ADAPALENE- adapalene gel
Glenmark Pharmaceuticals Inc., USA
----------
Adapalene Gel, 0.1%
DESCRIPTION
Adapalene gel 0.1%, containing adapalene USP, is used for the topical treatment of acne vulgaris. Each gram of adapalene gel 0.1% contains adapalene USP 0.1% (1 mg) in a vehicle consisting of carbomer homopolymer, edetate disodium, methylparaben, poloxamer 182, propylene glycol, purified water and sodium hydroxide.
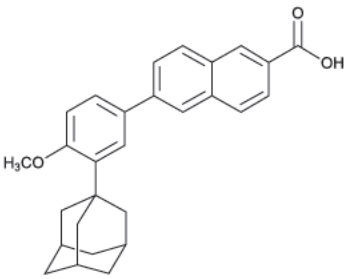
The chemical name of adapalene USP is 6-[3-(1-adamantyl)-4-methoxyphenyl]-2-naphthoic acid. Adapalene USP is a white to off-white powder which is sparingly soluble in tetrahydrofuran, very slightly soluble in diethyl ether. The molecular formula is C28H28O3 and molecular weight is 412.52. Adapalene USP is represented by the following structural formula:
CLINICAL PHARMACOLOGY
Adapalene is a chemically stable, retinoid-like compound. Biochemical and pharmacological profile studies have demonstrated that adapalene is a modulator of cellular differentiation, keratinization, and inflammatory processes all of which represent important features in the pathology of acne vulgaris.
Mechanistically, adapalene binds to specific retinoic acid nuclear receptors but does not bind to the cytosolic receptor protein. Although the exact mode of action of adapalene is unknown, it is suggested that topical adapalene may normalize the differentiation of follicular epithelial cells resulting in decreased microcomedone formation.
Pharmacokinetics:
Absorption of adapalene through human skin is low. Only trace amounts (<0.25 ng/mL) of parent substance have been found in the plasma of acne patients following chronic topical application of adapalene in controlled clinical trials. Excretion appears to be primarily by the biliary route.
CONTRAINDICATIONS
Adapalene should not be administered to individuals who are hypersensitive to adapalene or any of the components in the vehicle gel.
WARNINGS
Use of adapalene gel should be discontinued if hypersensitivity to any of the ingredients is noted. Patients with sunburn should be advised not to use the product until fully recovered.
PRECAUTIONS
General
If a reaction suggesting sensitivity or chemical irritation occurs, use of the medication should be discontinued. Exposure to sunlight, including sunlamps, should be minimized during the use of adapalene. Patients who normally experience high levels of sun exposure, and those with inherent sensitivity to sun, should be warned to exercise caution. Use of sunscreen products and protective clothing over treated areas is recommended when exposure cannot be avoided. Weather extremes, such as wind or cold, also may be irritating to patients under treatment with adapalene. Avoid contact with the eyes, lips, angles of the nose, and mucous membranes. The product should not be applied to cuts, abrasions, eczematous skin, or sunburned skin.
Certain cutaneous signs and symptoms such as erythema, dryness, scaling, burning, or pruritus may be experienced during treatment. These are most likely to occur during the first two to four weeks and will usually lessen with continued use of the medication. Depending upon the severity of adverse events, patients should be instructed to reduce the frequency of application or discontinue use.
Drug Interactions
As adapalene gel has the potential to produce local irritation in some patients, concomitant use of other potentially irritating topical products (medicated or abrasive soaps and cleansers, soaps and cosmetics that have a strong drying effect, and products with high concentrations of alcohol, astringents, spices, or lime) should be approached with caution. Particular caution should be exercised in using preparations containing sulfur, resorcinol, or salicylic acid in combination with adapalene gel. If these preparations have been used, it is advisable not to start therapy with adapalene gel until the effects of such preparations in the skin have subsided.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies with adapalene have been conducted in mice at topical doses of 0.3, 0.9, and 2.6 mg/kg/day and in rats at oral doses of 0.15, 0.5, and 1.5 mg/kg/day, approximately 4-75 times the maximal daily human topical dose. In the oral study, positive linear trends were observed in the incidence of follicular cell adenomas and carcinomas in the thyroid glands of female rats, and in the incidence of benign and malignant pheochromocytomas in the adrenal medullas of male rats.
No photocarcinogenicity studies were conducted. Animal studies have shown an increased tumorigenic risk with the use of pharmacologically similar drugs (e.g., retinoids) when exposed to UV irradiation in the laboratory or to sunlight. Although the significance of these studies to human use is not clear, patients should be advised to avoid or minimize exposure to either sunlight or artificial UV irradiation sources.
In a series of in vivo and in vitro studies, adapalene did not exhibit mutagenic or genotoxic activities.
Pregnancy
Teratogenic effects
Pregnancy Category C. No teratogenic effects were seen in rats at oral doses of adapalene 0.15 to 5.0 mg/kg/day, up to 120 times the maximal daily human topical dose. Cutaneous route teratology studies conducted in rats and rabbits at doses of 0.6, 2.0, and 6.0 mg/kg/day, up to 150 times the maximal daily human topical dose exhibited no fetotoxicity and only minimal increases in supernumerary ribs in rats. There are no adequate and well-controlled studies in pregnant women. Adapalene should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
ADVERSE REACTIONS
Some adverse effects such as erythema, scaling, dryness, pruritus, and burning will occur in 10-40% of patients. Pruritus or burning immediately after application also occurs in approximately 20% of patients. The following additional adverse experiences were reported in approximately 1% or less of patients: skin irritation, burning/stinging, erythema, sunburn, and acne flares. These are most commonly seen during the first month of therapy and decrease in frequency and severity thereafter. All adverse effects with use of adapalene gel during clinical trials were reversible upon discontinuation of therapy.
OVERDOSAGE
Adapalene gel is intended for cutaneous use only. If the medication is applied excessively, no more rapid or better results will be obtained and marked redness, peeling, or discomfort may occur. The acute oral toxicity of adapalene gel in mice and rats is greater than 10 mL/kg. Chronic ingestion of the drug may lead to the same side effects as those associated with excessive oral intake of Vitamin A.
DOSAGE AND ADMINISTRATION
Adapalene gel 0.1% should be applied once a day to affected areas after washing in the evening before retiring. A thin film of the gel should be applied, avoiding eyes, lips, and mucous membranes.
During the early weeks of therapy, an apparent exacerbation of acne may occur. This is due to the action of the medication on previously unseen lesions and should not be considered a reason to discontinue therapy. Therapeutic results should be noticed after eight to twelve weeks of treatment.
HOW SUPPLIED
Adapalene gel, 0.1% is supplied in the following size:
45 g laminate tube NDC 68462-403-55
| ADAPALENE
adapalene gel |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Glenmark Pharmaceuticals Inc., USA (130597813) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Glenmark Pharmaceuticals Limited | 677318665 | ANALYSIS(68462-403) , MANUFACTURE(68462-403) | |