Label: ISTALOL- timolol maleate solution/ drops
- NDC Code(s): 24208-004-01, 24208-004-02, 24208-004-03
- Packager: Bausch & Lomb Incorporated
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated March 31, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use ISTALOL safely and effectively. See full prescribing information for ISTALOL.
ISTALOL® (timolol maleate ophthalmic solution), for topical ophthalmic use
Initial U.S. Approval: 1978INDICATIONS AND USAGE
ISTALOL is a non-selective beta-adrenergic receptor blocking agent indicated for the treatment of elevated intraocular pressure in patients with ocular hypertension or open-angle glaucoma. (1)
DOSAGE AND ADMINISTRATION
One drop in the affected eye(s) once a day in the AM. (2)
DOSAGE FORMS AND STRENGTHS
Topical ophthalmic solution containing timolol maleate, 0.5% (5 mg/mL) (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- •
- Potentiation of Respiratory Reactions Including Asthma (5.1)
- •
- Cardiac Failure (5.2)
- •
- Obstructive Pulmonary Disease (5.3)
- •
- Increased Reactivity to Allergens (5.4)
- •
- Potentiation of Muscle Weakness (5.5)
- •
- Masking of Hypoglycemic Symptoms in Patients with Diabetes Mellitus (5.6)
- •
- Masking of Thyrotoxicosis (5.7)
ADVERSE REACTIONS
The most frequently reported adverse reactions have been burning and stinging upon instillation in 38% of patients treated with ISTALOL. Additional reactions reported with ISTALOL at a frequency of 4 to 10% include: blurred vision, cataract, conjunctival injection, headache, hypertension, infection, itching and decreased visual acuity.(6)
To report SUSPECTED ADVERSE REACTIONS, contact Bausch & Lomb Incorporated at 1-800-553-5340 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- •
- Concomitant use with systemic beta-blockers may potentiate systemic beta-blockade. (7.1)
- •
- Oral or intravenous calcium antagonists may cause atrioventricular conduction disturbances, left ventricular failure, and hypotension. (7.2)
- •
- Catecholamine-depleting drugs may have additive effects and produce hypotension and/or marked bradycardia. (7.3)
- •
- Digitalis and calcium antagonists may have additive effects in prolonging atrioventricular conduction time. (7.4)
- •
- Potentiated systemic beta-blockade (e.g., decreased heart rate) has been reported during combined treatment with quinidine and timolol. (7.5)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 3/2022
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
4.1 Asthma, COPD
4.2 Sinus Bradycardia, AV Block, Cardiac Failure, Cardiogenic Shock
4.3 Hypersensitivity Reactions
5 WARNINGS AND PRECAUTIONS
5.1 Potentiation of Respiratory Reactions Including Asthma
5.2 Cardiac Failure
5.3 Obstructive Pulmonary Disease
5.4 Increased Reactivity to Allergens
5.5 Potentiation of Muscle Weakness
5.6 Masking of Hypoglycemic Symptoms in Patients with Diabetes Mellitus
5.7 Masking of Thyrotoxicosis
5.8 Contamination of Topical Ophthalmic Products After Use
5.9 Impairment of Beta-adrenergically Mediated Reflexes During Surgery
5.10 Angle-Closure Glaucoma
5.11 Cerebrovascular Insufficiency
5.12 Choroidal Detachment
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Beta-Adrenergic Blocking Agents
7.2 Calcium Antagonists
7.3 Catecholamine-Depleting Drugs
7.4 Digitalis and Calcium Antagonists
7.5 CYP2D6 Inhibitors
7.6 Clonidine
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
- 2 DOSAGE AND ADMINISTRATION
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
4.1 Asthma, COPD
ISTALOL is contraindicated in patients with bronchial asthma; a history of bronchial asthma; severe chronic obstructive pulmonary disease [see Warnings and Precautions (5.1, 5.3)].
