FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
ADYNOVATE, Antihemophilic Factor (Recombinant), PEGylated, is a human antihemophilic factor indicated in children and adults with hemophilia A (congenital factor VIII deficiency) for:
- On-demand treatment and control of bleeding episodes
- Perioperative management
- Routine prophylaxis to reduce the frequency of bleeding episodes
Limitation of Use
ADYNOVATE is not indicated for the treatment of von Willebrand disease.
2 DOSAGE AND ADMINISTRATION
For intravenous use after reconstitution only.
2.1 Dose
- Each vial label of ADYNOVATE states the actual factor VIII potency in international units. This may be more or less than the nominal vial potency/content. One international unit corresponds to the activity of factor VIII contained in one milliliter of normal human plasma.
- Dosage and duration of treatment depend on the severity of factor VIII deficiency, the location and extent of the bleeding, and the patient's clinical condition. Careful monitoring of replacement therapy is necessary in cases of serious or life-threatening bleeding episodes.
- Potency assignment is determined using a one-stage clotting assay. Plasma factor VIII levels can be monitored clinically using a one-stage clotting assay.
- Calculate the dose of ADYNOVATE based on the empirical finding that one international unit of ADYNOVATE per kg body weight increases the plasma factor VIII level by 2 IU per dL of plasma. Use the following formula to estimate the expected in vivo peak increase in factor VIII level expressed as IU per dL (or % of normal) and the dose to achieve a desired in vivo peak increase in factor VIII level:
Estimated Increment of factor VIII (IU/dL or % of normal) = [Total Dose (IU)/body weight (kg)] × 2 (IU/dL per IU/kg)
Dose (IU) = Body Weight (kg) × Desired factor VIII Rise (IU/dL or % of Normal) × 0.5 (IU/kg per IU/dL)
- Patients vary in their pharmacokinetic (e.g., clearance, half-life, in vivo recovery) and clinical response. Base the dose and frequency of ADYNOVATE on the individual clinical response.
On-demand Treatment and Control of Bleeding Episodes
A guide for dosing of ADYNOVATE for the on-demand treatment and control of bleeding episodes is provided in Table 1. Maintain plasma factor VIII activity level at or above the described plasma levels (in IU per dL or % of normal).
| Type of Bleeding | Target Factor VIII Level (IU/dL or % of normal) | Dose*
(IU/kg) | Frequency of Dosing (hours) | Duration of Therapy |
|---|---|---|---|---|
|
||||
| Minor Early hemarthrosis, mild muscle bleeding, or mild oral bleeding episode. | 20-40 | 10-20 | 12-24 | Until the bleeding is resolved |
| Moderate Muscle bleeding, moderate bleeding into the oral cavity, definite hemarthroses, and known trauma. | 30-60 | 15-30 | 12-24 | Until the bleeding is resolved |
| Major Significant gastrointestinal bleeding, intracranial, intra-abdominal or intrathoracic bleeding, central nervous system bleeding, bleeding in the retropharyngeal or retroperitoneal spaces or iliopsoas sheath, fractures, head trauma. | 60-100 | 30-50 | 8-24 | Until bleeding is resolved. |
Perioperative Management
A guide for dosing ADYNOVATE during surgery (perioperative management) is provided in Table 2. Consideration should be given to maintain a factor VIII activity at or above the target range.
| Type of Surgery | Factor VIII Level Required (% of normal or IU/dL) | Dose (IU/kg) | Frequency of Doses (hours) | Duration of Treatment |
|---|---|---|---|---|
| Minor
Including tooth extraction | 60-100 | 30-50 | Within one hour before surgery. Repeat after 24 hours if necessary | Single dose or repeat as needed until bleeding is resolved. |
| Major
Intracranial, intra-abdominal, or intrathoracic surgery, joint replacement surgery | 80-120 (pre- and post-operative) | 40-60 | Within one hour before the operation to achieve 100% activity. Repeat every 8 to 24 hours (6 to 24 hours for patients <12 years of age) to maintain FVIII activity within the target range | Until adequate wound healing |
Routine Prophylaxis
Administer 40-50 IU/ kg body weight twice weekly in adults and adolescents (12 years and older). Administer 55 IU per kg body weight two times per week in children (< 12 years) with a maximum of 70 IU/kg. Adjust the dose and dosing intervals based on the patient's clinical response.
2.2 Preparation and Reconstitution
Preparation
- Do not remove ADYNOVATE or diluent vials from the external housing.
- Examine the packaging containing ADYNOVATE to ensure no damage or peeling of the lid is evident. Do not use if the lid is not completely sealed on the blister.
- Use aseptic technique (clean and germ free) and a flat work surface during the reconstitution procedure.
Reconstitution
- Allow the ADYNOVATE package to reach room temperature before use.
- Open the package by peeling away the lid. Remove ADYNOVATE from the package and verify that the expiration date on the label has not passed and the potency unit number is same as expected. Inspect parenteral drug products for discoloration and particulate matter. The ADYNOVATE powder should be white to off-white in color and the diluent free from foreign particles. Do not use if the criteria are not met.
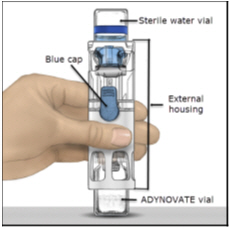
- Place the ADYNOVATE on a flat surface with the diluent vial on top (Figure A). The diluent vial has a blue stripe. Do not remove the blue cap until instructed in a later step.
Figure A 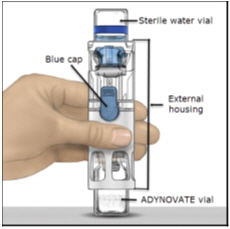
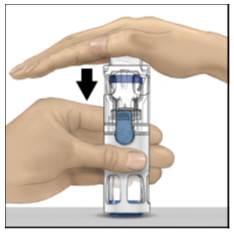
- With one hand holding the ADYNOVATE housing, press down firmly on the diluent vial with the other hand until the system is fully collapsed and the diluent flows down into the ADYNOVATE vial (Figure B). Do not tilt the system until the transfer is complete.
Figure B 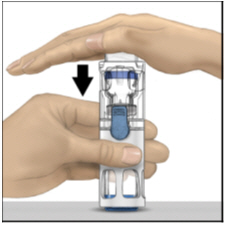
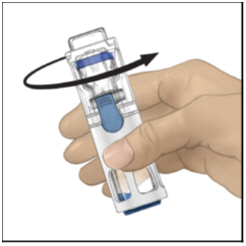
- Verify that diluent transfer is complete. Swirl gently until the powder is completely dissolved (Figure C). Do not shake. Do not refrigerate after reconstitution.
Figure C 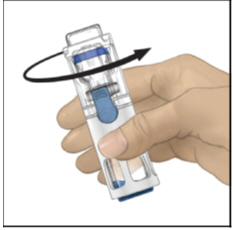
2.3 Administration
- Visually inspect the reconstituted ADYNOVATE solution for particulate matter and discoloration prior to administration, whenever solution and container permit. The final ADYNOVATE solution should be clear and colorless. Do not use if particulate matter or discoloration is observed.
- Administer ADYNOVATE as soon as possible, but no later than 3 hours after reconstitution.
Administration Steps:
- Remove the blue cap from the housing. Connect the syringe to the system (Figure D). Do not inject air into the ADYNOVATE.
