FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Instructions for Using the GlucaGen HypoKit to Treat Severe Hypoglycemia
GlucaGen is for subcutaneous, intramuscular, or intravenous injection. Administer intravenously ONLY under medical supervision.
Instruct patients and their caregivers on the signs and symptoms of severe hypoglycemia. Because severe hypoglycemia requires the help of others to recover, instruct the patient to inform those around them about GlucaGen and its Instructions for Use. Administer GlucaGen as soon as possible when severe hypoglycemia is recognized.
Instruct the patient or caregiver to read the Instructions for Use at the time they receive a prescription for GlucaGen. Emphasize the following instructions to the patient or caregiver:
- •
- Using the supplied prefilled syringe, carefully insert the needle through the rubber stopper of the vial containing GlucaGen powder and inject all the liquid from the syringe into the vial.
- •
- Shake the vial gently until the powder is completely dissolved and no particles remain in the fluid. The reconstituted solution should be clear and colorless. Inspect visually for particulate matter and discoloration. If the resulting solution is cloudy or contains particulate matter do not use.
- •
- The reconstituted solution is 1 mg per mL glucagon.
- •
- Immediately after reconstitution, using the same syringe, withdraw the correct dose of GlucaGen.
- •
- Inject the solution subcutaneously or intramuscularly in the upper arm, thigh, or buttocks. In addition, healthcare providers may administer intravenously.
- •
- Call for emergency assistance immediately after administering the dose.
- •
- If there has been no response after 15 minutes, an additional dose of GlucaGen may be administered while waiting for emergency assistance.
- •
- When the patient has responded to the treatment and is able to swallow, give oral carbohydrates to restore the liver glycogen and prevent recurrence of hypoglycemia.
- •
- Discard any unused portion.
2.2 Dosage in Adults and Pediatric Patients for Using the GlucaGen HypoKit to Treat Severe Hypoglycemia
Adults and Pediatric Patients Weighing 25 kg or More or for Pediatric Patients with Unknown Weight 6 Years and Older
- •
- The recommended dosage is 1 mg (1 mL) injected subcutaneously or intramuscularly into the upper arm, thigh, or buttocks, or intravenously.
- •
- If there has been no response after 15 minutes, an additional 1 mg dose (1 mL) of GlucaGen may be administered using a new kit while waiting for emergency assistance.
Pediatric Patients Weighing Less Than 25 kg or for Pediatric Patients with Unknown Weight Less Than 6 Years of Age
- •
- The recommended dosage is 0.5 mg (0.5 mL) injected subcutaneously or intramuscularly into the upper arm, thigh, or buttocks, or intravenously.
- •
-
If there has been no response after 15 minutes, an additional 0.5 mg dose (0.5 mL) of GlucaGen may be administered using a new kit while waiting for emergency assistance.
For GlucaGen Diagnostic Kit and the GlucaGen 10-pack:
2.3 Important Administration Instruction for Using GlucaGen Diagnostic Kit and the GlucaGen 10-pack as a Diagnostic Aid
- •
- Reconstitute GlucaGen with 1 mL of Sterile Water for Injection. Using a syringe, withdraw all of the Sterile Water for Injection (if supplied) or 1 mL Sterile Water for Injection and inject into the GlucaGen vial.
- •
- Shake the vial gently until the powder is completely dissolved and no particles remain in the fluid. The reconstituted fluid should be clear and colorless. Inspect visually for particulate matter and discoloration. If the resulting solution is cloudy or contains particulate matter do not use.
- •
- The reconstituted solution is 1 mg per mL glucagon.
- •
- Immediately after reconstitution, inject the solution intravenously or intramuscularly into upper arm, thigh, or buttocks.
- •
- Discard any unused portion.
- •
- After the end of the diagnostic procedure, give oral carbohydrates to patients who have been fasting, if this is compatible with the diagnostic procedure.
2.4 Dosage in Adults for Using GlucaGen Diagnostic Kit and GlucaGen for Injection Single-Dose Vial as a Diagnostic Aid
- •
- The recommended diagnostic dose for relaxation of the stomach, duodenal bulb, duodenum, and small bowel is 0.2 mg to 0.5 mg administered intravenously or 1 mg administered intramuscularly; the recommended dose to relax the colon is 0.5 mg to 0.75 mg administered intravenously or 1 mg to 2 mg administered intramuscularly [see Clinical Pharmacology (12.2)].