4.2 Sinus Bradycardia, AV Block, Cardiac Failure, Cardiogenic Shock
ISTALOL is contraindicated in patients with sinus bradycardia; second or third degree atrioventricular block; overt cardiac failure; cardiogenic shock [see Warnings and Precautions (5.2)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Potentiation of Respiratory Reactions Including Asthma
ISTALOL contains timolol maleate; and although administered topically, it can be absorbed systemically. Therefore, the same adverse reactions found with systemic administration of beta-adrenergic blocking agents may occur with topical administration. For example, severe respiratory reactions and cardiac reactions including death due to bronchospasm in patients with asthma, and rarely death in association with cardiac failure, have been reported following systemic or ophthalmic administration of timolol maleate [see Contraindications (4.1)].
5.2 Cardiac Failure
Sympathetic stimulation may be essential for support of the circulation in individuals with diminished myocardial contractility, and its inhibition of beta-adrenergic receptor blockade may precipitate more severe failure.
In patients without a history of cardiac failure, continued depression of the myocardium with beta-blocking agents over a period of time can, in some cases, lead to cardiac failure. At the first sign or symptom of cardiac failure, ISTALOL should be discontinued [see Contraindications (4.2)].
5.3 Obstructive Pulmonary Disease
Patients with chronic obstructive pulmonary disease (e.g., chronic bronchitis, emphysema) of mild or moderate severity, bronchospastic disease, or a history of bronchospastic disease [other than bronchial asthma or a history of bronchial asthma in which ISTALOL is contraindicated] should, in general, not receive beta-blocking agents, including ISTALOL [see Contraindications (4.1)].
5.4 Increased Reactivity to Allergens
While taking beta-blockers, patients with a history of atopy or a history of severe anaphylactic reactions to a variety of allergens may be more reactive to repeated accidental, diagnostic, or therapeutic challenge with such allergens. Such patients may be unresponsive to the usual doses of epinephrine used to treat anaphylactic reactions.
5.5 Potentiation of Muscle Weakness
Beta-adrenergic blockade has been reported to potentiate muscle weakness consistent with certain myasthenic symptoms (e.g., diplopia, ptosis, and generalized weakness). Timolol has been reported rarely to increase muscle weakness in some patients with myasthenia gravis or myasthenic symptoms.
5.6 Masking of Hypoglycemic Symptoms in Patients with Diabetes Mellitus
Beta-adrenergic blocking agents should be administered with caution in patients subject to spontaneous hypoglycemia or to diabetic patients (especially those with labile diabetes) who are receiving insulin or oral hypoglycemic agents. Beta-adrenergic receptor blocking agents may mask the signs and symptoms of acute hypoglycemia.
5.7 Masking of Thyrotoxicosis
Beta-adrenergic blocking agents may mask certain clinical signs (e.g., tachycardia) of hyperthyroidism. Patients suspected of developing thyrotoxicosis should be managed carefully to avoid abrupt withdrawal of beta-adrenergic blocking agents that might precipitate a thyroid storm.
5.8 Contamination of Topical Ophthalmic Products After Use
There have been reports of bacterial keratitis associated with the use of multidose containers of topical ophthalmic products. These containers had been inadvertently contaminated by patients who, in most cases, had a concurrent corneal disease or a disruption of the ocular epithelial surface [see Patient Counseling Information (17)].
5.9 Impairment of Beta-adrenergically Mediated Reflexes During Surgery
The necessity or desirability of withdrawal of beta-adrenergic blocking agents prior to major surgery is controversial. Beta-adrenergic receptor blockade impairs the ability of the heart to respond to beta-adrenergically mediated reflex stimuli. This may augment the risk of general anesthesia in surgical procedures. Some patients receiving beta-adrenergic receptor blocking agents have experienced protracted severe hypotension during anesthesia. Difficulty in restarting and maintaining the heartbeat has also been reported. For these reasons, in patients undergoing elective surgery, some authorities recommend gradual withdrawal of beta-adrenergic receptor blocking agents. If necessary during surgery, the effects of beta-adrenergic blocking agents may be reversed by sufficient doses of adrenergic agonists.
