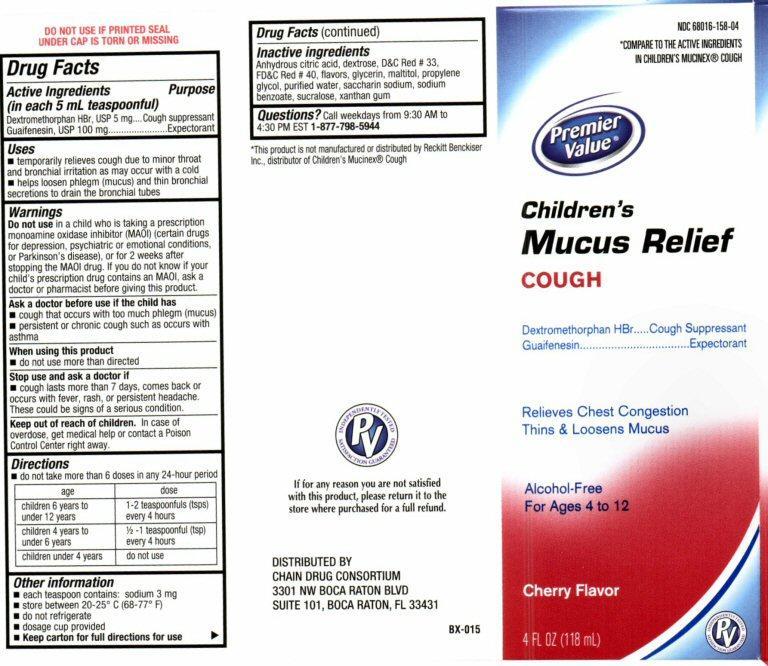
Active ingredients
Drug Facts
Active ingredients (in each 5 mL teaspoonful)
Dextromethorphan HBr 5 mg
Guaifenesin 100 mg
Keep out of reach of children
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control center right away.
Uses
- temporarily relieves cough due to minor throat and bronchial irritation as may occur with a cold
- helps to loosen phlegm (mucus) and thin bronchial secretions to drain the bronchial tubes
Warnings
Do not use
in a child who is taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for two weeks after stopping the MAOI drug. If you do not know if your child’s prescription drug contains an MAOI, ask a doctor or a pharmacist before taking this product.
Ask a doctor before use if the child has
- cough that occurs with too much phlegm (mucus)
- persistent or chronic cough such as occurs with asthma
Stop use and ask a doctor if
- cough lasts more than 7 days, comes back or occurs with fever, rash, or persistent headache.
- These could be signs of a serious condition.
Other information
each teaspoon contains: sodium 3 mg
- store between 20-25 ° C (68-77° F)
- do not refrigerate
- dosage cup provided
- Keep carton for full directions for use
Inactive ingredients
citric acid anhydrous, dextrose, D and C Red # 33, FD and C Red # 40, flavors, glycerin, maltitol, propylene glycol, purified water, saccharin sodium, sodium benzoate, sucralose, Xanthan gum
Product Label
NDC 68016-158-04
*COMPARE TO THE ACTIVE INGREDIENTS IN CHILDRENS MUCINEX® COUGH
Premier Value®
Childrens
Mucus Relief
COUGH
Dextromethorphan HBr ..... Cough Suppressant
Guaifenesin ..................... Expectorant
Relieves Chest Congestion
Thins and Loosens Mucus
Alcohol Free
For Ages 4 to 12
Cherry Flavor
4 FL OZ (118mL)
INDEPENDENTLY TESTED SATISFACTION GUARANTEED PV
DO NOT USE IF PRINTED SEAL UNDER CAP IS TORN OR MISSING
*This product is not manufactured or distributed by Reckitt Benckiser Inc. distributor of Mucinex® Cough
If for Any reason you are not satisfied with this product, please return it to the store where purchased for a full refund.
DISTRIBUTED BY
CHAIN DRUG CONSORTIUM
3301 NW BOCA RATON BLVD
SUITE 101, BOCA RATON, FL 33431
BX-015