Figure D 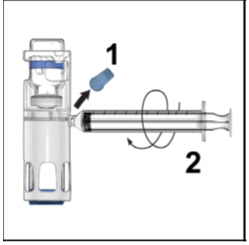
- Turn the system upside down (ADYNOVATE vial now on top). Draw the ADYNOVATE solution into the syringe by pulling the plunger back slowly (Figure E).
Figure E 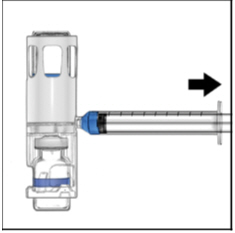
- Disconnect the syringe, attach a suitable needle, and inject intravenously as instructed. If a patient is to receive more than one ADYNOVATE -BAXJECT III system or a combination of an ADYNOVATE -BAXJECT II and an ADYNOVATE -BAXJECT III system, the contents may be drawn into the same syringe.
- Administer ADYNOVATE intravenously over a period of less than or equal to 5 minutes (maximum infusion rate 10 mL per min).
3 DOSAGE FORMS AND STRENGTHS
ADYNOVATE is a lyophilized powder in single-dose vials containing nominally (approximately) 250, 500, 750, 1000, 1500, 2000, and 3000 International Units (IU, units). The 250-1500 IU strengths come with 2 mL Sterile Water for Injection (sWFI); the 2000 and 3000 IU strengths come with 5 mL of sWFI. The actual factor VIII potency/content is labeled on each ADYNOVATE vial.
The potency assignment employs a factor VIII concentrate standard that is referenced to a WHO (World Health Organization) international standard for factor VIII concentrates and is evaluated by appropriate methodology to ensure accuracy of the results.
4 CONTRAINDICATIONS
ADYNOVATE is contraindicated in patients who have had prior anaphylactic reaction to ADYNOVATE, to the parent molecule (ADVATE), mouse or hamster protein, or excipients of ADYNOVATE (e.g. Tris, mannitol, trehalose, glutathione, and/or polysorbate 80).
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions
Hypersensitivity reactions including anaphylaxis, have been reported with ADYNOVATE. Hypersensitivity reactions that can progress to anaphylaxis may include angioedema, chest tightness, dyspnea, wheezing, urticaria, pruritus, and nausea and vomiting. Immediately discontinue administration and initiate appropriate treatment if hypersensitivity reactions occur.
5.2 Neutralizing Antibodies
Formation of neutralizing antibodies (inhibitors) to factor VIII can occur following administration of ADYNOVATE. Monitor patients regularly for the development of factor VIII inhibitors by appropriate clinical observations and laboratory tests. Perform an assay that measures factor VIII inhibitor concentration if the plasma factor VIII level fails to increase as expected, or if bleeding is not controlled with expected dose.
5.3 Monitoring Laboratory Tests
- Monitor plasma factor VIII activity by performing a validated one-stage clotting assay to confirm the adequate factor VIII levels have been achieved and maintained [see Dosage and Administration (2)].
- Monitor for the development of factor VIII inhibitors. Perform the Bethesda inhibitor assay to determine if factor VIII inhibitor is present. If expected factor VIII activity plasma levels are not attained, or if bleeding is not controlled with the expected dose of ADYNOVATE, use Bethesda Units (BU) to determine inhibitor levels.
6 ADVERSE REACTIONS
The most common adverse reactions (≥1% of subjects) reported in the clinical studies were headache, diarrhea, rash, nausea, dizziness and urticaria.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in clinical trials of another drug and may not reflect the rates observed in practice.
The safety of ADYNOVATE was evaluated in 365 previously treated patients (PTPs) and previously untreated patients (PUPs) with severe hemophilia A (factor VIII less than 1% of normal), who received at least one dose of ADYNOVATE in 6 completed multi-center, prospective, open label clinical studies and 1 ongoing clinical studies. The total number of infusions within the safety database is 74487. Table 3 lists the adverse reactions reported during clinical studies.
| MedDRA System Organ Class | MedDRA Preferred Term | Number of Subjects n (%) (N=365) |
|---|---|---|
|
||
| Gastrointestinal Disorders | Diarrhea | 25 (6.8%) |
| Nausea | 8 (2.2%) | |
| Eye Disorders | Ocular Hyperaemia | 3 (0.8%) |
| Immune System Disorder | Hypersensitivity* | 2 (0.5%) |
| Nervous System Disorders | Headache | 41 (11.2%) |
| Dizziness | 7 (1.9%) | |
| Skin and Subcutaneous Tissue Disorders | Rash | 10 (2.7%) |
| Urticaria | 7 (1.9%) | |
| Rash Pruritic | 1 (0.3%) | |
| Vascular Disorders | Flushing | 1 (0.27%) |
| Investigations | Eosinophil Count Increased | 2 (0.5%) |
| Injury, Poisoning and Procedural Complications | Infusion Related Reaction | 2 (0.5%) |
Two cases of acute pancreatitis, with no precipitating cause identified in one case, were reported in adults during an extension study of the clinical trial which evaluated 216 subjects. Administration of ADYNOVATE continued and both cases resolved.
6.2 Immunogenicity
Clinical trial subjects were monitored for neutralizing (inhibitory) antibodies to FVIII. Of the 6 completed clinical trials in previously treated patients (PTPs), in the randomized controlled trial comparing different dosing regimens of Adynovate, one previously treated patient developed a transient low titer FVIII inhibitor at 0.6 BU while receiving more frequent dosing with Adynovate. In a continuation study with Adynovate, one patient developed a transient low titer (0.6BU) FVIII inhibitor. Repeat testing did not confirm the presence of inhibitor. Both of these subjects continued treatment without change in the dose of Adynovate.
Immunogenicity also was evaluated by measuring the development of binding IgG and IgM antibodies against factor VIII, PEGylated (PEG)-factor VIII, PEG and Chinese hamster ovary (CHO) protein using validated ELISA assays. Persistent treatment-emergent binding antibodies against FVIII, PEG-FVIII or PEG were not detected. Out of 365 subjects, thirty six subjects in total showed pre-existing antibodies to factor VIII (n=5), PEG-factor VIII (n=31) and/or PEG (n=6) prior to the first exposure to ADYNOVATE. Twenty four subjects who tested negative at screening developed transient antibodies against factor VIII (n= 10), PEG-FVIII (n= 16) and/or PEG (n=3) at one or two consecutive study visits. Antibodies were transient and not detectable at subsequent visits. Two subjects showed positive results for binding antibodies at study completion or at the time of data cutoff. Binding antibodies that were detected prior to exposure to ADYNOVATE, that transiently developed during the trial or were still detectable at study completion or data cutoff could not be correlated to any impaired treatment efficacy or altered PK parameters. There was no causal relationship between observed adverse events and binding antibodies except in one subject where a causal relationship cannot be ruled out based on available data. No subject had pre-existing or treatment-emergent antibodies to CHO protein.
From an ongoing study in previously untreated patients <6 years with severe hemophilia A, 9 cases of FVIII inhibitor development associated with treatment with Adynovate were reported.
The detection of antibodies that are reactive to factor VIII is highly dependent on many factors, including: the sensitivity and specificity of the assay, sample handling, timing of sample collection, concomitant medications and underlying disease. For these reasons, comparison of the incidence of antibodies to ADYNOVATE with the incidence of antibodies to other products may be misleading.
6.3 Postmarketing Experience
Because post-marketing reporting of adverse reactions is voluntary and from a population of uncertain size, it is not always possible to reliably estimate the frequency of these reactions or establish a causal relationship to product exposure.