5.10 Angle-Closure Glaucoma
In patients with angle-closure glaucoma, the immediate objective of treatment is to reopen the angle. This may require constricting the pupil. Timolol maleate has little or no effect on the pupil. ISTALOL should not be used alone in the treatment of angle-closure glaucoma.
5.11 Cerebrovascular Insufficiency
Because of potential effects of beta-adrenergic blocking agents on blood pressure and pulse, these agents should be used with caution in patients with cerebrovascular insufficiency. If signs or symptoms suggesting reduced cerebral blood flow develop following initiation of therapy with ISTALOL, alternative therapy should be considered.
-
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The most frequently reported adverse reactions have been burning and stinging upon instillation in 38% of patients treated with ISTALOL. Additional reactions reported with ISTALOL at a frequency of 4 to 10% include: blurred vision, cataract, conjunctival injection, headache, hypertension, infection, itching and decreased visual acuity.
The following additional adverse reactions have been reported less frequently with ocular administration of this or other timolol maleate formulations.
Timolol (Ocular Administration)
Body as a Whole: Asthenia/fatigue and chest pain; Cardiovascular: Bradycardia, arrhythmia, hypotension, syncope, heart block, cerebral vascular accident, cerebral ischemia, cardiac failure, worsening of angina pectoris, palpitation, cardiac arrest, pulmonary edema, edema, claudication, Raynaud’s phenomenon and cold hands and feet; Digestive: Nausea, diarrhea, dyspepsia, anorexia, and dry mouth; Immunologic: Systemic lupus erythematosus; Nervous System/Psychiatric: Dizziness, increase in signs and symptoms of myasthenia gravis, paresthesia, somnolence, insomnia, nightmares, behavioral changes and psychic disturbances including depression, confusion, hallucinations, anxiety, disorientation, nervousness and memory loss; Skin: Alopecia and psoriasiform rash or exacerbation of psoriasis; Hypersensitivity: Signs and symptoms of systemic allergic reactions, including angioedema, urticaria, and localized and generalized rash; Respiratory: Bronchospasm (predominantly in patients with pre-existing bronchospastic disease), respiratory failure, dyspnea, nasal congestion, cough and upper respiratory infections; Endocrine: Masked symptoms of hypoglycemia in diabetic patients [see Warnings and Precautions (5.6)]; Special Senses: Signs and symptoms of ocular irritation including conjunctivitis, blepharitis, keratitis, ocular pain, discharge (e.g., crusting), foreign body sensation, itching and tearing, and dry eyes; ptosis, decreased corneal sensitivity; cystoid macular edema; visual disturbances including refractive changes and diplopia; pseudopemphigoid; choroidal detachment following filtration surgery [see Warnings and Precautions (5.12)]; Urogenital: Retroperitoneal fibrosis, decreased libido, impotence, and Peyronie’s disease.
6.2 Postmarketing Experience
Oral Timolol/Oral Beta-Blockers
The following additional adverse effects have been reported in clinical experience with ORAL timolol maleate or other ORAL beta-blocking agents and may be considered potential effects of ophthalmic timolol maleate: Allergic: Erythematous rash, fever combined with aching and sore throat, laryngospasm with respiratory distress; Body as a Whole: Extremity pain, decreased exercise tolerance, weight loss; Cardiovascular: Worsening of arterial insufficiency, vasodilatation; Digestive: Gastrointestinal pain, hepatomegaly, vomiting, mesenteric arterial thrombosis, ischemic colitis; Hematologic: Nonthrombocytopenic purpura; thrombocytopenic purpura, agranulocytosis; Endocrine: Hyperglycemia, hypoglycemia; Skin: Pruritus, skin irritation, increased pigmentation, sweating; Musculoskeletal: Arthralgia; Nervous System/Psychiatric: Vertigo, local weakness, diminished concentration, reversible mental depression progressing to catatonia, an acute reversible syndrome characterized by disorientation for time and place, emotional lability, slightly clouded sensorium and decreased performance on neuropsychometrics; Respiratory: Rales, bronchial obstruction; Urogenital: Urination difficulties.
