Label: SULFADIAZINE tablet
- NDC Code(s): 0185-0757-01, 0185-0757-10, 0185-0757-30
- Packager: Eon Labs, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated June 30, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
Sulfadiazine is an oral sulfonamide antibacterial agent.
Each tablet, for oral administration, contains 500 mg sulfadiazine. In addition, each tablet contains the following inactive ingredients: croscarmellose sodium, docusate sodium, microcrystalline cellulose, povidone, sodium benzoate, sodium starch glycolate and stearic acid.
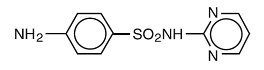
Sulfadiazine occurs as a white or slightly yellow powder. It is odorless or nearly so and slowly darkens on exposure to light. It is practically insoluble in water and slightly soluble in alcohol. The chemical name of sulfadiazine is N1-2-pyrimidinylsulfanilamide. The molecular formula is C10H10N4O2S. It has a molecular weight of 250.27. The structural formula is shown below:
Most sulfonamides slowly darken on exposure to light.
-
CLINICAL PHARMACOLOGY
The systemic sulfonamides are bacteriostatic agents having a similar spectrum of activity. Sulfonamides competitively inhibit bacterial synthesis of folic acid (pteroylglutamic acid) from aminobenzoic acid. Resistant strains are capable of utilizing folic acid precursors or preformed folic acid.
Sulfonamides exist in the blood in 3 forms - free, conjugated (acetylated and possibly others) and protein bound. The free form is considered to be the therapeutically active one.
Sulfadiazine given orally is readily absorbed from the gastrointestinal tract. After a single 2 g oral dose, a peak of 6.04 mg/100 mL is reached in 4 hours; of this, 4.65 mg/100 mL is free drug.
When a dose of 100 mg/kg of body weight is given initially and followed by 50 mg/kg every 6 hours, blood levels of free sulfadiazine are about 7 mg/100mL. Protein binding is 38% to 48%. Sulfadiazine diffuses into the cerebrospinal fluid; free drug reaches 32% to 65% of blood levels and total drug 40% to 60%.
Sulfadiazine is excreted largely in the urine, where concentrations are 10 to 25 times greater than serum levels. Approximately 10% of a single oral dose is excreted in the first 6 hours, 50% within 24 hours and 60% to 85% in 48 to 72 hours. Of the amount excreted in the urine, 15% to 40% is in the acetyl form.
-
INDICATIONS AND USAGE
sulfADIAZINE Tablets, USP are indicated in the following conditions:
Chancroid
Trachoma
Inclusion conjunctivitis
Nocardiosis
Urinary tract infections (primarily pyelonephritis, pyelitis and cystitis) in the absence of obstructive uropathy or foreign bodies, when these infections are caused by susceptible strains of the following organisms: Escherichia coli, Klebsiella species, Enterobacter species, Staphylococcus aureus, Proteus mirabilis and P. vulgaris. Sulfadiazine should be used for urinary tract infections only after use of more soluble sulfonamides has been unsuccessful.
Toxoplasmosis encephalitis in patients with and without acquired immunodeficiency syndrome, as adjunctive therapy with pyrimethamine.
Malaria due to chloroquine-resistant strains of Plasmodium falciparum, when used as adjunctive therapy.
Prophylaxis of meningococcal meningitis when sulfonamide-sensitive group A strains are known to prevail in family groups or larger closed populations (the prophylactic usefulness of sulfonamides when group B or C infections are prevalent is not proved and may be harmful in closed population groups).
Meningococcal meningitis, when the organism has been demonstrated to be susceptible.
Acute otitis media due to Haemophilus influenzae, when used concomitantly with adequate doses of penicillin.
Prophylaxis against recurrences of rheumatic fever, as an alternative to penicillin.
H. influenzae meningitis, as adjunctive therapy with parental streptomycin.
IMPORTANT NOTES
In vitro sulfonamide susceptibility tests are not always reliable. The test must be carefully coordinated with bacteriologic and clinical response. When the patient is already taking sulfonamides, follow-up cultures should have aminobenzoic acid added to the culture media.