Immune System Disorders: Anaphylactic Reaction
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no data with ADYNOVATE use in pregnant women to inform a drug-associated risk. Animal reproduction studies have not been conducted with ADYNOVATE. It is unknown whether ADYNOVATE can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity.
In the U.S. general population, the estimated background risk of major birth defect and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
8.2 Lactation
Risk Summary
There is no information regarding the presence of ADYNOVATE in human milk, the effect on the breastfed infant, or the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for ADYNOVATE and any potential adverse effects on the breastfed infant from ADYNOVATE or from the underlying maternal condition.
8.4 Pediatric Use
Safety and efficacy studies have been performed in 91 previously treated, pediatric patients age 1 year to <18 years who received at least one dose of ADYNOVATE as part of routine prophylaxis, on-demand treatment of bleeding episodes, or perioperative management. Adolescent subjects age 12 to <18 (n=25) were enrolled in the adult and adolescent safety and efficacy trial, and subjects <12 years of age (n=66) were enrolled in a pediatric trial. The safety and efficacy of ADYNOVATE in routine prophylaxis and the treatment of bleeding episodes were comparable between children and adults. [see Clinical Studies (14)]
Pharmacokinetic studies in children (<12 years) have demonstrated higher clearance, a shorter half-life and lower incremental recovery of factor VIII compared to adults. Because clearance (based on per kg body weight) has been demonstrated to be higher in children (<12 years), dose adjustment or more frequent dosing based on per kg body weight may be needed in this population. [see Clinical Pharmacology (12.3)]
11 DESCRIPTION
ADYNOVATE, Antihemophilic Factor (Recombinant), PEGylated, is formulated as a sterile, non-pyrogenic, white to off-white lyophilized powder for reconstitution for intravenous injection. The product is supplied in single-dose vials containing nominal (approximate) potencies of 250, 500, 750, 1000, 1500, 2000, or 3000 international units (IU). Each vial of ADYNOVATE is labeled with the actual factor VIII activity in IU determined using one-stage clotting assay, using a reference material calibrated against a World Health Organization (WHO) International Standard for factor VIII concentrates. One IU, as defined by the WHO standard for blood coagulation factor VIII, human, is approximately equal to the level of factor VIII activity found in 1 mL of fresh pooled human plasma.
When reconstituted with 2 mL or 5 mL sterile water for injection, the final solution contains the following excipients and stabilizers in targeted amounts per mL of reconstituted product:
| Stabilizer and Excipient | 2 mL Reconstitution (for 250, 500, 750, 1000, 1500 IU) Target (per mL) | 5 mL Reconstitution (for 2000, 3000 IU) Target (per mL) |
|---|---|---|
| Tris (hydroxymethyl) aminomethane | 3.05 mg | 1.22 mg |
| Calcium Chloride | 0.60 mg | 0.24 mg |
| Mannitol | 80 mg | 32 mg |
| Sodium Chloride | 13.15 mg | 5.26 mg |
| Trehalose Dihydrate | 20 mg | 8 mg |
| Glutathione | 0.2 mg | 0.08 mg |
| Histidine | 3.90 mg | 1.56 mg |
| Polysorbate 80 | 0.25 mg | 0.10 mg |
ADYNOVATE contains no preservative. The specific activity of ADYNOVATE is 2700 - 8000 IU/mg protein.
ADYNOVATE is a recombinant full-length human coagulation factor VIII (2,332 amino acids with a molecular weight (MW) of 280 kDa) covalently conjugated with one or more molecules of polyethylene glycol (MW 20 kDa) [see Clinical Pharmacology (12.1)]. The therapeutic activity of ADYNOVATE is derived from its parent drug substance, ADVATE [Antihemophilic Factor (Recombinant)], which is produced by recombinant DNA technology from the CHO cell line. ADVATE is purified from the culture medium using a series of chromatography columns. The purification process includes an immunoaffinity chromatography step in which a monoclonal antibody directed against factor VIII is employed to selectively isolate the factor VIII from the medium. The production process includes a dedicated, viral inactivation solvent-detergent treatment step. The ADVATE molecule is then covalently conjugated with the polyethylene glycol, which mainly targets lysine residues.
The cell culture, pegylation, purification process and formulation used in the manufacture of ADYNOVATE do not use additives of human or animal origins.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
ADYNOVATE, a PEGylated form of recombinant antihemophilic factor (ADVATE), [see Description (11)], temporarily replaces the missing coagulation factor VIII needed for effective hemostasis in congenital hemophilia A patients. ADYNOVATE exhibits an extended terminal half-life through pegylation of the parent molecule, ADVATE, which reduces binding to the physiological factor VIII clearance receptor (LRP1).
12.2 Pharmacodynamics
Hemophilia A is a disorder characterized by a deficiency of functional coagulation factor VIII, resulting in a prolonged, patient plasma clotting time as measured by the activated partial thromboplastin time (aPTT). Treatment with ADYNOVATE normalizes the aPTT over the effective dosing period. The administration of ADYNOVATE increases plasma levels of factor VIII and can temporarily correct the coagulation defect in hemophilia A patients.
12.3 Pharmacokinetics
The pharmacokinetics (PK) of ADYNOVATE were evaluated in a multi-center, prospective, open label clinical trial and compared with ADVATE in 26 subjects prior to initiation of prophylactic treatment with ADYNOVATE and in 22 subjects after 6 months of treatment with ADYNOVATE. A single dose of 45 IU/kg was utilized for both products. The PK parameters, as shown in Table 4, were based on plasma coagulation factor VIII activity measured by the one-stage clotting assay and are presented by age groups.
Incremental recovery was comparable between both products. The PK parameters determined after 6 months of prophylactic treatment with ADYNOVATE were consistent with the initial parameter estimates.
Pediatric Pharmacokinetics
Pharmacokinetic parameters calculated from 39 subjects <18 years of age (intent-to-treat analysis) are available for 14 children (2 to <6 years), 17 older children (6 to <12 years) and 8 adolescent subjects (12 to <18 years of age), as shown in Table 4. The mean clearance (based on body weight) of ADYNOVATE was higher and the mean half-life was lower in children <12 years of age than adults. A dose adjustment may be required in children <12 years of age.
