mean infusion rate was 2.35 ± 0.54 mL/kg/hour for GAMMAGARD S/D and 2.33 ± 0.71 for Gamimune N. Two subjects withdrew from the study. One subject was pregnant, and the other subject was withdrawn by his parents after the eighth infusion for reasons other than adverse events.
The use of GAMMAGARD S/D as a 10% solution and the maximal rate of infusion were evaluated in a postmarketing study of 27 subjects with PI. Subjects were treated with GAMMAGARD S/D at 400 mg/kg every 4 weeks for up to 12 months. Each subject received an initial infusion of GAMMAGARD S/D 5% solution at 4 mL/kg/hour. Subsequently, the concentration was increased to 7.5% and then to 10% as tolerated. Thereafter, the infusion rate was gradually increased to a maximal 8 mL/kg/hour as tolerated. There were 276 infusions administered and 26 of the 27 subjects were able to reach the maximum infusion rate and concentration.
14.2 B-cell Chronic Lymphocytic Leukemia (CLL)
The efficacy of GAMMAGARD in reducing bacterial infections of B-cell CLL patients has been demonstrated in a double-blind, placebo-controlled trial of 81 subjects.4 Subjects were treated with 400 mg/kg/dose of GAMMAGARD or saline solution every 3 weeks for a total of 17 infusions. Forty-one subjects received GAMMAGARD and 40 subjects received saline. The infection outcomes, including the frequency of bacterial/viral/fungal infections, mean time to first bacterial infections, were compared between the two groups and are shown in Table 7.
| Outcome | GAMMAGARD S/D | Placebo |
Significance P value |
| Number of subjects | 41 | 40 | - |
| Frequency of bacterial infections | 56.1% | 105% | 0.01 |
| Mean time to first bacterial infection | > 365 | 192 | 0.026 |
| Total Bacterial Infections | 23 | 42 | 0.01 |
| Total Viral Infections | 40 | 37 | 0.65 |
| Fungal or Candida infection | 3 | 2 | - |
| Patients free of any infection | 13 | 11 | 0.68 |
Patients receiving GAMMAGARD had fewer infections with Streptococcus pneumoniae and Haemophilus influenza, but the incidence of other gram negative infections was similar.
14.3 Idiopathic Thrombocytopenic Purpura (ITP)
The efficacy of GAMMAGARD has been demonstrated in a clinical study involving 16 patients: thirteen had chronic ITP (11 adults, 2 children), and 3 had acute ITP (one adult, 2 children). All 16 patients (100%) demonstrated a rise in platelet count to a level greater than 40,000/mm3 following the administration of GAMMAGARD. Ten of the 16 patients (62.5%) exhibited a platelets rise to greater than 80,000/mm3. Of these 10 patients, 7 had chronic ITP (5 adults, 2 children), and 3 had acute ITP (one adult, 2 children).
The rise in platelet count to greater than 40,000/mm3 occurred after a single 1 g/kg infusion of GAMMAGARD in 8 patients with chronic ITP (6 adults, 2 children), and in 2 patients with acute ITP (one adult, one child). A similar response was observed after two 1 g/kg infusions in 3 adult patients with chronic ITP, and one child with acute ITP. The remaining 2 adult patients with chronic ITP received more than two 1 g/kg infusions before achieving a platelet count greater than 40,000/mm3. The rise in platelet count occurred within 5 days. However, this rise was transient and not considered curative. Platelet count rises lasted 2 to 3 weeks, with a range of 12 days to 6 months. It should be noted that childhood ITP may resolve spontaneously without treatment.
14.4 Kawasaki Syndrome
The efficacy of GAMMAGARD S/D in reducing the incidence of coronary artery aneurysm in patients with Kawasaki syndrome has been demonstrated in a clinical study of 44 patients.7
The incidence of coronary artery aneurysm in patients with Kawasaki syndrome receiving GAMMAGARD either at a single dose of 1 g/kg (n=22) or at a dose of 400 mg/kg for four consecutive days (n=22), beginning within seven days of onset of fever, was 3/44 (6.8%). This was significantly different (p=0.008) from a comparable group of patients that received aspirin only in previous trials and of whom 42/185 (22.7%) experienced coronary artery aneurysms. All patients in the GAMMAGARD trial received concomitant aspirin therapy and none experienced hypersensitivity-type reactions (urticaria, bronchospasm or generalized anaphylaxis).