Currently, the increasing frequency of resistant organisms limits the usefulness of antibacterial agents, including the sulfonamides, especially in the treatment of recurrent and complicated urinary tract infections.
Wide variation in blood levels may result with identical doses. Blood levels should be measured in patients receiving sulfonamides for serious infections. Free sulfonamide blood levels of 5 mg to 15 mg per 100 mL may be considered therapeutically effective for most infections and blood levels of 12 mg to 15 mg per 100 mL may be considered optimal for serious infections. Twenty mg per 100 mL should be the maximum total sulfonamide level, since adverse reactions occur more frequently above this level.
-
CONTRAINDICATIONS
Sulfadiazine is contraindicated in the following circumstances: Hypersensitivity to sulfonamides.
In infants less than 2 months of age (except as adjunctive therapy with pyrimethamine in the treatment of congenital toxoplasmosis).
In pregnancy at term and during the nursing period, because sulfonamides cross the placenta and are excreted in breast milk and may cause kernicterus.
-
WARNINGS
The sulfonamides should not be used for the treatment of group A betahemolytic streptococcal infections. In an established infection, they will not eradicate the streptococcus and, therefore, will not prevent sequelae such as rheumatic fever and glomerulonephritis.
Deaths associated with the administration of sulfonamides have been reported from hypersensitivity reactions, agranulocytosis, aplastic anemia and other blood dyscrasias.
The presence of such clinical signs as sore throat, fever, pallor, purpura or jaundice may be early indications of serious blood disorders.
The frequency of renal complications is considerably lower in patients receiving the more soluble sulfonamides.
-
PRECAUTIONS
General
Sulfonamides should be given with caution to patients with impaired renal or hepatic function and to those with severe allergy or bronchial asthma.
Hemolysis may occur in individuals deficient in glucose-6-phosphate dehydrogenase. This reaction is dose related.
Adequate fluid intake must be maintained in order to prevent crystalluria and stone formation.
Information for Patients
Patients should be instructed to drink an eight ounce glass of water with each dose of medication and at frequent intervals throughout the day. Caution patients to report promptly the onset of sore throat, fever, pallor, purpura or jaundice when taking this drug, since these may be early indications of serious blood disorders.
Laboratory Tests
Complete blood counts and urinalyses with careful microscopic examinations should be done frequently in patients receiving sulfonamides.
Drug Interactions
Administration of a sulfonamide may increase the effect of oral anticoagulants and methotrexate, probably by displacement of these drugs from binding sites on plasma albumin. Potentiation of the action of sulfonylurea hypoglycemic agents, thiazide diuretics and uricosuric agents may also be noted. This may also be due to displacement of the drugs from albumin or a pharmacodynamic mechanism may play a role. Conversely, agents such as indomethacin, probenecid and salicylates may displace sulfonamides from plasma albumin and increase the concentrations of free drug in plasma.
Carcinogenesis, Mutagenesis, Impairment of Fertility
The sulfonamides bear certain chemical similarities to some goitrogens. Rats appear to be especially susceptible to the goitrogenic effects of sulfonamides and long-term administration has produced thyroid malignancies in rats.
Pregnancy
Teratogenic Effects
The safe use of sulfonamides in pregnancy has not been established. The teratogenic potential of most sulfonamides has not been thoroughly investigated in either animals or humans. However, a significant increase in the incidence of cleft palate and other bony abnormalities in offspring has been observed when certain sulfonamides of the short, intermediate and long acting types were given to pregnant rats and mice in high oral doses (7 to 25 times the human therapeutic dose).
Nursing Mothers
Sulfadiazine is contraindicated for use in nursing mothers because the sulfonamides cross the placenta, are excreted in breast milk and may cause kernicterus.
Because of the potential for serious adverse reactions in nursing infants from sulfadiazine, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother. See CONTRAINDICATIONS.
Pediatric Use
Sulfadiazine is contraindicated in infants less than 2 months of age (except as adjunctive therapy with pyrimethamine in the treatment of congenital toxoplasmosis). See CONTRAINDICATIONS and DOSAGE AND ADMINISTRATION.