| PK Parameters | Pediatric Population PK with Sparse Sampling* | Adult and Adolescent Individual PK with Full Sampling† |
||
|---|---|---|---|---|
| <6 years N=14 | 6 to <12 years N=17 | 12 to <18 years N = 8 | ≥18 years N = 18 |
|
| Abbreviations: MRT: mean residence time; CL: clearance; CI: confidence interval; AUC: area under the curve; Vss: body weight adjusted volume of distribution at steady-state; Cmax: maximum observed activity; Tmax: time to reach the maximum concentration. | ||||
|
||||
| Terminal half-life [h] | 11.8 ± 2.43 | 12.4 ± 1.67 | 13.43 ± 4.05 | 14.69 ± 3.79 |
| MRT [h] | 17.0 ± 3.50 | 17.8 ± 2.42 | 17.96 ± 5.49 | 20.27 ± 5.23 |
| CL [mL/(kg∙h)] | 3.53 ± 1.29 | 3.11 ± 0.76 | 3.87 ± 3.31 (2.73 ± 0.93)‡ | 2.27 ± 0.84 |
| Incremental Recovery [(IU/dL)/(IU/kg)] | 1.89 ± 0.49 | 1.95 ± 0.47 | 2.12 ± 0.60 | 2.66 ± 0.68 |
| AUC0-Inf [IU∙h/dL] | 1947 ± 757 | 2012 ± 495 | 1642 ± 752 | 2264 ± 729 |
| Vss [dL/kg] | 0.56 ± 0.12 | 0.54 ± 0.09 | 0.56 ± 0.18 | 0.43 ± 0.11 |
| Cmax [IU/dL] | 115 ± 30 | 115 ± 33 | 95 ± 25 | 122 ± 29 |
| Tmax [h] | -§ | -§ | 0.26 ± 0.10 | 0.46 ± 0.29 |
14 CLINICAL STUDIES
Original Safety and Efficacy Clinical Trial
The safety, efficacy, and PK of ADYNOVATE were evaluated in a multicenter, open-label, prospective, non-randomized, two-arm clinical trial that compared the efficacy of a twice weekly prophylactic treatment regimen to on-demand treatment and determined hemostatic efficacy in the treatment of bleeding episodes. A total of 137 male PTPs (12 to 65 years of age) with severe hemophilia A received at least one infusion with ADYNOVATE. Twenty-five of the 137 subjects were adolescents (12 to less than 18 years of age).
Subjects received either prophylactic treatment (n = 120) with ADYNOVATE at a dose of 40-50 IU per kg twice weekly or on-demand treatment (n = 17) with ADYNOVATE at a dose of 10-60 IU per kg for a 6-month period. The mean (SD) dose per prophylaxis infusion was 44.4 (3.9) IU per kg with a median dosing interval of 3.6 days. There were 91 out of 98 (93%) subjects previously treated prophylactically prior to enrollment, who experienced a reduction in dosing frequency during routine prophylaxis in the trial, with a median reduction of 33.7% (approximately one more day between doses). One hundred eighteen of 120 (98%) prophylaxis subjects remained on the starting recommended regimen without dose adjustment, and 2 subjects increased their dose to 60 IU/kg during prophylaxis due to bleeding in target joints.
On-demand Treatment and Control of Bleeding Episodes
A total of 518 bleeding episodes were treated with ADYNOVATE in the per-protocol population, i.e. dosed according to the protocol specific dosing requirements. Of these, 361 bleeding episodes (n=17 subjects) occurred in the on-demand arm and 157 (n=61 subjects) occurred in the prophylaxis arm. The median dose per infusion to treat all bleeding episodes in the per-protocol population was 29 (Q1: 20.0; Q3: 39.2) IU per kg. The median dose per infusion to treat a minor, moderate, or severe/major bleeding episode in the per-protocol population was 25.5 (Q1: 16.9; Q3: 37.6) IU/kg, 30.9 (Q1: 23.0; Q3: 43.1) IU/kg, or 36.4 (Q1: 29.0; Q3: 44.5) IU/kg, respectively.
A total of 591 bleeding episodes were treated with ADYNOVATE in the treated population, which was identical to the safety analysis set of subjects assigned to routine prophylaxis or on-demand treatment with ADYNOVATE and who received at least one dose of the product. Of these, 361 bleeding episodes (n=17 subjects) occurred in the on-demand arm and 230 bleeding episodes (n=75 subjects) occurred in the routine prophylaxis arm. Efficacy in control of bleeding episodes is summarized in Table 5.
| Bleeding Episode Etiology | All | Joint | Non-joint | |
|---|---|---|---|---|
|
||||
| Number of bleeds treated | 591 | 455 | 136 | |
| Number of infusions to treat bleeding episodes | 1 infusions: | 85.4% | 85.9% | 83.8% |
| 2 infusions: | 10.8% | 10.8% | 11.0% | |
| Total (1 or 2 infusions): | 96.2% | 96.7% | 94.8% | |
| Rate of success to treat bleeding episodes* | Excellent or good | 95.3% | 95.8% | 93.4% |
Routine Prophylaxis
A total of 120 subjects (treated population) received a twice a week regimen in the prophylaxis arm, and an additional 17 subjects were treated episodically in the on-demand arm. In the treated population, the median [mean] annualized bleed rate (ABR) in the on-demand treatment arm was 41.5 [40.8] compared to 1.9 [4.7] while on a twice a week prophylaxis regimen (Table 6). In the per-protocol population, the median [mean] annualized bleed rate (ABR) in the on-demand treatment arm was 41.5 [40.8] compared to 1.9 [3.7] while on a twice a week prophylaxis regimen. Using a negative binomial model to estimate the ABR, there was a significant reduction in the ABR (p <0.0001) for subjects in the prophylaxis arm compared to the on-demand arm.
| Bleeding Episode Etiology | On-Demand Treatment | Routine Prophylaxis Treatment | ||
|---|---|---|---|---|
| Median | Mean (SD) | Median | Mean (SD) | |
| Overall | 41.5 | 40.8 (16.3) | 1.9 | 4.7 (8.6) |
| Joint | 38.1 | 34.7 (15.1) | 0.0 | 2.9 (8.0) |
| Non-Joint | 3.7 | 6.1 (6.7) | 0.0 | 1.8 (3.0) |
| Spontaneous | 21.6 | 26.0 (19.6) | 0.0 | 2.9 (7.1) |
| Traumatic | 9.3 | 14.9 (15.3) | 0.0 | 1.8 (3.1) |
In the treated population, the median [mean] ABR for the 23 adolescent subjects age 12 to <18 years of age on routine prophylaxis was 2.1 [5.2] compared to a median [mean] ABR of 1.9 [4.6] for the 97 subjects 18 years and older. Reduction in ABR between the treatment arms was observed regardless of baseline subgroups examined, including age, presence or absence of target joints, and pre-trial treatment regimen. The majority of the bleeding episodes during prophylaxis (95%) were of minor/moderate severity. Forty-five out of 120 subjects (38%) experienced no bleeding episodes and 68 out of 120 subjects (57%) experienced no joint bleeding episodes in the prophylaxis arm. Of those subjects who were compliant to regimen (per-protocol population), 40 out of 101 subjects (40%) experienced no bleeding episodes. All subjects in the on-demand arm experienced a bleeding episode, including a joint bleeding episode.
Routine Prophylaxis Clinical Trial in Pediatric Subjects (<12 years of age)
The safety and efficacy of ADYNOVATE was evaluated in a total of 73 pediatric PTPs with severe hemophilia A, of which 66 subjects were dosed (32 subjects aged <6 years and 34 subjects aged 6 to <12 years) in a separate pediatric clinical trial. The prophylactic regimen was 40 to 60 IU/kg of ADYNOVATE twice a week, with a mean (SD) dose of 51.1 IU/kg (5.5). The median [mean] overall ABR was 2.0 [3.61] for the 66 subjects in the treated population and the median [mean] ABRs for spontaneous and joint bleeding episodes were both 0 [1.18 and 1.12, respectively]. Of the 66 subjects treated prophylactically, 25 (38%) experienced no bleeding episodes, 44 (67%) experienced no spontaneous bleeding episodes, and 48 (73%) experienced no joint bleeding episodes.
Of the 70 bleeding episodes observed during the pediatric trial, 82.9% were controlled with 1 infusion and 91.4% were controlled with 1 or 2 infusions. Control of bleeding was rated excellent or good in 63 out of 70 (90%) bleeding episodes. The definitions of excellent or good in the pediatric clinical trial were unchanged as compared to the previously conducted prophylaxis clinical trial in adolescent and adult subjects.
