DESCRIPTION
50% dextrose solution is sterile, non-pyrogenic for fluid and calorie replacement, and is supplied in single-dose containers for intravenous administration. Discard the unused portion. Does not contain antimicrobial agents.
Glucose (dextrose) is a monosaccharide sugar that is obtained by acid hydrolysis of corn starch. Chemically, glucose contains a pyramidal ring-5-atom of carbon and one of oxygen and in aqueous solution, there are two forms that are in equilibrium with the aldehyde form. The use can be anhydrous or with a water molecule.
The Plastic container, a semi-rigid bottle, is made of a low density polyethylene which is a flexible and resistant material that provides an excellent compatibility with a maximum number of pharmaceuticals, reducing the risk of interactions. No venting is necessary during infusion.
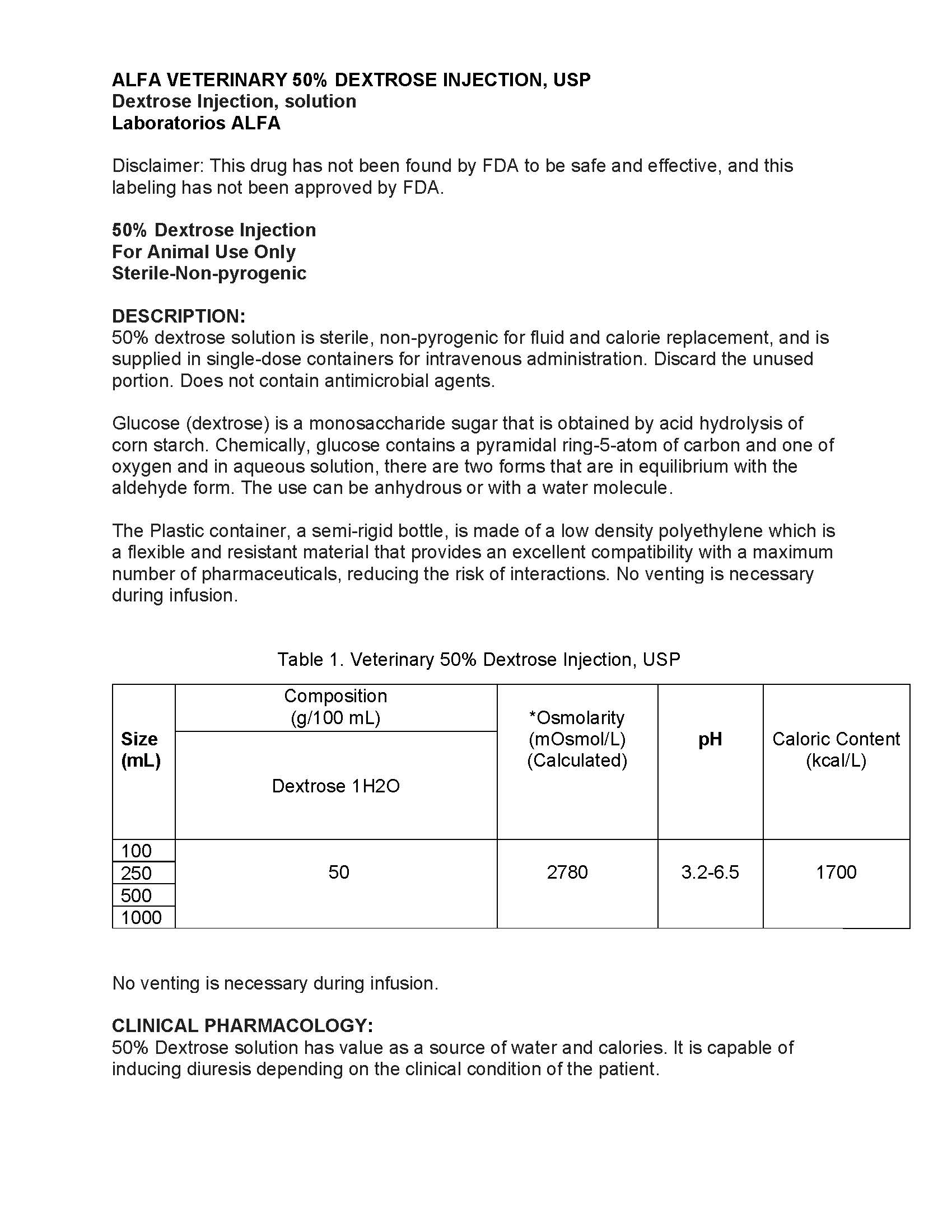
| Size (mL) | Composition (g/100 mL) | *Osmolarity (mOsmol/L) (Calculated) | pH | Caloric Content (kcal/L) |
| Dextrose 1H2 | ||||
| 100 | 50 | 2780 | 3.2-6.5 | 1700 |
| 250 | ||||
| 500 | ||||
| 1000 |
No venting is necessary during infusion.