- •
- The onset of action after an injection will depend on the organ under examination and route of administration [see Clinical Pharmacology (12.2)].
3 DOSAGE FORMS AND STRENGTHS
GlucaGen for injection is a white lyophilized powder supplied as follows:
Treatment of Severe Hypoglycemia
- •
- 1 mg single-dose vial of GlucaGen with a 1 mL single-dose syringe of Sterile Water for Injection, USP (GlucaGen HypoKit®)
Use as a Diagnostic Aid
- •
- 1 mg single-dose vial of GlucaGen
- •
- 1 mg single-dose vial of GlucaGen with a 1 mL single-dose vial of Sterile Water for Injection, USP (Diagnostic Kit)
4 CONTRAINDICATIONS
GlucaGen is contraindicated in patients with:
- •
- Pheochromocytoma because of the risk of substantial increase in blood pressure [see Warnings and Precautions (5.1)]
- •
- Insulinoma because of the risk of hypoglycemia [see Warnings and Precautions (5.2)]
- •
- Known hypersensitivity to glucagon or the excipients in GlucaGen. Allergic reactions have been reported with glucagon and include anaphylactic shock with breathing difficulties and hypotension [see Warnings and Precautions (5.3)]
- •
- Glucagonoma when used as a diagnostic aid because of risk of hypoglycemia [see Warnings and Precautions (5.8)]
5 WARNINGS AND PRECAUTIONS
5.1 Substantial Increase in Blood Pressure in Patients with Pheochromocytoma
GlucaGen is contraindicated in patients with pheochromocytoma because GlucaGen may stimulate the release of catecholamines from the tumor [see Contraindications (4)]. If the patient develops a substantial increase in blood pressure and a previously undiagnosed pheochromocytoma is suspected, 5 to 10 mg of phentolamine mesylate, administered intravenously, has been shown to be effective in lowering blood pressure for the short time that control would be needed.
5.2 Hypoglycemia in Patients with Insulinoma
In patients with insulinoma, administration of glucagon may produce an initial increase in blood glucose; however, GlucaGen administration may directly or indirectly (through an initial rise in blood glucose) stimulate exaggerated insulin release from an insulinoma and cause hypoglycemia. GlucaGen is contraindicated in patients with insulinoma [see Contraindications (4)]. If a patient develops symptoms of hypoglycemia after a dose of GlucaGen, give glucose orally or intravenously.
5.3 Hypersensitivity and Allergic Reactions
Allergic reactions have been reported with glucagon, these include generalized rash, and in some cases anaphylactic shock with breathing difficulties and hypotension. GlucaGen is contraindicated in patients with a prior hypersensitivity reaction [see Contraindications (4)].
5.4 Lack of Efficacy in Patients with Decreased Hepatic Glycogen
GlucaGen is effective in treating hypoglycemia only if sufficient hepatic glycogen is present. Patients in states of starvation, with adrenal insufficiency or chronic hypoglycemia may not have adequate levels of hepatic glycogen for GlucaGen administration to be effective. Patients with these conditions should be treated with glucose.
5.5 Necrolytic Migratory Erythema
Necrolytic migratory erythema (NME), a skin rash commonly associated with glucagonomas (glucagon-producing tumors) and characterized by scaly, pruritic erythematous plaques, bullae, and erosions, has been reported postmarketing following continuous glucagon infusion. NME lesions may affect the face, groin, perineum and legs or be more widespread. In the reported cases NME resolved with discontinuation of the glucagon, and treatment with corticosteroids was not effective. Should NME occur, consider whether the benefits of continuous glucagon infusion outweigh the risks.
5.6 Hyperglycemia in Patients with Diabetes Mellitus when Used as a Diagnostic Aid
Treatment with GlucaGen in patients with diabetes mellitus may cause hyperglycemia. Monitor diabetic patients for changes in blood glucose levels during treatment and treat if indicated.
5.7 Blood Pressure and Heart Rate Increase in Patients with Cardiac Disease when used as a Diagnostic Aid
GlucaGen may increase myocardial oxygen demand, blood pressure, and pulse rate which may be life-threatening in patients with cardiac disease. Cardiac monitoring is recommended in patients with cardiac disease during use of GlucaGen as a diagnostic aid, and an increase in blood pressure and pulse rate may require therapy.