An extension study in adult and pediatric patients evaluated the safety and efficacy of prophylactic treatment regimen in 216 previously treated patients with severe hemophilia A. Majority had completed the adult and adolescent study or the pediatric study. Similar efficacy was noted in this extension study.
Perioperative Management Clinical Trial
Twenty-one major surgical procedures comprised of 14 orthopedic, and 7 non-orthopedic procedures, and 5 additional minor surgeries were performed in 21 subjects. The preoperative loading dose ranged from 36 IU/kg to 99 IU/kg (median: 60 IU/kg) and the total postoperative dose ranged from 23 IU/kg to 769 IU/kg (median: 183 IU/kg). The median total dose (including all administrations from pre-surgical PK and loading doses to post-hospital follow up) was 629 IU/kg (range: 464 – 1,457 IU/kg) for major orthopedic surgeries, 489 IU/kg (range: 296 – 738 IU/kg) for major non-orthopedic surgeries.
Overall hemostatic efficacy was rated as excellent (blood loss less than or equal to that expected for the same type of procedure performed in a non-hemophilic patient, for all 24 (21 major, 3 minor) procedures with available assessments.
16 HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
ADYNOVATE in a BAXJECT III system is packaged with 2 mL or 5 mL of Sterile Water for Injection, one Terumo Microbore Infusion set (2 mL only), one full prescribing physician insert and one patient insert. Components not made with natural rubber latex.
ADYNOVATE is available in single-dose vials that contain the following nominal product strengths:
| Nominal Strength | Potency Color Code | Carton NDC (Includes 2 mL sWFI Diluent) | Carton NDC (Includes 5 mL sWFI Diluent) |
|---|---|---|---|
| 250 IU | Light Blue | 0944-4622-01 | |
| 500 IU | Pink | 0944-4623-01 | |
| 750 IU | Red | 0944-4626-01 | |
| 1000 IU | Light Green | 0944-4624-01 | |
| 1500 IU | Purple | 0944-4627-01 | |
| 2000 IU | Orange | 0944-4625-01 | |
| 3000 IU | Silver | 0944-4628-01 |
Actual factor VIII activity in IU is stated on the label of each ADYNOVATE carton and housing.
Storage and Handling
- Store ADYNOVATE in powder form at 2°C to 8°C (36°F to 46°F).
- Do not freeze.
- ADYNOVATE may be stored at room temperature not to exceed 30°C (86°F) for a period of up to 3 months not to exceed the expiration date. If stored at room temperature, write the date on the carton when ADYNOVATE is removed from refrigeration.
- After storage at room temperature, do not return the product to the refrigerator.
- Do not use beyond expiration date printed on the carton or housing.
- Store ADYNOVATE in the original box and protect from extreme exposure to light.
17 PATIENT COUNSELING INFORMATION
Advise the patients to:
- Read the FDA-approved patient labeling (Patient Information and Instructions for Use).
- Call their healthcare provider or go to the emergency department right away if a hypersensitivity reaction occurs. Early signs of hypersensitivity reactions may include rash, hives, itching, facial swelling, tightness of the chest, and wheezing. Advise patients to discontinue use of the product if these symptoms occur and seek immediate emergency treatment.
- Contact their healthcare provider or treatment facility for further treatment and/or assessment if they experience a lack of a clinical response to factor VIII therapy because this may be a sign of inhibitor development.
- Consult with their physicians or healthcare provider prior to travel. While traveling, advise patients to bring an adequate supply of ADYNOVATE based on their current regimen of treatment.
To enroll in the confidential, industry-wide Patient Notification System, call 1-888-873-2838.
Takeda Pharmaceuticals U.S.A., Inc.
Lexington, MA 02421
U.S. License No. 1898
ADYNOVATE®, ADVATE® and BAXJECT® are registered trademarks of Baxalta Incorporated.
ADYNOVATE® is a registered trademark of Baxalta Incorporated.
TAKEDA® and the TAKEDA Logo® are registered trademarks of Takeda Pharmaceutical Company Limited.
Patented: see www.takeda.com/en-us/patents
FDA-Approved Patient Labeling
Patient Information
ADYNOVATE
[Antihemophilic Factor (Recombinant), PEGylated]
This leaflet summarizes important information about ADYNOVATE. Please read it carefully before using this medicine. This information does not take the place of talking with your healthcare provider, and it does not include all of the important information about ADYNOVATE. If you have any questions after reading this, ask your healthcare provider.
What is the most important information I need to know about ADYNOVATE?
Do not attempt to do an infusion to yourself unless you have been taught how by your healthcare provider or hemophilia center.
You must carefully follow your healthcare provider's instructions regarding the dose and schedule for infusing ADYNOVATE so that your treatment will work best for you.
What is ADYNOVATE?
ADYNOVATE is an injectable medicine that is used to help treat and control bleeding in children and adults with hemophilia A (congenital Factor VIII deficiency). Your healthcare provider may give you ADYNOVATE when you have surgery.
ADYNOVATE can reduce the number of bleeding episodes when used regularly (prophylaxis).
ADYNOVATE is not used to treat von Willebrand disease.
Who should not use ADYNOVATE?
You should not use ADYNOVATE if you:
- Are allergic to mice or hamster protein
- Are allergic to any ingredients in ADYNOVATE or ADVATE
Tell your healthcare provider if you are pregnant or breastfeeding because ADYNOVATE may not be right for you.
How should I use ADYNOVATE?
ADYNOVATE is given directly into the bloodstream.
You may infuse ADYNOVATE at a hemophilia treatment center, at your healthcare provider's office or in your home. You should be trained on how to do infusions by your healthcare provider or hemophilia treatment center. Many people with hemophilia A learn to infuse their ADYNOVATE by themselves or with the help of a family member.
Your healthcare provider will tell you how much ADYNOVATE to use based on your individual weight, level of physical activity, the severity of your hemophilia A, and where you are bleeding.
Reconstituted product (after mixing dry product with wet diluent) must be used within 3 hours and cannot be stored or refrigerated. Discard any ADYNOVATE left in the vial at the end of your infusion as directed by your healthcare professional.
You may have to have blood tests done after getting ADYNOVATE to be sure that your blood level of factor VIII is high enough to clot your blood.
Call your healthcare provider right away if your bleeding does not stop after taking ADYNOVATE.
What should I tell my healthcare provider before I use ADYNOVATE?
You should tell your healthcare provider if you:
- Have or have had any medical problems.
- Take any medicines, including prescription and non-prescription medicines, such as over-the-counter medicines, supplements or herbal remedies.
- Have any allergies, including allergies to mice or hamsters.
- Are breastfeeding. It is not known if ADYNOVATE passes into your milk and if it can harm your baby.
- Are pregnant or planning to become pregnant. It is not known if ADYNOVATE may harm your unborn baby.
- Have been told that you have inhibitors to factor VIII (because ADYNOVATE may not work for you).
What are the possible side effects of ADYNOVATE?
You can have an allergic reaction to ADYNOVATE.
Call your healthcare provider right away and stop treatment if you get a rash or hives, itching, tightness of the throat, chest pain or tightness, difficulty breathing, lightheadedness, dizziness, nausea or fainting.
The common side effects of ADYNOVATE are headache, diarrhea, rash, nausea dizziness and hives. Tell your healthcare provider about any side effects that bother you or do not go away.