15 REFERENCES
- Orange JS, Hossny EM, Weiler CR, Ballow M, Berger M, Bonilla FA, Buckley R, Chinen J, El-Gamal Y, Mazer BD, Nelson Jr. RP, Patel DD, Secord E, Sorenson RU, Wasserman RL, Cunningham-Rundles C. Use of Intravenous Immunoglobulin in Human Disease: A Review of Evidence by Members of the Primary Immunodeficiency Committee of the American Academy of Allergy, Asthma, and Immunology. J Allergy Clin Immunol 2006; 117:S525-53.
- Bonilla FA, Bernstein IL, Khan DA, Ballas ZK, Chinen J, Frank MM, et al. Practice parameter for the diagnosis and management of primary immunodeficiency. Ann Allergy Asthma Immunol. 2005; 94(suppl 1):S1-63.
- Ochs HD, Lee ML, Fischer SH, et al. Efficacy of a New Intravenous Immunoglobulin Preparation in Primary Immunodeficient Patients. Clinical Therapeutics. 1987; 9:512-522.
- Cooperative Group for the Study of Immunoglobulin in Chronic Lymphocytic Leukemia: Intravenous immunoglobulin for the prevention of infection in Chronic Lymphocytic Leukemia: A randomized, controlled clinical trial. N Eng J Med. 1988; 319:902-907.
- Newburger J, Takahashi M, Burns JG, et al. The Treatment of Kawasaki Syndrome with Intravenous Gamma Globulin. New England Journal of Medicine. 1986; 315:341-347.
- Eijkhout HW, Der Meer JW, Kallenbert CG, et al. The effect of two different dosages of intravenous immunoglobulin on the incidence of recurrent infections in patients with primary hypogammaglobulinemia. A randomized, double-blind, multicenter crossover trial. Ann Intern Med. 2001; 135:165-174.
- Barron KS, Murphy DJ, Siverman ED, Ruttenberg HD, Wright GB, Franklin W, Goldberg SJ, Higashino SM, Cox DG, Lee M. Treatment of Kawasaki syndrome: a comparison of two dosage regimens of intravenously administered immune globulin. J Pediatr. 1990; 117:638-644.
- Engle MA, Fatica NS, Bussel JB, O'Laughlin JE, Snyder MS, Lesser ML. Clinical Trial of Single-Dose Intravenous Gammaglobulin in Acute Kawasaki Disease. AJDC. 1989; 143:1300-1304.
- Polmar SH, Smith TF, Pirofsky B, Cox DG, Rechtman D. Rapid infusion of intravenous immunoglobulin in patients with primary immunodeficiency disease. J Allergy Clin Immunol. 1992; 69:166.
- Pierce LR, Jain N. Risks associated with the use of intravenous immunoglobulin. Transfusion Med Rev. 2003; 17:241-251.
- Katz U, Sheonfeld Y. Review: intravenous immunoglobulin therapy and thromboembolic complications. Lupus 2005; 14:802-8.
- Hamrock DJ. Adverse events associated with intravenous immunoglobulin therapy. International Immunopharmacology 2006; 6:535-42.
- Daw Z, Padmore R, Neurath D, Cober N, Tokessy M, Desjardins D, et al. Hemolytic transfusion reactions after administration of intravenous immune (gamma) globulin: a case series analysis. Transfusion 2008; 48:1598-601.
16 HOW SUPPLIED/STORAGE AND HANDLING
GAMMAGARD S/D is supplied in single use bottles containing the labeled amount of functionally active IgG. The following presentation of GAMMAGARD S/D is available:
| GramsProtein | NDC |
| 2.5g | 0944-2620-02 |
| 5 g | 0944-2620-03 |
| 10 g | 0944-2620-04 |
Each bottle of GAMMAGARD S/D is furnished with a suitable volume of Sterile Water for Injection, USP, a transfer device and an administration set which contains an integral airway and a 15 micron filter.
GAMMAGARD S/D is to be stored at a temperature not to exceed 25ºC (77ºF) for 24 months.
Do not Freeze.