5.8 Hypoglycemia in Patients with Glucagonoma
Glucagon administered to patients with glucagonoma may cause secondary hypoglycemia. Test patients suspected of having glucagonoma for blood levels of glucagon prior to use as a diagnostic aid as GlucaGen is contraindicated in this setting [see Contraindications (4)].
6 ADVERSE REACTIONS
The following important adverse reactions are described below and elsewhere in the labeling:
- •
- Hypersensitivity and Allergic Reactions [see Warnings and Precautions (5.3)]
- •
- Necrolytic Migratory Erythema [see Warnings and Precautions (5.5)]
- •
- Hyperglycemia in Patients with Diabetes Mellitus when Used as a Diagnostic Aid [see Warnings and Precautions (5.6)]
- •
- Blood Pressure and Heart Rate Increase in Patients with Cardiac Disease when Used as a Diagnostic Aid [see Warnings and Precautions (5.7)]
The following adverse reactions have been identified during post-approval use of glucagon. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- •
- Injection site reactions
- •
- Nausea
- •
- Vomiting
- •
- Headache
- •
- Dizziness
- •
- Asthenia
- •
- Pallor
- •
- Diarrhea
- •
- Somnolence
- •
- Generalized allergic reactions including anaphylactic shock with breathing difficulties and hypotension
- •
- Hypertension and tachycardia
- •
- Decreased blood pressure. Hypotension has been reported up to 2 hours after administration in patients receiving GlucaGen as premedication for upper gastrointestinal endoscopy procedures.
- •
- Hypoglycemia and hypoglycemic coma. Patients taking indomethacin may be more likely to experience hypoglycemia following glucagon administration [see Drug Interactions (7)].
- •
- Necrolytic Migratory Erythema (NME) cases have been reported post marketing in patients receiving continuous infusion of glucagon.
7 DRUG INTERACTIONS
Table 1: Clinically Significant Drug Interactions with GlucaGen
|
Beta-Blockers |
|
|
Clinical Impact: |
Patients taking beta-blockers may have a transient increase in pulse and blood pressure when given GlucaGen. |
|
Intervention: |
The increase in blood pressure and heart rate may require therapy in patients with coronary artery disease. |
|
Indomethacin |
|
|
Clinical Impact: |
In patients taking indomethacin, GlucaGen may lose its ability to raise blood glucose or may even produce hypoglycemia. |
|
Intervention: |
Monitor blood glucose levels during GlucaGen treatment of patients taking indomethacin. |
|
Anticholinergic Drugs |
|
|
Clinical Impact: |
The concomitant use of anticholinergic drugs and GlucaGen increase the risk of gastrointestinal adverse reactions due to additive effects on inhibition of gastrointestinal motility. |
|
Intervention: |
Concomitant use of anticholinergic drugs with GlucaGen for use as a diagnostic aid is not recommended. |
|
Warfarin |
|
|
Clinical Impact: |
GlucaGen may increase the anticoagulant effect of warfarin. |
|
Intervention: |
Monitor patients for unusual bruising or bleeding, as adjustments in warfarin dosage may be required. |
|
Insulin |
|
|
Clinical Impact: |
Insulin acts antagonistically to glucagon. |
|
Intervention: |
Monitor blood glucose when GlucaGen is used as a diagnostic aid in patients receiving insulin. |
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Available data from case reports and a small number of observational studies with glucagon use in pregnant women over decades of use have not identified a drug-associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. Multiple small studies have demonstrated a lack of transfer of pancreatic glucagon across the human placental barrier during early gestation. In rat and rabbit reproduction studies, no embryofetal toxicity was observed with glucagon administered by injection during the period of organogenesis at doses representing up to 100 and 200 times the human dose, respectively, based on body surface area (mg/m2) (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2%-4% and 15%-20%, respectively.
Data
Animal Data
In rats and rabbits given glucagon by injection at doses of 0.4, 2, and 10 mg/kg (up to 100 and 200 times the human dose based on mg/m2 for rats and rabbits, respectively) there was no evidence of increased malformations or embryofetal lethality.
8.2 Lactation
Risk Summary
There is no information available on the presence of glucagon in human or animal milk, the effects of glucagon on the breastfed child or the effects of glucagon on milk production. However, glucagon is a peptide and would be expected to be broken down to its constituent amino acids in the infant's digestive tract and is therefore, unlikely to cause harm to an exposed infant.
