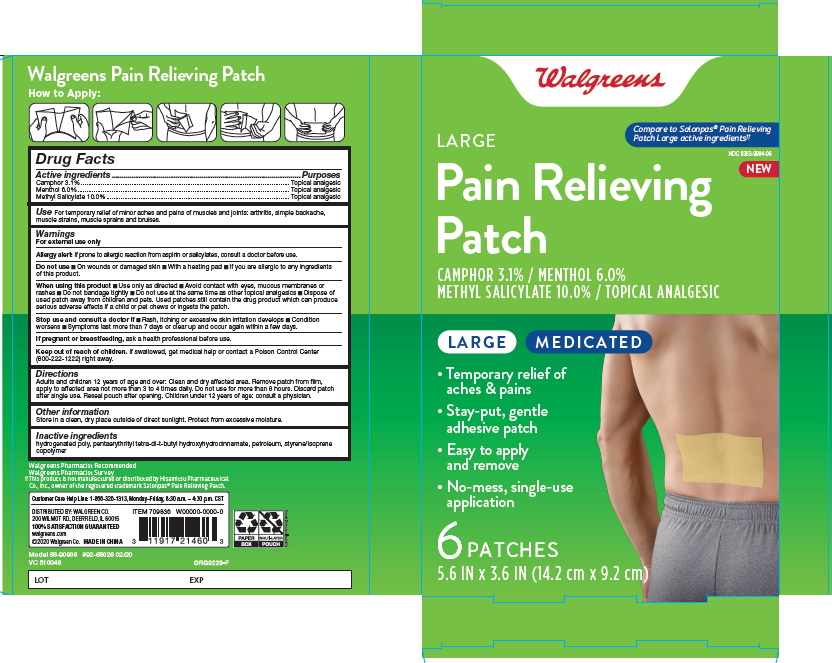
ACTIVE INGREDIENT
| Active Ingredient | Purpose |
| Camphor 3.1% | Topical Analgesic |
| Menthol 6.0% | Topical Analgesic |
| Methyl Salicylate 10.0% | Topical Analgesic |
INACTIVE INGREDIENThydrogenated poly, pentaerythrityl tetra-di-t-butyl hydroxyhydrocinnamate, petroleum, styrene/isoprene copolymer
KEEP OUT OF REACH OF CHILDREN
If swallowed, get medical help or contact a Poison Control Center (800-222-1222) right away.
INDICATIONS & USAGE
For temporary relief of minor aches and pains of muscles and joins: arthritis, simple backache, muscle strains, muscle sprains and bruises.
WARNINGS
For External Use Only
Allergy Alert:If prone to allergic reactions from aspirine or salicytates, consult a doctor before use.
DOSAGE & ADMINISTRATION
Adults and children over 12 years:
Clean and dry affected area. Remove patch from film, apply to affected area not more than 3-4 times a day.
Do not use for more than 8 hours.
Discard patch after use.
Reseal pouch after opening.
Children under 12 years of age, consult a physician.
When using this product:
■ Use only as directed ■ Avoid contact with eyes, mucous membranes or rashes ■ Do not bandage tightly ■ Do not use at the same time as other topical analgesics ■ Dispose of used patch away from children and pets. Used patches still contain the drug product which can produce serious adverse effects if a child or pet chews or ingests the patch.
Do not use
- On wounds or damaged skin
- With a heating pad
- If you are allergic to any ingredients in this product.
Stop use and consult a doctor
- If rash, itching or excessive skin irritation develops
- If condition worsens
- If symptoms last more than 7 days or clear up and occur again in a few days.