-
7 DRUG INTERACTIONS
7.1 Beta-Adrenergic Blocking Agents
Patients who are receiving a beta-adrenergic blocking agent orally and ISTALOL should be observed for potential additive effects of beta-blockade, both systemic and on intraocular pressure. The concomitant use of two topical beta-adrenergic blocking agents is not recommended.
7.2 Calcium Antagonists
Caution should be used in the co-administration of beta-adrenergic blocking agents, such as ISTALOL, and oral or intravenous calcium antagonists because of possible atrioventricular conduction disturbances, left ventricular failure, and hypotension. In patients with impaired cardiac function, co-administration should be avoided.
7.3 Catecholamine-Depleting Drugs
Close observation of the patient is recommended when a beta-blocker is administered to patients receiving catecholamine-depleting drugs such as reserpine, because of possible additive effects and the production of hypotension and/or marked bradycardia, which may result in vertigo, syncope, or postural hypotension.
7.4 Digitalis and Calcium Antagonists
The concomitant use of beta-adrenergic blocking agents with digitalis and calcium antagonists may have additive effects in prolonging atrioventricular conduction time.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Teratogenic Effects
Teratogenicity studies have been performed in animals.
Teratogenicity studies with timolol in mice, rats, and rabbits at oral doses up to 50 mg/kg/day (7,000 times the systemic exposure following the maximum recommended human ophthalmic dose) demonstrated no evidence of fetal malformations. Although delayed fetal ossification was observed at this dose in rats, there were no adverse effects on postnatal development of offspring. Doses of 1,000 mg/kg/day (142,000 times the systemic exposure following the maximum recommended human ophthalmic dose) were maternotoxic in mice and resulted in an increased number of fetal resorptions. Increased fetal resorptions were also seen in rabbits at doses of 14,000 times the systemic exposure following the maximum recommended human ophthalmic dose, in this case without apparent maternotoxicity.
There are no adequate and well-controlled studies in pregnant women. ISTALOL should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.8.3 Nursing Mothers
Timolol has been detected in human milk following oral and ophthalmic drug administration. Because of the potential for serious adverse reactions from ISTALOL in nursing infants, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
-
10 OVERDOSAGE
There have been reports of inadvertent overdosage with ISTALOL resulting in systemic effects similar to those seen with systemic beta-adrenergic blocking agents such as dizziness, headache, shortness of breath, bradycardia, bronchospasm, and cardiac arrest.
An in vitro hemodialysis study, using 14C timolol added to human plasma or whole blood, showed that timolol was readily dialyzed from these fluids; however, a study of patients with renal failure showed that timolol did not dialyze readily. -
11 DESCRIPTION
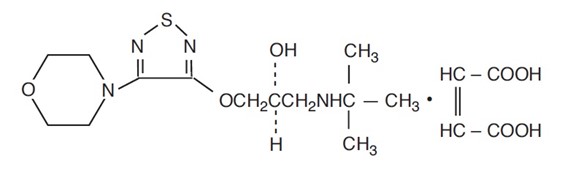
ISTALOL (timolol maleate ophthalmic solution) 0.5% is a non-selective beta-adrenergic receptor blocking agent. Its chemical name is (-)-1-(tert-butylamino)-3-[(4-morpholino-1,2,5-thiadiazol-3-yl)oxy]-2-propanol maleate (1:1) (salt). Timolol maleate possesses an asymmetric carbon atom in its structure and is provided as the levo-isomer.
Its molecular formula is C13H24N4O3S-C4H4O4 and its structural formula is:
Timolol maleate has a molecular weight of 432.49. It is a white, odorless, crystalline powder which is soluble in water, methanol, and alcohol. ISTALOL is stable at room temperature. ISTALOL ophthalmic solution is supplied as a sterile, isotonic, buffered, aqueous solution of timolol maleate in a single strength. It has a pH of 6.5 to 7.5 and an osmolality of 275 to 330 mOsm/kg.