These are not all the possible side effects with ADYNOVATE. You can ask your healthcare provider for information that is written for healthcare professionals.
What are the ADYNOVATE dosage strengths?
ADYNOVATE with 2 mL or 5 mL Sterile Water for Injection in a BAXJECT III system comes in seven different dosage strengths: 250 International Units (IU), 500 IU, 750 IU, 1000 IU, 1500 IU, 2000 IU, and 3000 IU. The actual strength will be imprinted on the label and on the box. The seven different strengths are color coded, as follows:
|
| Dosage strength of approximately 250 International Units per vial (with 2 mL sWFI) |
|
| Dosage strength of approximately 500 International Units per vial (with 2 mL sWFI) |
|
| Dosage strength of approximately 750 International Units per vial (with 2 mL sWFI) |
|
| Dosage strength of approximately 1000 International Units per vial (with 2 mL sWFI) |
|
| Dosage strength of approximately 1500 International Units per vial (with 2 mL sWFI) |
|
| Dosage strength of approximately 2000 International Units per vial (with 5 mL sterile Water For Injection) |
|
| Dosage strength of approximately 3000 International Units per vial (with 5 mL sterile Water For Injection) |
Always check the actual dosage strength printed on the label to make sure you are using the strength prescribed by your healthcare provider. Always check the expiration date printed on the box. Do not use the product after the expiration date printed on the box.
How do I store ADYNOVATE?
- Do not freeze.
- Store at refrigerated temperature 2°C to 8°C (36°F to 46°F).
- May store at room temperature not to exceed 30°C (86°F) for up to 3 months.
- Write the date on the carton when ADYNOVATE is removed from refrigeration.
- After storage at room temperature, do not return product back to the refrigerator.
- Do not use beyond the expiration date printed on the carton or vial.
- Store ADYNOVATE in the original box and protect from extreme exposure to light.
What else should I know about ADYNOVATE and Hemophilia A?
Your body may form inhibitors to Factor VIII. An inhibitor is part of the body's normal defense system. If you form inhibitors, it may stop ADYNOVATE from working properly. Consult with your healthcare provider to make sure you are carefully monitored with blood tests for the development of inhibitors to Factor VIII.
Medicines are sometimes prescribed for purposes other than those listed here. Do not use ADYNOVATE for a condition for which it is not prescribed. Do not share ADYNOVATE with other people, even if they have the same symptoms that you have.
Takeda Pharmaceuticals U.S.A., Inc.
Lexington, MA 02421
U.S. License No. 1898
ADYNOVATE®, ADVATE® and BAXJECT® are registered trademarks of Baxalta Incorporated.
ADYNOVATE® is a registered trademark of Baxalta Incorporated.
TAKEDA® and the TAKEDA Logo® are registered trademarks of Takeda Pharmaceutical Company Limited.
Patented: see www.takeda.com/en-us/patents
Revised: 3/2023
FDA-Approved Patient Labeling
Instructions for Use
ADYNOVATE
[Antihemophilic Factor (Recombinant),
PEGylated]
(For intravenous use only)
Do not attempt to do an infusion to yourself unless you have been taught how by your healthcare provider or hemophilia center.
Step-by-step instructions for reconstituting ADYNOVATE are found at the end of this leaflet.
Always follow the specific instructions given by your healthcare provider. The steps listed below are general guidelines for using ADYNOVATE. If you are unsure of the procedures, please call your healthcare provider before using.
Call your healthcare provider right away if bleeding is not controlled after using ADYNOVATE.
Your healthcare provider will prescribe the dose that you should take.
Reconstituted product (after mixing dry product with wet diluent) must be used within 3 hours and cannot be stored or refrigerated.
Your healthcare provider may need to take blood tests from time to time.
Talk to your healthcare provider before traveling. Plan to bring enough ADYNOVATE for your treatment during this time.
Dispose of all materials, including any leftover reconstituted ADYNOVATE product, in an appropriate container.
- Prepare a clean flat surface and gather all the materials you will need for the infusion.
- Check the expiration date, and let the ADYNOVATE warm up to room temperature.
- Wash your hands and put on clean exam gloves. If infusing yourself at home, the use of gloves is optional.
- Open the ADYNOVATE package by peeling away the lid. Remove the ADYNOVATE from the package and visually inspect the contents of the product and diluent vial. The ADYNOVATE powder should be white to off-white in color and the diluent should not contain particles. Do not use if discoloration or particles are seen.
- Place on a flat surface with the diluent vial on top. The diluent vial has a blue stripe.
- With one hand holding the ADYNOVATE housing, press down firmly on the diluent vial with the other hand until the system is fully collapsed and the diluent flows down into the ADYNOVATE vial. Both vials will move into the housing when pressed. If you don't see the diluent transfer to the product vial, press the vials again to assure they are completely inserted. Do not remove the blue cap until instructed in a later step.
- Swirl the ADYNOVATE gently and continuously until the ADYNOVATE is completely dissolved. Do not shake. Do not refrigerate after reconstitution. Inspect the ADYNOVATE solution for particulate matter and discoloration prior to administration. The solution should be clear and colorless in appearance. If not, do not use the solution and notify your healthcare provider immediately.
- Take off the blue cap from the housing and connect the syringe. Be careful to not inject air into the ADYNOVATE.
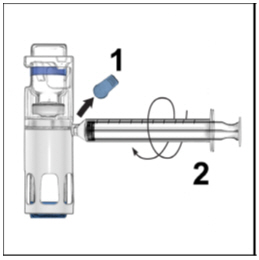
- Turn over the ADYNOVATE so that the vial containing the ADYNOVATE solution is on top. Draw the ADYNOVATE solution into the syringe by pulling back the plunger slowly. If the solution does not draw into the syringe, be sure that both vials are pressed firmly together. The contents of more than one vial may be drawn into a single, appropriately sized syringe if you are using more than one vial of ADYNOVATE.
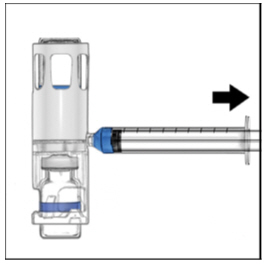
- Disconnect the syringe from the system. Attach the infusion needle to the syringe using a winged (butterfly) infusion set, if available. Point the needle up and remove any air bubbles by gently tapping the syringe with your finger and slowly and carefully pushing air out of the syringe and needle.
- Apply a tourniquet and get the injection site ready by wiping the skin well with an alcohol swab (or other suitable solution suggested by your healthcare provider or hemophilia center).
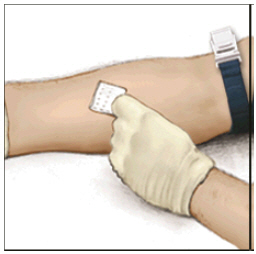
- Insert the needle into the vein and remove the tourniquet. Slowly infuse the ADYNOVATE. Do not infuse any faster than 10 mL per minute.
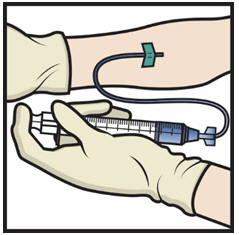
- Take the needle out of the vein and use sterile gauze to put pressure on the infusion site for several minutes.
- Remove the peel-off label from blister lid and place it in your logbook. Clean any spilled blood with a freshly prepared mixture of 1 part bleach and 9 parts water, soap and water, or any household disinfecting solution.
