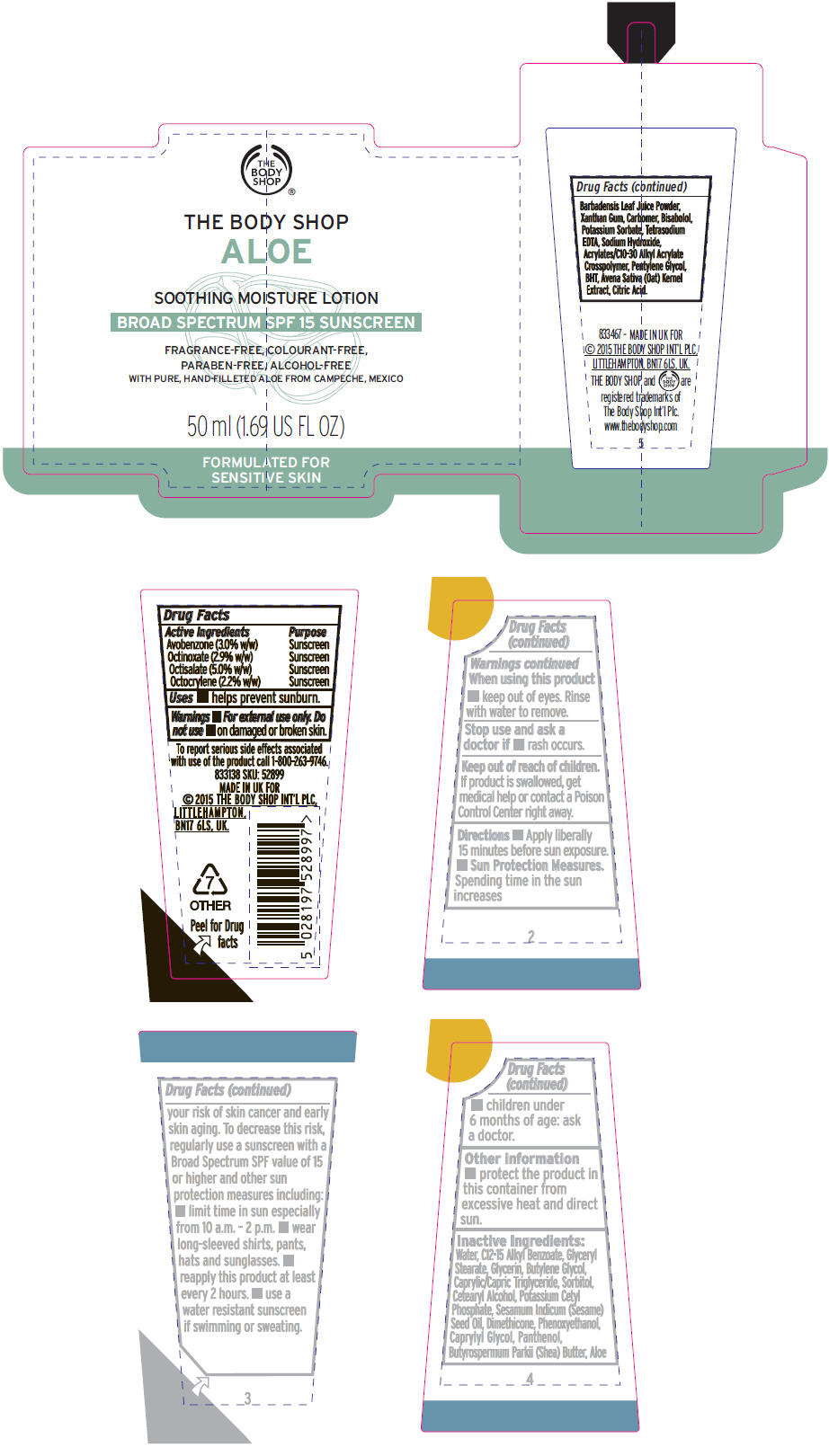
Directions
- Apply liberally 15 minutes before sun exposure.
-
Sun Protection Measures.
Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in sun especially from 10 a.m. – 2 p.m.
- wear long-sleeved shirts, pants, hats and sunglasses.
- reapply this product at least every 2 hours.
- use a water resistant sunscreen if swimming or sweating.
- children under 6 months of age: ask a doctor.
Inactive Ingredients
Water, C12-15 Alkyl Benzoate, Glyceryl Stearate, Glycerin, Butylene Glycol, Caprylic/Capric Triglyceride, Sorbitol, Cetearyl Alcohol, Potassium Cetyl Phosphate, Sesamum Indicum (Sesame) Seed Oil, Dimethicone, Phenoxyethanol, Caprylyl Glycol, Panthenol, Butyrospermum Parkii (Shea) Butter, Aloe Barbadensis Leaf Juice Powder, Xanthan Gum, Carbomer, Bisabolol, Potassium Sorbate, Tetrasodium EDTA, Sodium Hydroxide, Acrylates/C10-30 Alkyl Acrylate Crosspolymer, Pentylene Glycol, BHT, Avena Sativa (Oat) Kernel Extract, Citric Acid.