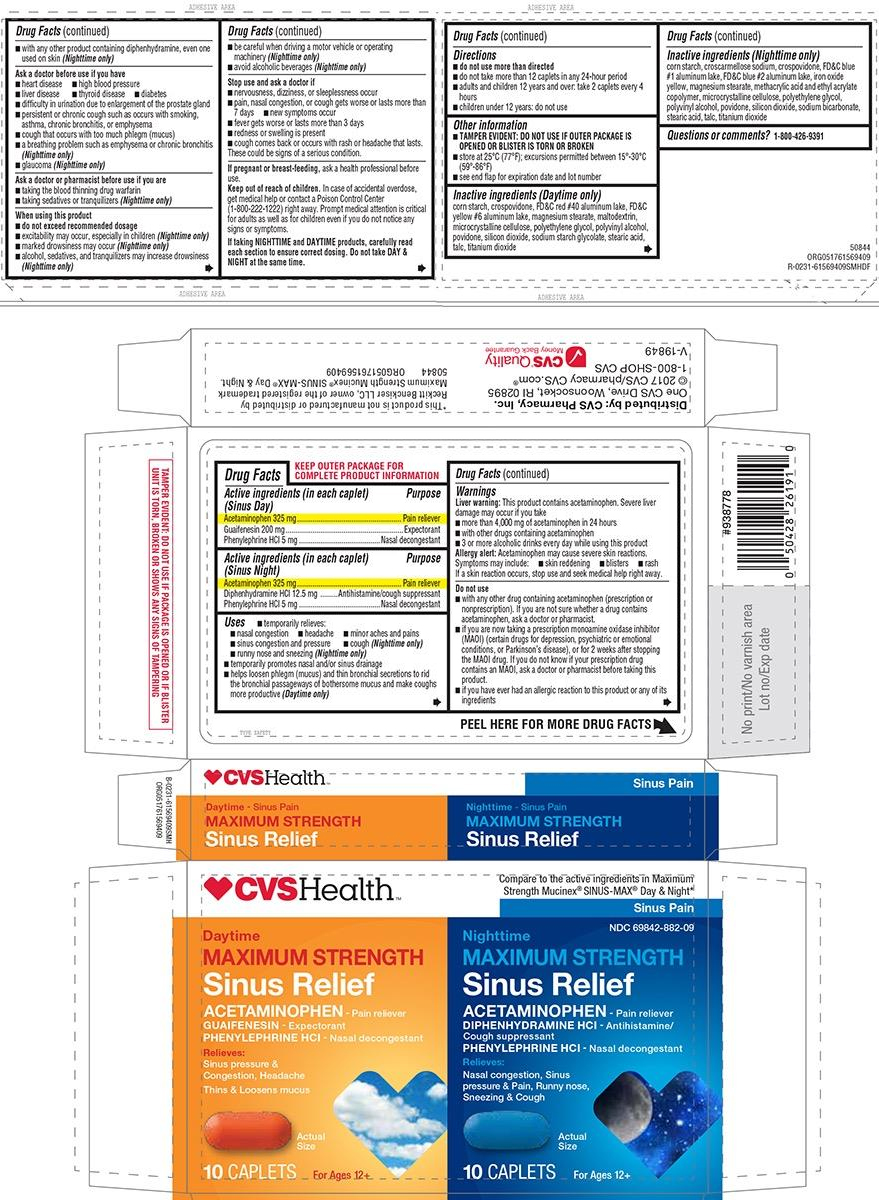
Active ingredients (in each caplet) (Sinus day)
Acetaminophen 325 mg
Guaifenesin 200 mg
Phenylephrine HCl 5 mg
Active ingredients (in each caplet) (Sinus night)
Acetaminophen 325 mg
Diphenhydramine HCl 12.5 mg
Phenylephrine HCl 5 mg
Uses
- temporarily relieves these common cold symptoms:
- nasal congestion
- headache
- minor aches and pains
- sinus congestion and pressure
- cough (Nighttime only)
- runny nose and sneezing (Nighttime only)
- temporarily promotes nasal and/or sinus drainage
- helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive (Daytime only)
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
- more than 4,000 mg of acetaminophen in 24 hours
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- if you have ever had an allergic reaction to this product or any of its ingredients
- with any other product containing diphenhydramine, even one used on skin (Nighttime only)
Ask a doctor before use if you have
- heart disease
- high blood pressure
- liver disease
- thyroid disease
- diabetes
- difficulty in urination due to enlargement of the prostate gland
- persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
- cough that occurs with too much phlegm (mucus)
- a breathing problem such as emphysema or chronic bronchitis (Nighttime only)
- glaucoma (Nighttime only)
Ask a doctor or pharmacist before use if you are
- taking the blood thinning drug warfarin
- taking sedatives or tranquilizers (Nighttime only)
When using this product
- do not exceed recommended dosage
- excitability may occur, especially in children (Nighttime only)
- marked drowsiness may occur (Nighttime only)
- alcohol, sedatives, and tranquilizers may increase drowsiness (Nighttime only)
- use caution when driving a motor vehicle or operating machinery (Nighttime only)
- avoid alcoholic beverages (Nighttime only)
Stop use and ask a doctor if
- nervousness, dizziness, or sleeplessness occur
- pain, nasal congestion, or cough gets worse or lasts more than 7 days
- new symptoms occur
- fever gets worse or lasts more than 3 days
- redness or swelling is present
- cough comes back or occurs with rash or headache that lasts.
These could be signs of a serious condition.
Directions
-
do not use more than directed
- do not take more than 12 caplets in any 24-hour period
- adults and children 12 years and over: take 2 caplets every 4 hours
- children under 12 years: do not use
Other information
- each caplet contains: sodium 3 mg (Daytime only)
- TAMPER EVIDENT: DO NOT USE IF OUTER PACKAGE IS OPENED OR BLISTER IS TORN OR BROKEN
- store at 25°C (77°F); excursions permitted between 15°-30°C (59°-86°F)
- see end flap for expiration date and lot number
Inactive ingredients (Daytime only)
corn starch, crospovidone, FD&C red #40 aluminum lake, FD&C yellow #6 aluminum lake, magnesium stearate, maltodextrin, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, povidone, silicon dioxide, sodium starch glycolate, stearic acid, talc, titanium dioxide
Inactive ingredients (Nighttime only)
corn starch, croscarmellose sodium, crospovidone, FD&C blue #1 aluminum lake, FD&C blue #2 aluminum lake, iron oxide yellow, magnesium stearate, methacrylic acid and ethyl acrylate copolymer, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, povidone, silicon dioxide, sodium bicarbonate, stearic acid, talc, titanium dioxide
Principal Display Panel
| ♥︎CVSHealth™ |
Sinus Pain NDC 69842-882-09 |
|
Daytime
ACETAMINOPHEN- Pain reliever Relieves: Actual Size 10 CAPLETS For Ages 12 + |
Nighttime
ACETAMINOPHEN- Pain reliever Relieves: Actual Size 10 CAPLETS For Ages 12 + |
TAMPER EVIDENT: DO NOT USE IF PACKAGE IS OPENED OR IF BLISTER
UNIT IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERING
50844 REV0723A61569409
Distributed by: CVS Pharmacy, Inc.
One CVS Drive, Woonsocket, RI 02895
© 2023 CVS/pharmacy CVS.com®
1-800-SHOP CVS
V-19849
✓CVS® Quality
Money Back Guarantee
CVS 615694