DEXCHLORPHENIRAMINE MALEATE- dexchlorpheniramine maleate solution
Morton Grove Pharmaceuticals, Inc.
----------
DEXCHLORPHENIRAMINE MALEATE ORAL SOLUION, USP
2mg/5mL
DESCRIPTION
Each 5 mL (teaspoonful) contains:
Dexchlorpheniramine Maleate, USP 2 mg
Alcohol not more than 7.0%
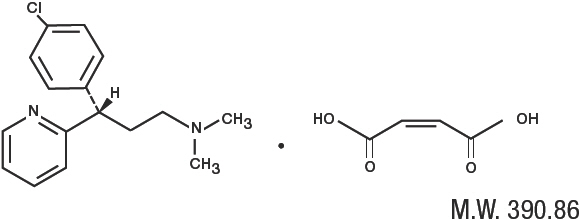
Dexchlorpheniramine Maleate, USP, an antihistamine agent, is a white, odorless crystalline powder that is freely soluble in water. The molecular formula is C16H19CIN2•C4H4O4, designated chemically as (+)-2-[p-Chloro-α-[2-(dimethylamino)ethyl]benzyl]pyridine maleate (1:1).
Inactive Ingredients
Citric Acid; Dehydrated Alcohol; FD&C Red No. 40; Glycerin; Liquid Sugar; Menthol; Methylparaben; Natural and Artificial Orange Juice Flavor; Propylene Glycol; Propylparaben and Purified Water. May also contain Sodium Citrate for pH adjustment. The pH range is between 5.0 and 6.5.
CLINICAL PHARMACOLOGY
Dexchlorpheniramine maleate is an antihistamine with anticholinergic (drying) and sedative side effects. Antihistamines appear to compete with histamine for cell receptor sites on effector cells.
INDICATIONS AND USAGE
Perennial and seasonal allergic rhinitis
Vasomotor rhinitis
Allergic conjunctivitis due to inhalant allergens and foods
Mild, uncomplicated allergic skin manifestations of urticaria and angioedema
Amelioration of allergic reactions to blood or plasma
Dermographism
As therapy for anaphylactic reactions adjunctive to epinephrine and other standard measures after the acute manifestations have been controlled.
CONTRAINDICATIONS
Use in Nursing Mothers:
Because of the higher risk of antihistamines for infants generally and for newborns and prematures in particular, antihistamine therapy is contraindicated in nursing mothers.
Use in Lower Respiratory Disease:
Antihistamines should NOT be used to treat lower respiratory tract symptoms including asthma.
Antihistamines are also contraindicated in the following conditions:
- Hypersensitivity to dexchlorpheniramine maleate or other antihistamines of similar chemical structure
- Monoamine oxidase inhibitor therapy (See Drug Interaction section)
WARNINGS
Antihistamines should be used with considerable caution in patients with:
- Narrow angle glaucoma
- Stenosing peptic ulcer
- Pyloroduodenal obstruction
- Symptomatic prostatic hypertrophy
- Bladder neck obstruction
Use in Children
In infants and children, especially, antihistamines in overdosage may cause hallucinations, convulsions, or death.
As in adults, antihistamines may diminish mental alertness in children. In the young child, particularly, they may produce excitation.
Use in Pregnancy:
Experience with this drug in pregnant women is inadequate to determine whether there exists a potential for harm to the developing fetus.
Use with CNS Depressants:
Dexchlorpheniramine Maleate, USP has additive effects with alcohol and other CNS depressants (hypnotics, sedatives, tranquilizers, etc.).
PRECAUTIONS
Dexchlorpheniramine Maleate, USP has an atropine-like action and, therefore, should be used with caution in patients with:
- History of bronchial asthma
- Increased intraocular pressure
- Hyperthyroidism
- Cardiovascular disease
- Hypertension
ADVERSE REACTIONS
- General: Urticaria, drug rash, anaphylactic shock, photosensitivity, excessive perspiration, chills, dryness of mouth, nose and the throat.
- Cardiovascular System: Hemolytic anemia, thrombocytopenia, agranulocytosis.
- Hematologic System: Hemolytic anemia, thrombocytopenia, agranulocytosis.
- Nervous System: Sedation, sleepiness, dizziness, disturbed coordination, fatigue, confusion, restlessness, excitation, nervousness, tremor, irritability, insomnia, euphoria, paresthesias, blurred vision, diplopia, vertigo, tinnitus, acute labyrinthitis, hysteria, neuritis, convulsions.
- G.I. System: Epigastric distress, anorexia, nausea, vomiting, diarrhea, constipation.
- G.U. System: Urinary frequency, difficult urination, urinary retention, early menses.
- Respiratory System: Thickening of bronchial secretions, tightness of chest and wheezing, nasal stuffiness.
OVERDOSAGE
Antihistamine overdosage reactions may vary from central nervous system depression to stimulation. Stimulation is particularly likely in children. Atropine-like signs and symptoms—dry mouth, fixed, dilated pupils, flushing, and gastrointestinal symptoms may also occur.
If vomiting has not occurred spontaneously the patient should be induced to vomit. This is best done by having the patient drink a glass of water or milk after which the patient should be made to gag. Precautions against aspiration must be taken, especially in infants and children.
Saline cathartics, such as milk of magnesia, draw water into the bowel by osmosis and therefore, are valuable for their action in rapid dilution of bowel content.
Stimulants should not be used.
Vasopressors may be used to treat hypotension.
DOSAGE AND ADMINISTRATION
DOSAGE SHOULD BE INDIVIDUALIZED ACCORDING TO THE NEEDS AND THE RESPONSE OF THE PATIENT.
HOW SUPPLIED
Dexchlorpheniramine Maleate Oral Solution, USP 2 mg/5 mL is supplied as a red-orange colored, orange flavored liquid in the following sizes:
16 fl oz (473 mL)
| DEXCHLORPHENIRAMINE MALEATE
dexchlorpheniramine maleate solution |
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| Labeler - Morton Grove Pharmaceuticals, Inc. (801897505) |
| Registrant - Morton Grove Pharmaceuticals, Inc. (801897505) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Morton Grove Pharmaceuticals, Inc. | 801897505 | ANALYSIS(60432-539) , MANUFACTURE(60432-539) , PACK(60432-539) | |