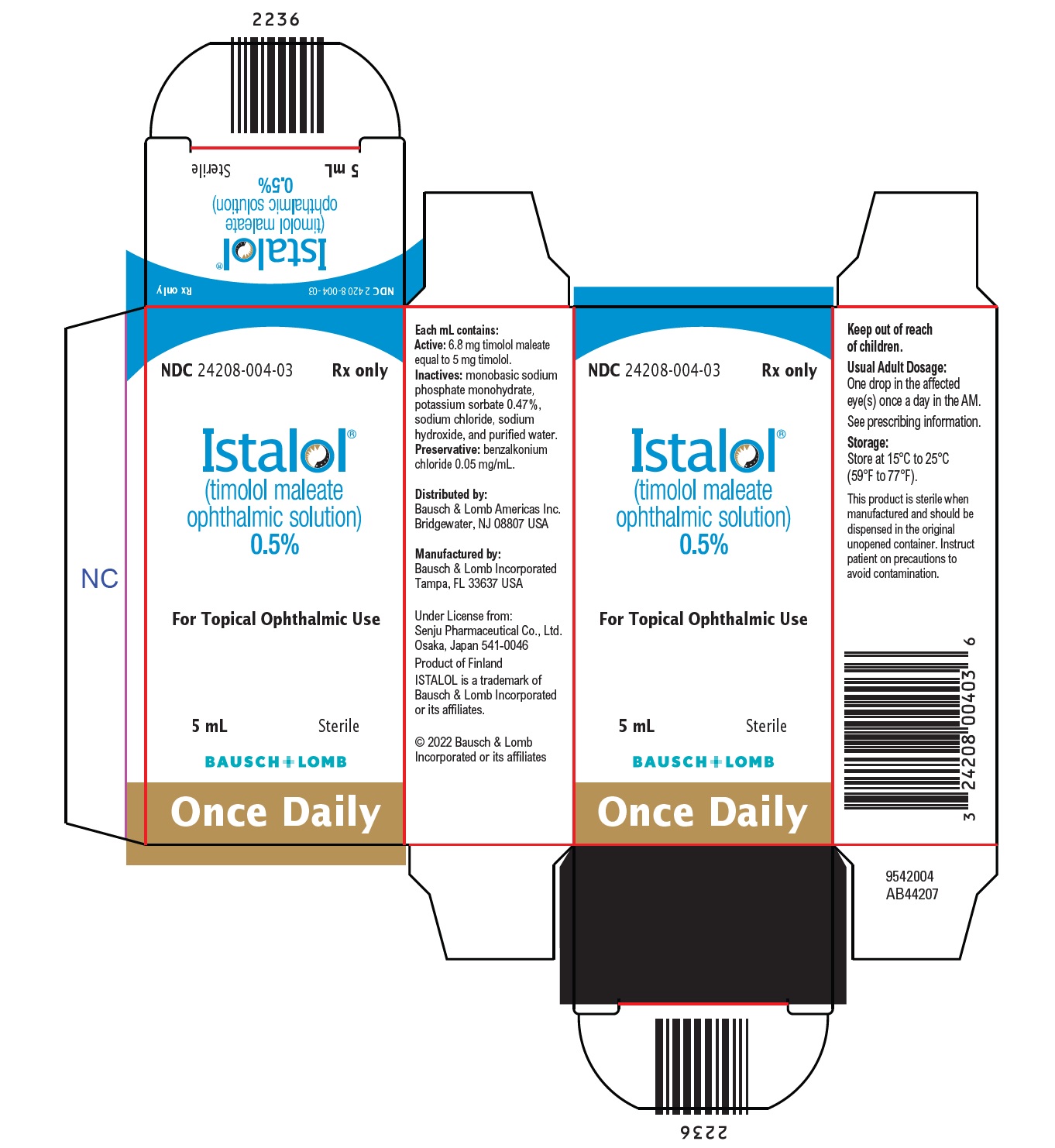
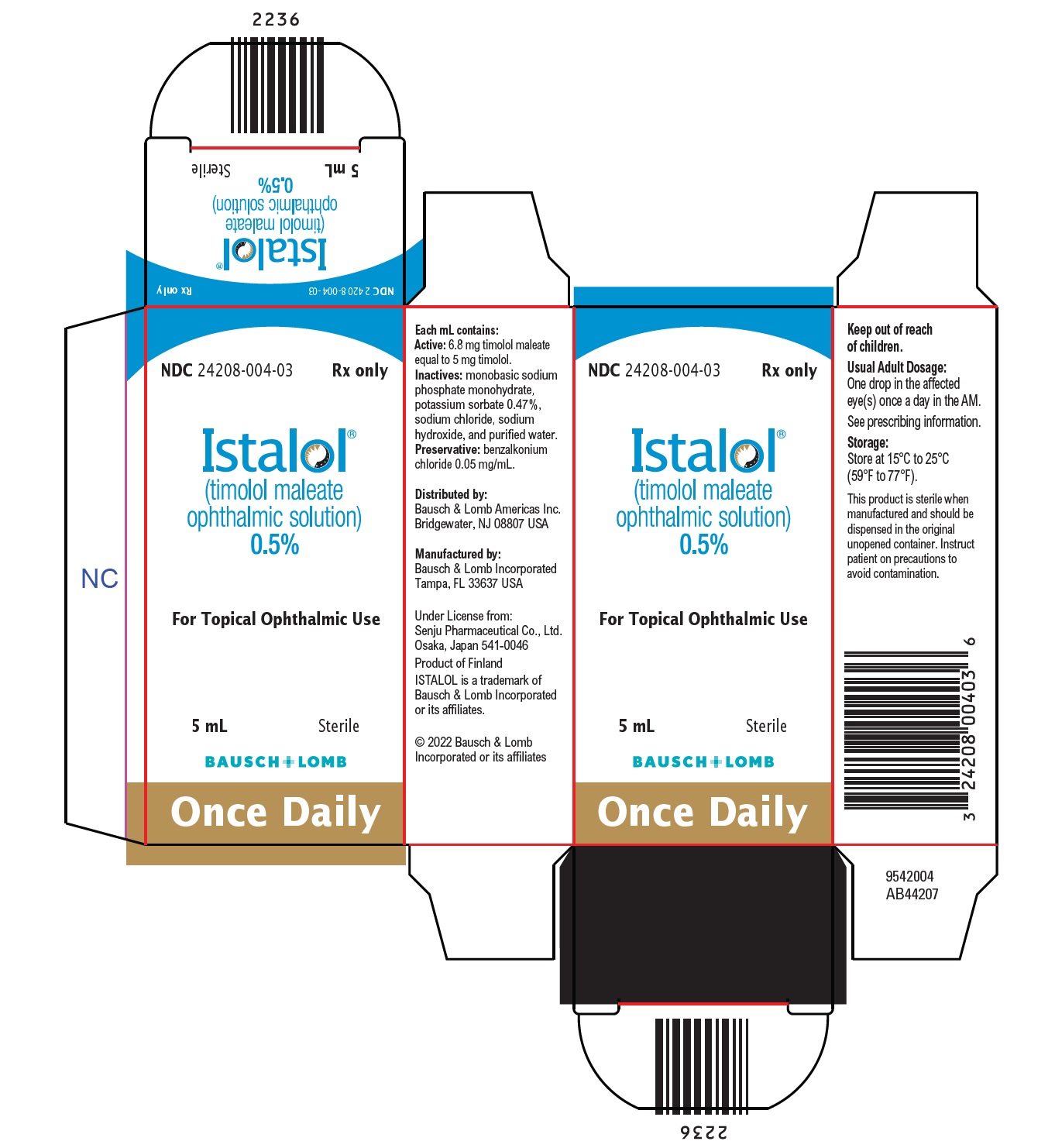
Each mL of ISTALOL contains the active ingredient 5 mg of timolol (6.8 mg of timolol maleate) with the inactive ingredients monobasic sodium phosphate monohydrate, potassium sorbate 0.47%, sodium chloride, sodium hydroxide, and purified water. Preservative added: benzalkonium chloride (0.05 mg/mL).