-
ADVERSE REACTIONS
Blood Dyscrasias
Agranulocytosis, aplastic anemia, thrombocytopenia, leukopenia, hemolytic anemia, purpura, hypoprothrombinemia and methemoglobinemia.
Allergic Reactions
Erythema multiforme (Stevens-Johnson syndrome), generalized skin eruptions, epidermal necrolysis, urticaria, serum sickness, pruritus, exfoliative dermatitis, anaphylactoid reactions, periorbital edema, conjunctival and scleral injection, photosensitization, arthralgia, allergic myocarditis, drug fever and chills.
Gastrointestinal Reactions
Nausea, emesis, abdominal pains, hepatitis, diarrhea, anorexia, pancreatitis and stomatitis.
C.N.S. Reactions
Headache, peripheral neuritis, mental depression, convulsions, ataxia, hallucinations, tinnitus, vertigo and insomnia.
Renal
Crystalluria, stone formation, toxic nephrosis with oliguria and anuria; periarteritis nodosa and lupus erythematosus phenomenon have been noted.
Miscellaneous Reactions
The sulfonamides bear certain chemical similarities to some goitrogens, diuretics (acetazolamide and the thiazides) and oral hypoglycemic agents. Goiter production, diuresis and hypoglycemia have occurred rarely in patients receiving sulfonamides. Cross-sensitivity may exist with these agents.
-
DOSAGE AND ADMINISTRATION
SYSTEMIC SULFONAMIDES ARE CONTRAINDICATED IN INFANTS UNDER 2 MONTHS OF AGE except as adjunctive therapy with pyrimethamine in the treatment of congenital toxoplasmosis.
Usual Dosage for Infants over 2 Months of Age and Children
Initially, one-half the 24-hour dose. Maintenance, 150 mg/kg or 4 g/m2, divided into 4 to 6 doses, every 24 hours, with a maximum of 6 g every 24 hours. Rheumatic fever prophylaxis, under 30 kg (66 pounds), 500 mg every 24 hours; over 30 kg (66 pounds), 1 g every 24 hours.
-
HOW SUPPLIED
sulfADIAZINE Tablets, USP for oral administration are available as
500 mg
White, unscored, capsule-shaped tablets, debossed “E 757” on one face and supplied as:
NDC 0185-0757-30 bottles of 30
NDC 0185-0757-01 bottles of 100
NDC 0185-0757-10 bottles of 1000
Storage
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Protect from moisture.
Dispense contents in a tight, light-resistant container as defined in the USP with a child-resistant closure, as required.
KEEP TIGHTLY CLOSED.
KEEP OUT OF THE REACH OF CHILDREN.
To report SUSPECTED ADVERSE REACTIONS, contact Sandoz Inc. at 1-800-525-8747 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Manufactured for
Sandoz Inc.
Princeton, NJ 08540
Manufactured by
Epic Pharma, LLC
Laurelton, NY 11413
OS7190
Rev. 06/2020
MF0757REV06/20
- sulfADIAZINE Tablets USP, 500 mg x 100 Tablets - Label
-
INGREDIENTS AND APPEARANCE
SULFADIAZINE
sulfadiazine tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0185-0757 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SULFADIAZINE (UNII: 0N7609K889) (SULFADIAZINE - UNII:0N7609K889) SULFADIAZINE 500 mg Inactive Ingredients Ingredient Name Strength CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) DOCUSATE SODIUM (UNII: F05Q2T2JA0) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) STEARIC ACID (UNII: 4ELV7Z65AP) Product Characteristics Color WHITE Score no score Shape CAPSULE Size 17mm Flavor Imprint Code E757 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0185-0757-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 05/22/2013 10/22/2017 2 NDC:0185-0757-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 07/29/1994 05/31/2024 3 NDC:0185-0757-10 1000 in 1 BOTTLE; Type 0: Not a Combination Product 07/29/1994 12/31/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA040091 07/29/1994 05/31/2024 Labeler - Eon Labs, Inc. (012656273)