CLINICAL PHARMACOLOGY
Veterinary 50% Dextrose Injection, USP solution has value as a source of water and calories. It is capable of inducing diuresis depending on the clinical condition of the patient.
Glucose is a nutrient of the first order, provides 4.1 Kcal per gram and like all carbohydrates has the property of decreasing protein catabolism
INDICATIONS AND USAGE
50% dextrose solution is indicated as a source of water and calories. It is used to decrease the excessive pressure of spinal brain fluid, also as sclerosing to treat varicose veins and decrease intracranial pressure
WARNING
Veterinary 50% Dextrose Injection, USP should not be administered simultaneously with blood through the same administration set because of the possibility of pseudo agglutination or hemolysis.
The intravenous administration of 5% Dextrose Injection can cause fluid and/or solute overloading resulting in dilution of serum electrolyte concentrations, over hydration, congested states, or pulmonary edema.
The risk of dilutive states is inversely proportional to the electrolyte concentrations of the injections.
The risk of solute overload causing congested states with peripheral and pulmonary edema is directly proportional to the electrolyte concentrations of the injections.
Excessive administration of dextrose injections may result in significant hypokalemia.
The container label for these injections bears the statement: Do not administer simultaneously with blood.
Keep out of the reach of children.
ADVERSE REACTIONS
Reactions which may occur because of the injection or the technique of administration include febrile response, infection at the site of injection, venous thrombosis or phlebitis extending from the site of injection, extravasation and hypervolemia.
If an adverse reaction does occur, discontinue the infusion, evaluate the patient, institute appropriate therapeutic countermeasures and save the remainder of the fluid for examination if deemed necessary.
PRECAUTIONS
Veterinary 50% Dextrose Injection, USP should be used with caution in patients with known overt or subclinical diabetes mellitus.
Clinical evaluation and periodic laboratory determinations are necessary to monitor changes in fluid balance, electrolyte concentrations and acid base balance during prolonged parenteral therapy or whenever the condition of the patient warrants such evaluation.
This is a hypotonic solution and as such should not be used for resuscitation.
Do not administer unless solution is clear and both seal and container are intact.
DOSAGE AND ADMINISTRATION
As directed by a veterinarian. Dosage is dependent upon the age, weight and clinical condition of the patient as well as laboratory determinations.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
All injections in plastic containers are intended for intravenous administration using sterile equipment.
Additives may be incompatible. Complete information is not available. Those additives known to be incompatible should not be used. Consult with veterinarian, if available. If, in the informed judgment of the veterinarian, it is deemed advisable to introduce additives, use aseptic technique. Mix thoroughly when additives have been introduced.
Do not store solutions containing additives. Discard unused portion.
OVERDOSAGE
In an event of over hydration or solute overload, re-evaluate the patient and institute appropriate corrective measures. See Warnings, Precautions, and Adverse Reactions.
PRECAUTION FOR USE OF THE BOTTLE
This is a single dose container and does not contain preservatives.
Use the solution immediately after the bottle is opened, discard the remaining one.
Squeeze and inspect the bottle, discard if leaks are found or if the solution contains visible and solid particles.
Do not administer simultaneously with blood.
Do not use it unless solution is clear, and seal is intact.
DIRECTIONS FOR USE PLASTIC CONTAINER:
This is a single dose container and does not contain preservatives. If leaks are found, discard solution as sterility may be impaired. Use the solution immediately after the bottle is opened, discard the remaining one. Discard unused portion. If supplemental medication is desired follow directions below:
Preparation and administration
1. Check for minute leaks by squeezing the container firmly. If leaks are found, discard solution as sterility may be impaired.
2. Suspend container from eyelet support.
3. Remove Plastic protector from ports area at the bottom of container.
4. Hold the bottle in vertical position and inset pyrogen free IV administration set in the outlet port. Use aseptic Technique
To add medication
WARNING: Additives may be incompatible.
To add medication before solution administration
1. Prepare medication site.
2. Using syringe with 18 to 21 gauge needle, puncture inlet port and inject.
3. Mix solution and medication thoroughly.
To add medication during solution administration
1. Close clamp on the set.
2. Prepare medication site.
3. Using syringe with 18 to 21 gauge needle, puncture inlet port and inject.
4. Remove container from IV pole and/or turn to an upright position.
5. Mix solution and medication thoroughly.
6. Return container to in use position and continue administration.
CAUTION: Federal law (USA) restricts this drug to use by or on the order of a licensed veterinarian.