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Timolol maleate is a beta1 and beta2 (non-selective) adrenergic receptor blocking agent that does not have significant intrinsic sympathomimetic, direct myocardial depressant, or local anesthetic (membrane-stabilizing) activity.
Beta-adrenergic receptor blockade reduces cardiac output in both healthy subjects and patients with heart disease. In patients with severe impairment of myocardial function, beta-adrenergic receptor blockade may inhibit the stimulatory effect of the sympathetic nervous system necessary to maintain adequate cardiac function.Beta-adrenergic receptor blockade in the bronchi and bronchioles results in increased airway resistance from unopposed parasympathetic activity. Such an effect in patients with asthma or other bronchospastic conditions is potentially dangerous.
ISTALOL ophthalmic solution, when applied topically in the eye, has the action of reducing elevated as well as normal intraocular pressure, whether or not accompanied by glaucoma. Elevated intraocular pressure is a major risk factor in the pathogenesis of glaucomatous visual field loss. The higher the level of intraocular pressure, the greater the likelihood of glaucomatous visual field loss and optic nerve damage.
The onset of reduction in intraocular pressure following administration of ISTALOL can usually be detected within one-half hour after a single dose. The maximum effect usually occurs in one to two hours and significant lowering of intraocular pressure can be maintained for periods as long as 24 hours with a single dose. Repeated observations over a period of one year indicate that the intraocular pressure lowering effect of ISTALOL is well maintained.
The precise mechanism of the ocular hypotensive action of ISTALOL is not clearly established at this time. Tonography and fluorophotometry studies in man suggest that its predominant action may be related to reduced aqueous formation. However, in some studies a slight increase in outflow facility was also observed.12.3 Pharmacokinetics
In a study of plasma drug concentration in 12 healthy subjects, the systemic exposure to timolol was determined following twice daily administration of ISTALOL (exaggerated regimen) for eight days. With ISTALOL, mean plasma concentrations of timolol were 0.68 ng/mL and 0.88 ng/mL two hours after the first dose and the dose on the eighth day, respectively.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a two-year study of timolol maleate administered orally to rats, there was a statistically significant increase in the incidence of adrenal pheochromocytomas in male rats administered 300 mg/kg/day (approximately 42,000 times the systemic exposure following the maximum recommended human ophthalmic dose). Similar differences were not observed in rats administered oral doses equivalent to approximately 14,000 times the maximum recommended human ophthalmic dose.
In a lifetime oral study in mice, there were statistically significant increases in the incidence of benign and malignant pulmonary tumors, benign uterine polyps and mammary adenocarcinomas in female mice at 500 mg/kg/day, (approximately 71,000 times the systemic exposure following the maximum recommended human ophthalmic dose), but not at 5 or 50 mg/kg/day (approximately 700 or 7,000, respectively, times the systemic exposure following the maximum recommended human ophthalmic dose). In a subsequent study in female mice, in which post-mortem examinations were limited to the uterus and the lungs, a statistically significant increase in the incidence of pulmonary tumors was again observed at 500 mg/kg/day.
The increased occurrence of mammary adenocarcinomas was associated with elevations in serum prolactin which occurred in female mice administered oral timolol at 500 mg/kg/day, but not at doses of 5 or 50 mg/kg/day. An increased incidence of mammary adenocarcinomas in rodents has been associated with administration of several other therapeutic agents that elevate serum prolactin, but no correlation between serum prolactin levels and mammary tumors has been established in humans. Furthermore, in adult human female subjects who received oral dosages of up to 60 mg of timolol maleate (the maximum recommended human oral dosage), there were no clinically meaningful changes in serum prolactin.