8.4 Pediatric Use
The safety and effectiveness of GlucaGen for the treatment of severe hypoglycemia in pediatric patients with diabetes have been established.
Safety and effectiveness for use as a diagnostic aid during radiologic examinations to temporarily inhibit movement of the gastrointestinal tract in pediatric patients have not been established.
10 OVERDOSAGE
If overdosage occurs, the patient may experience nausea, vomiting, inhibition of gastrointestinal tract motility, increase in blood pressure and pulse rate. In case of suspected overdosing, the serum potassium may decrease and should be monitored and corrected if needed. If the patient develops a dramatic increase in blood pressure, phentolamine mesylate has been shown to be effective in lowering blood pressure for the short time that control would be needed.
11 DESCRIPTION
Glucagon is an antihypoglycemic agent and a gastrointestinal motility inhibitor. It is produced by expression of recombinant DNA in a Saccharomyces cerevisiae vector with subsequent purification. The chemical structure of the glucagon is identical to human glucagon. Glucagon with the empirical formula of C153H225N43O49S, and a molecular weight of 3483, is a single-chain polypeptide containing 29 amino acid residues. The structure of glucagon is:
GlucaGen is a sterile, lyophilized white powder for reconstitution for subcutaneous, intramuscular or intravenous use, supplied in a 2 mL vial (appearance of the powder may vary, and occasionally the powder may appear compacted). Each vial for reconstitution contains 1 mg of glucagon, 107 mg of lactose monohydrate, hydrochloric acid and sodium hydroxide. Hydrochloric acid and/or sodium hydroxide may be used to adjust the pH before lyophilization. The reconstituted solution of GlucaGen contains glucagon 1 mg/mL at pH 2.5-3.5, and is soluble in water.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Glucagon increases blood glucose concentration by activating hepatic glucagon receptors, thereby stimulating glycogen breakdown and release of glucose from the liver. Hepatic stores of glycogen are necessary for glucagon to produce an antihypoglycemic effect. Extrahepatic effects of glucagon include relaxation of the smooth muscle of the stomach, duodenum, small bowel, and colon.
12.2 Pharmacodynamics
Treatment of Severe Hypoglycemia:
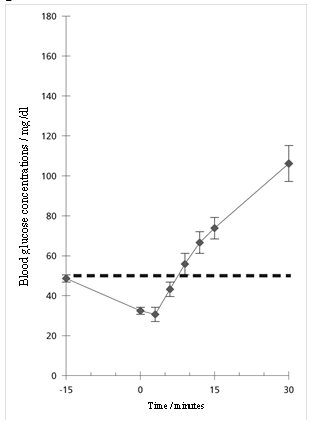
Blood glucose concentration rises within 10 minutes of injection and maximal concentrations are attained at approximately 30 minutes after injection (see Figure 1). The duration of hyperglycemic action after intravenous or intramuscular injection is 60 – 90 minutes.
Figure 1. Recovery from Insulin Induced Hypoglycemia (mean blood glucose) after Intramuscular Injection of
1 mg GlucaGen in Type 1 Diabetic Men
Diagnostic Aid:
Table 2: Pharmacodynamic Properties of Glucagon for Intravenous Route
|
Route of Administration |
Dosea |
Time of Maximal Glucose Concentration |
Time of Onset of Action for GI Smooth Muscle Relaxation |
Duration of Smooth Muscle Relaxation |
|
Intravenous |
0.25 to 0.5 mg |
5 to 20 minutes |
45 seconds |
9 to 17 minutes |
- aDose is determined based on the length of the procedure.
Table 3: Pharmacodynamic Properties of GlucaGen for Intramuscular Route
|
Route of Administration |
Dosea |
Time of Maximal Glucose Concentration |
Time of Onset of Action for GI Smooth Muscle Relaxation |
Duration of Smooth Muscle Relaxation |
|
Intramuscular |
1 mg |
30 minutes |
8 to 10 minutes |
12 to 27 minutes |
|
2 mg |
30 minutes |
4 to 7 minutes |
21 to 32 minutes |
- aDose is determined based on the length of the procedure.
The time of maximal glucose concentration for GlucaGen administered subcutaneously is 30-45 minutes.