17 PATIENT COUNSELING INFORMATION
Inform patients to immediately report the following signs and symptoms to their healthcare provider:
- Decreased urine output, sudden weight gain, fluid retention/edema, and/or shortness of breath (seeWARNINGS AND PRECAUTIONS [5.2])
- Acute chest pain, shortness of breath, leg pain, and swelling of the legs/feet, numbness in the face or extremities, weakness or paralysis, severe headache, confusion, and visual disturbances (seeWARNINGS AND PRECAUTIONS [5.3]).
- Severe headache, neck stiffness, drowsiness, fever, sensitivity to light, painful eye movements, nausea, and vomiting (seeWARNINGS AND PRECAUTIONS [5.4])
- Increased heart rate, fatigue, yellowing of the skin or eyes, and dark-colored urine (seeWARNINGS AND PRECAUTIONS [5.5])
- Trouble breathing, chest pain, blue lips or extremities, fever(seeWARNINGS AND PRECAUTIONS [5.6])
Inform patients that GAMMAGARD S/D is made from human plasma and may contain infectious agents that can cause disease (e.g., viruses and, theoretically, the vCJD agent). The risk of GAMMAGARD S/D transmitting an infectious agent has been reduced by screening plasma donors for prior exposure, testing donated plasma, and inactivating or removing certain viruses during manufacturing; patients should report any symptoms that concern them or that might be caused by infections ( see WARNINGS AND PRECAUTIONS [5.7]).
Inform patients that GAMMAGARD S/D can interfere with their immune response to live viral vaccines such as measles, mumps and rubella. Inform patients to notify their healthcare professional of this potential interaction when they are receiving vaccinations (seeDRUG INTERACTIONS [7]).
To enroll in the confidential, industry-wide Patient Notification System, call 1-888-UPDATE U (1-888-873-2838)
Baxter, Gammagard and Viaflex are trademarks of Baxter International Inc., registered in the U.S. Patent and Trademark Office.
Baxter Healthcare Corporation
Westlake Village, CA 91362 USA
U.S. License No. 140
Revised: December 2011
PRINCIPLE DISPLAY PANEL

GAMMAGARD SD 2.5g IgA less than 2.2 µg/mL in a 5% solution unit carton
50 mL size dried
NDC 0944-2620-02
Immune Globulin Intravenous (Human)
GAMMAGARD S/D
2.5g
IgA less than or equal to 2.2 µg/mL in a 5% solution
Single dose container. For Intravenous administration only.
Dosage and Administration: See enclosed package insert.
Reconstitution Expiry Information: See Dosage and Administration in enclosed package insert.
Storage temperature: Not to exceed (25°C (77°F). Do not freeze.
This product is prepared from large pools of human plasma. Human blood and its components may transmit infectious agents. The patient and physician should discuss the risks and benefits of this product.
Rx Only
Baxter Healthcare Corporation
Westlake Village, CA 91362 USA
U.S. License No. 140

GAMMAGARD SD 2.5g IgA less than 2.2 µg/mL in a 5% solution vial label
50 mL size, dried
NDC 0944-2620-06
single dose container
Immune Globulin Intravenous (Human)
GAMMAGARD S/D
2.5g
IgA less than or equal to 2.2 µg/mL in a 5% solution
Dosage and Administration: See enclosed package insert.
Storage temperature: not to exceed (25°C (77°F). Do not freeze.
Rx Only
Baxter and Gammagard are trademarks of Baxter International, Inc.
Baxter Healthcare Corporation, Westlake Village, CA 91362 USA
U.S. License No.140

50 mL Sterile Water for Injection vial label
1A7136
50 mL
Nonpyrogenic Single-Dose Container NDC 0338-0001-65
Baxter
Sterile Water for Injection, USP for reconstitution of accompanying product
See accompanying directions for use. Do not use unless clear. No antimicrobial agent or other substance has been added. Do not use for intravascular injection without making approximately isotonic by addition of suitable solute. Discard unused portion.
This Product Contains Dry Natural Rubber. Rx Only.
Manufactured by: Baxter Healthcare Corporation Deerfield, IL 60015 USA
Imported by Baxter Corporation Toronto, Ontario
Baxter is a trademark of Baxter International Inc.
| GAMMAGARD
S/D
human immunoglobulin g kit |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Baxalta US Inc. (079887619) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Baxter Healthcare Corporation | 001728059 | MANUFACTURE | |