Timolol maleate was devoid of mutagenic potential when tested in vivo(mouse) in the micronucleus test and cytogenetic assay (doses up to 800 mg/kg) and in vitroin a neoplastic cell transformation assay (up to 100 mcg/mL). In Ames tests, the highest concentrations of timolol employed, 5,000 or 10,000 mcg/plate, were associated with statistically significant elevations of revertants observed with tester strain TA100 (in seven replicate assays), but not in the remaining three strains. In the assays with tester strain TA100, no consistent dose response relationship was observed, and the ratio of test to control revertants did not reach 2. A ratio of 2 is usually considered the criterion for a positive Ames test.
Reproduction and fertility studies in rats demonstrated no adverse effect on male or female fertility at doses up to 21,000 times the systemic exposure following the maximum recommended human ophthalmic dose.
-
14 CLINICAL STUDIES
In a controlled, double-masked, parallel study in 332 patients with untreated intraocular pressures of 22 mm Hg or greater, ISTALOL 0.5% administered once daily (AM) was equivalent to timolol maleate ophthalmic solution 0.5% administered twice daily. In both groups, mean intraocular pressure decreased from 25 mm Hg at baseline to 18 mm Hg at peak and 19 mm Hg at trough. ISTALOL was generally well tolerated, and 3% of patients had treatment discontinued for adverse events judged related to treatment. There was a slight decrease in cardiovascular function consistent with known systemic absorption of β-adrenoceptor antagonists.
- 16 HOW SUPPLIED/STORAGE AND HANDLING
-
17 PATIENT COUNSELING INFORMATION
Patients with bronchial asthma, a history of bronchial asthma, severe chronic obstructive pulmonary disease, sinus bradycardia, second or third degree atrioventricular block, or cardiac failure should be advised not to take this product [see Contraindications (4.1, 4.2)].
Patients should also be instructed that ocular solutions, if handled improperly or if the tip of the dispensing container contacts the eye or surrounding structures, can become contaminated by common bacteria known to cause ocular infections. Serious damage to the eye and subsequent loss of vision may result from using contaminated solutions [see Warnings and Precautions (5.8)].
Patients should also be advised that if they have ocular surgery or develop an intercurrent ocular condition (e.g., trauma or infection), they should immediately seek their physician’s advice concerning the continued use of the present multidose container.
If more than one topical ophthalmic drug is being used, the drugs should be administered at least five minutes apart.
Patients should be advised that ISTALOL contains benzalkonium chloride which may be absorbed by soft contact lenses. Contact lenses should be removed prior to administration of the solution. Lenses may be reinserted 15 minutes following ISTALOL administration.
-
SPL UNCLASSIFIED SECTION
Distributed by:
Bausch & Lomb Americas Inc.
Bridgewater, NJ 08807 USAManufactured by:
Bausch & Lomb Incorporated
Tampa, FL 33637 USAUnder License from:
Senju Pharmaceutical Co., Ltd.
Osaka, Japan 541-0046
ISTALOL is a trademark of Bausch & Lomb Incorporated or its affiliates.
© 2022 Bausch & Lomb Incorporated or its affiliates9733101 (Folded)
9733201 (Flat) - PACKAGE/LABEL PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ISTALOL
timolol maleate solution/ dropsProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:24208-004 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TIMOLOL MALEATE (UNII: P8Y54F701R) (TIMOLOL ANHYDROUS - UNII:5JKY92S7BR) TIMOLOL ANHYDROUS 5 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) 0.05 mg in 1 mL SODIUM PHOSPHATE, MONOBASIC, MONOHYDRATE (UNII: 593YOG76RN) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM HYDROXIDE (UNII: 55X04QC32I) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:24208-004-03 1 in 1 CARTON 08/02/2004 1 5 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 2 NDC:24208-004-01 1 in 1 CARTON 08/02/2004 2 2.5 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 3 NDC:24208-004-02 1 in 1 CARTON 08/02/2004 3 2.5 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021516 08/02/2004 Labeler - Bausch & Lomb Incorporated (196603781) Establishment Name Address ID/FEI Business Operations Bausch & Lomb, Incorporated 079587625 MANUFACTURE(24208-004)