12.3 Pharmacokinetics
Absorption
Intramuscular injection of 1 mg GlucaGen resulted in a mean Cmax (CV%) of 1686 pg/mL (43%) and median Tmax of 12.5 minutes.
Elimination
The mean apparent half-life of 45 minutes after intramuscular injection probably reflects prolonged absorption from the injection site.
Metabolism
Glucagon is degraded in the liver, kidney, and plasma.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Long term studies in animals to evaluate carcinogenic potential have not been performed.
Mutagenesis
The mutagenic potential tested in the Ames and human lymphocyte assays, was borderline positive under certain conditions for both glucagon (pancreatic) and glucagon (rDNA) origin. Doses of 100 and 200 mg/kg of glucagon of both pancreatic and recombinant origins gave slightly higher incidences of micronucleus formation in male mice but there was no effect in females. The weight of evidence indicates that synthetic and recombinant glucagon are not different and do not pose a genotoxic risk to humans.
Impairment of Fertility
Glucagon was not tested in animal fertility studies. Studies in rats have shown that pancreatic glucagon does not cause impaired fertility.
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
GlucaGen for injection is supplied as a lyophilized white powder available as follows:
|
Presentation |
NDC |
Strength |
Description |
|
Treatment of Severe Hypoglycemia |
|||
|
GlucaGen HypoKit |
0169-7065-15 |
1 mg per vial |
1 mL single-dose vial of GlucaGen with 1 mL single-dose syringe of Sterile Water for Injection, USP for reconstitution |
|
Use as a Diagnostic Aid |
|||
|
GlucaGen 10-pack: 10 Single-dose vials |
0597-0053-45 |
1 mg per vial |
1 mL single-dose vial of GlucaGen |
|
GlucaGen Diagnostic Kit |
0597-0260-10 |
1 mg per vial |
1 mL single-dose vial of GlucaGen with 1 mL single-dose vial of Sterile Water for Injection, USP for reconstitution |
16.2 Recommended Storage
Before Reconstitution:
The GlucaGen package may be stored up to 24 months at controlled room temperature 20o to 25oC (68o to 77oF) prior to reconstitution. Do not freeze. Keep in the original package to protect from light.
After Reconstitution:
Use the reconstituted GlucaGen solution immediately. Discard any unused portion [see Dosage and Administration (2)].
17 PATIENT COUNSELING INFORMATION
Recognition of Severe Hypoglycemia
Inform patient and family members or caregivers on how to recognize the signs and symptoms of severe hypoglycemia and the risks of prolonged hypoglycemia.
Serious Hypersensitivity
Inform patients that allergic reactions can occur with GlucaGen. Advise patients to seek immediate medical attention if they experience any symptoms of serious hypersensitivity reactions [see Warnings and Precautions (5.3)].
Manufactured for:
Boehringer Ingelheim Pharmaceuticals, Inc.
Ridgefield, CT 06877
By:
Novo Nordisk A/S
2880 Bagsvaerd, Denmark
GlucaGen® and HypoKit® are registered trademarks of Novo Nordisk A/S
© 1998-2021 Novo Nordisk
For information contact:
Boehringer Ingleheim Pharmaceuticals, Inc.
Ridgefield, CT 06877
1-800-243-0127
PRINCIPAL DISPLAY PANEL - GlucaGen diagnostic kit
NDC 0597-0260-10
GlucaGen®
(glucagon) for injection
1 mg per vial
For intramuscular or intravenous injection
GlucaGen® should be reconstituted
with Sterile Water for Reconstitution
immediately before use
Single dose. Discard unused portion.
Protect from Light.
See package insert for complete product
information.
FOR DIAGNOSTIC USE ONLY - NOT INTENDED FOR
USE BY PATIENTS TO TREAT SEVERE
HYPOGLYCEMIA
Rx ONLY
Boehringer
Ingelheim
PRINCIPAL DISPLAY PANEL - GlucaGen diagnostic kit - (10 Pack)
NDC 0597-0053-45
10 vials each containing 1 mg per vial
GlucaGen®
(glucagon) for injection
1 mg per vial
For intramuscular or intravenous injection
Protect from Light
FOR DIAGNOSTIC USE ONLY - NOT INTENDED FOR
USE BY PATIENTS TO TREAT SEVERE HYPOGLYCEMIA
Rx ONLY
Boehringer
Ingelheim