- Do not recap the needle. Place needle, syringe and ADYNOVATE system in a hard-walled Sharps container for proper disposal. Do not dispose of these supplies in ordinary household trash.
Important: Contact your healthcare provider or local hemophilia treatment center if you experience any problems.
Takeda Pharmaceuticals U.S.A., Inc.
Lexington, MA 02421
U.S. License No. 1898
ADYNOVATE®, ADVATE® and BAXJECT® are registered trademarks of Baxalta Incorporated.
ADYNOVATE® is a registered trademark of Baxalta Incorporated.
TAKEDA® and the TAKEDA Logo® are registered trademarks of Takeda Pharmaceutical Company Limited.
Patented: see www.takeda.com/en-us/patents
Revised: 3/2023
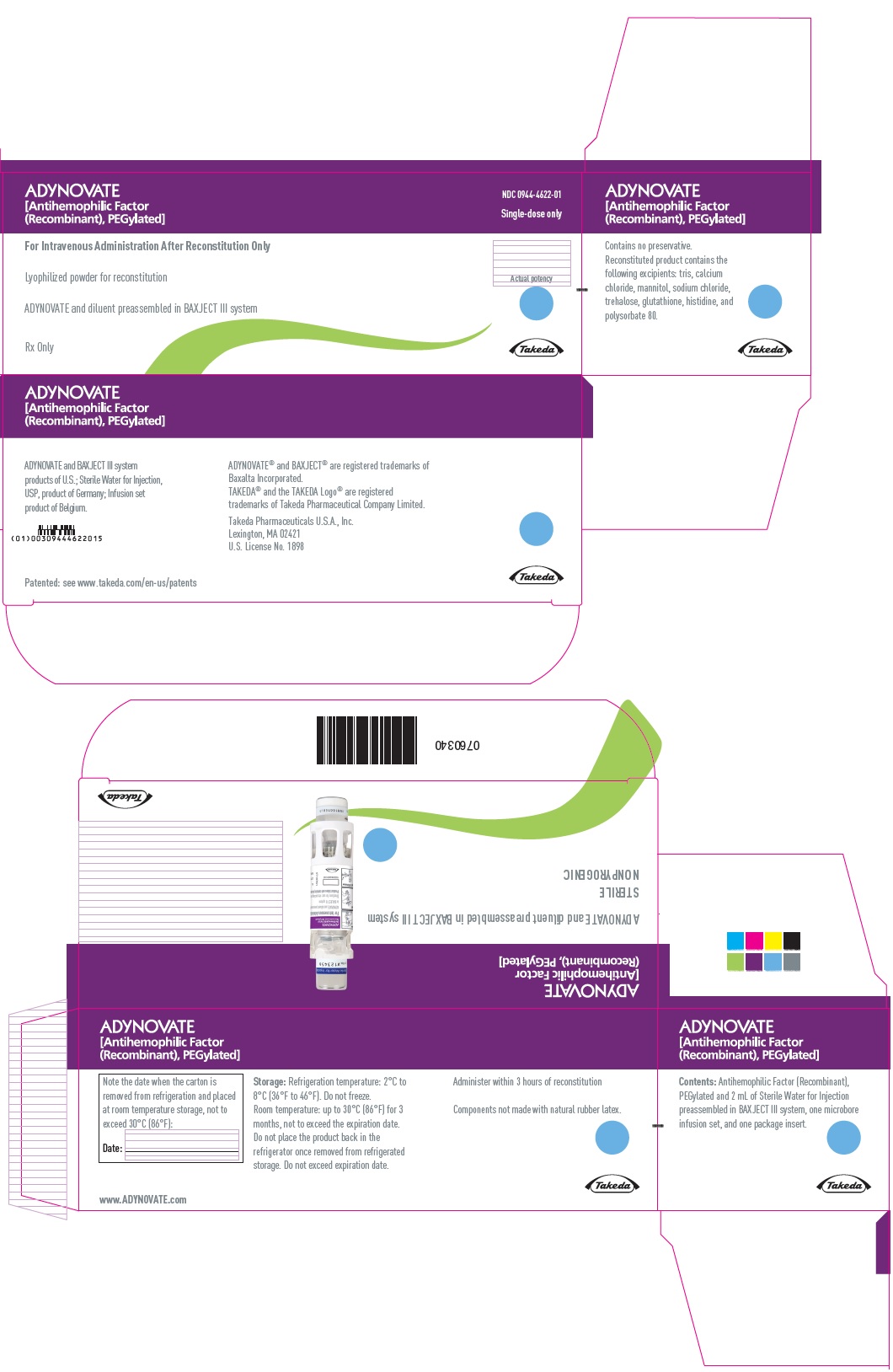
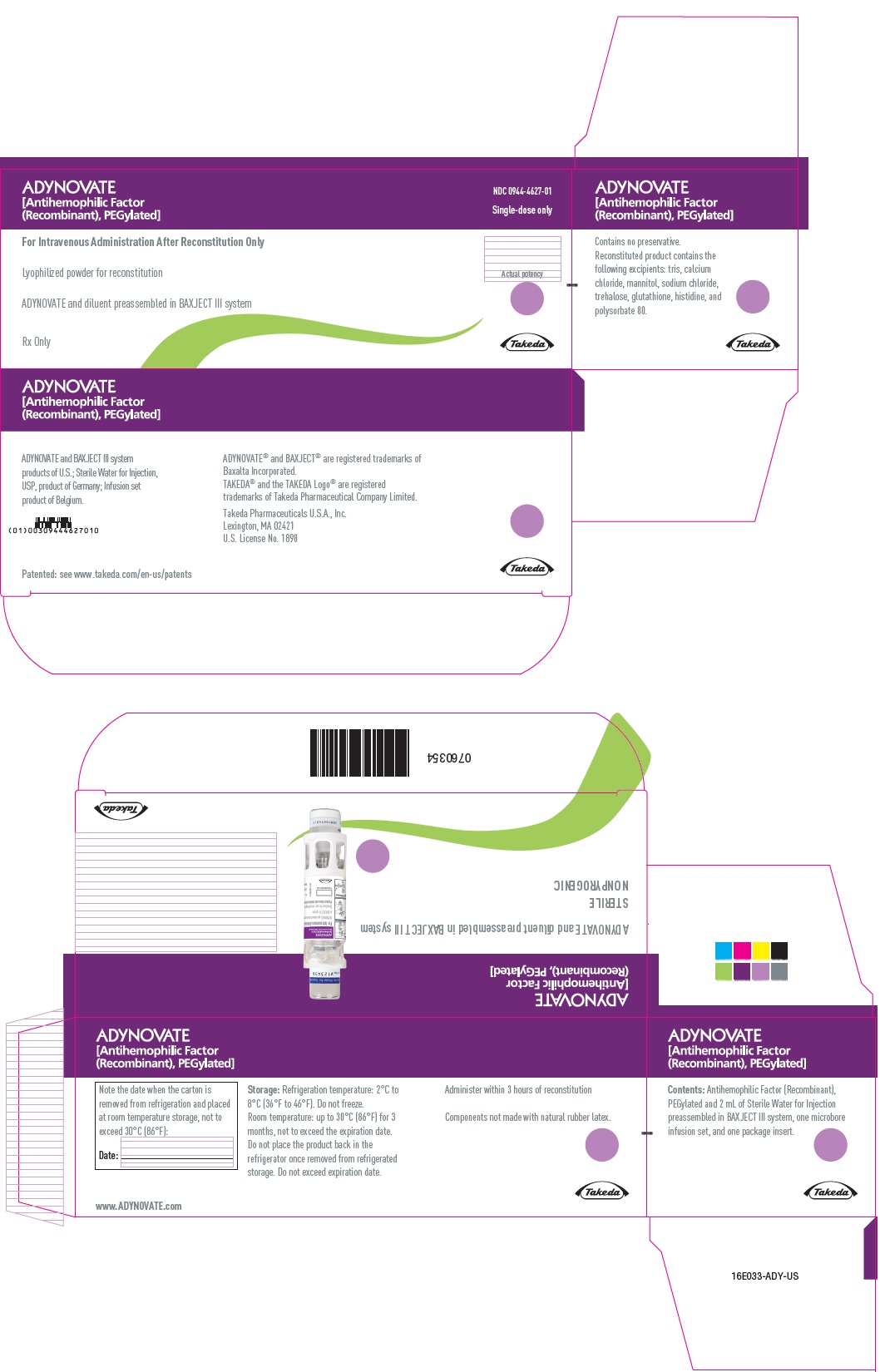
PRINCIPAL DISPLAY PANEL - Kit Carton - 250 IU
ADYNOVATE
[Antihemophilic Factor
(Recombinant), PEGylated]
NDC 0944-4622-01
Single-dose only
For Intravenous Administration After Reconstitution Only
Lyophilized powder for reconstitution
ADYNOVATE and diluent preassembled in BAXJECT III system
Rx Only
Actual potency
Takeda
PRINCIPAL DISPLAY PANEL - Barrel Label - 250 IU
ADYNOVATE
[Antihemophilic Factor
(Recombinant), PEGylated]
For Intravenous Administration Only
ADYNOVATE and diluent preassembled
in BAXJECT III system
Directions for use: see package insert
Rx Only
IU:
LOT:
EXP:
Takeda
0754163

PRINCIPAL DISPLAY PANEL - Blister Label - 250 IU
ADYNOVATE
[Antihemophilic Factor
(Recombinant), PEGylated]
NDC 0944-4622-02
Single-dose only
0760339
For Intravenous Administration Only
ADYNOVATE and diluent preassembled in BAXJECT III system
Do not open until time of use
Do not use if packaging is opened or damaged
Rx Only
Takeda

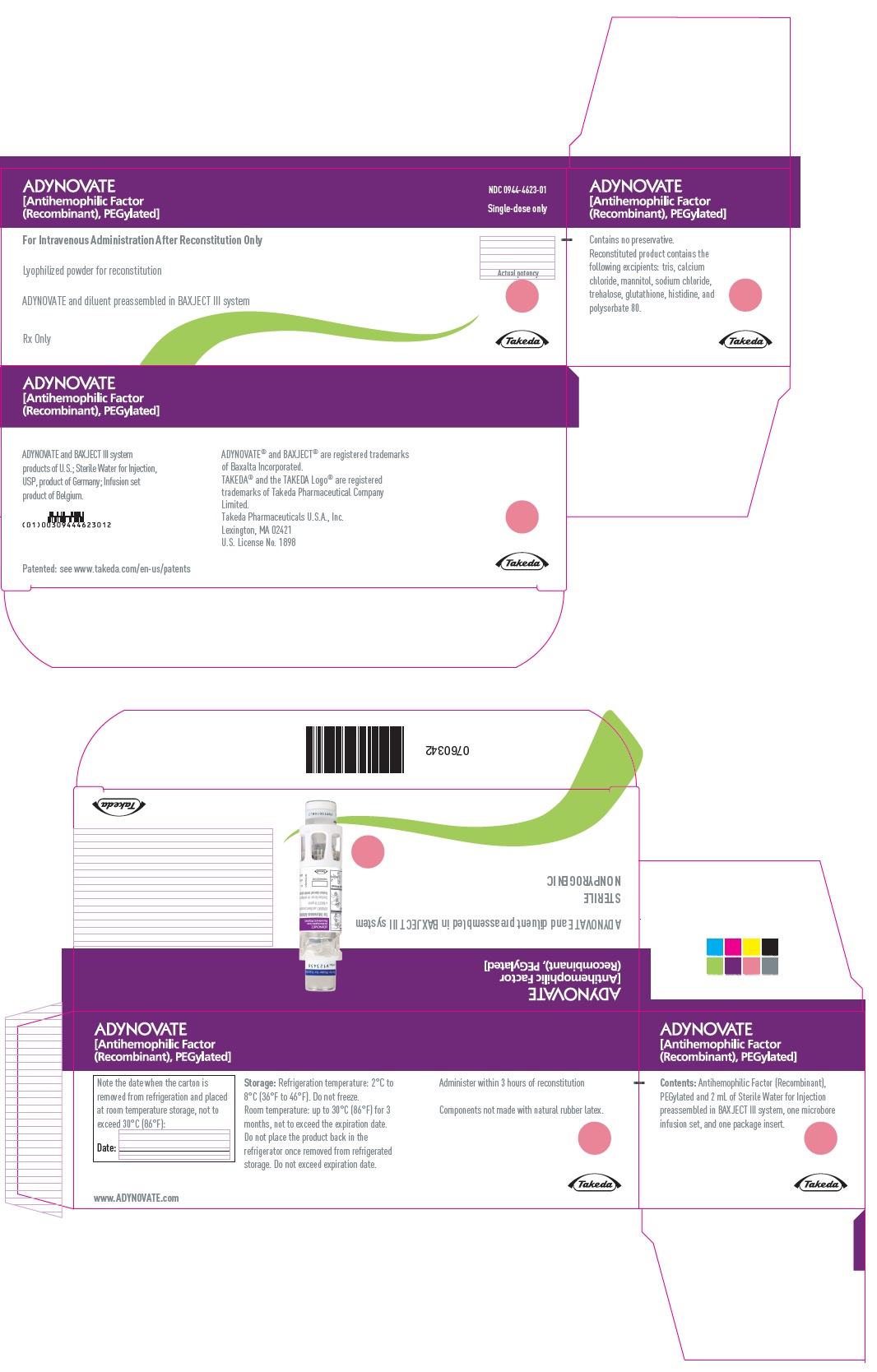
PRINCIPAL DISPLAY PANEL - Kit Carton - 500 IU
ADYNOVATE
[Antihemophilic Factor
(Recombinant), PEGylated]
NDC 0944-4623-01
Single-dose only
For Intravenous Administration After Reconstitution Only
Lyophilized powder for reconstitution
ADYNOVATE and diluent preassembled in BAXJECT III system
Rx Only
Actual potency
Takeda
PRINCIPAL DISPLAY PANEL - Barrel Label - 500 IU
ADYNOVATE
[Antihemophilic Factor
(Recombinant), PEGylated]
For Intravenous Administration Only
ADYNOVATE and diluent preassembled
in BAXJECT III system
Directions for use: see package insert
Rx Only
IU:
LOT:
EXP:
Takeda
0754167

PRINCIPAL DISPLAY PANEL - Blister Label - 500 IU
ADYNOVATE
[Antihemophilic Factor
(Recombinant), PEGylated]
NDC 0944-4623-02
Single-dose only
0760341
For Intravenous Administration Only
ADYNOVATE and diluent preassembled in BAXJECT III system
Do not open until time of use
Do not use if packaging is opened or damaged
Rx Only
Takeda

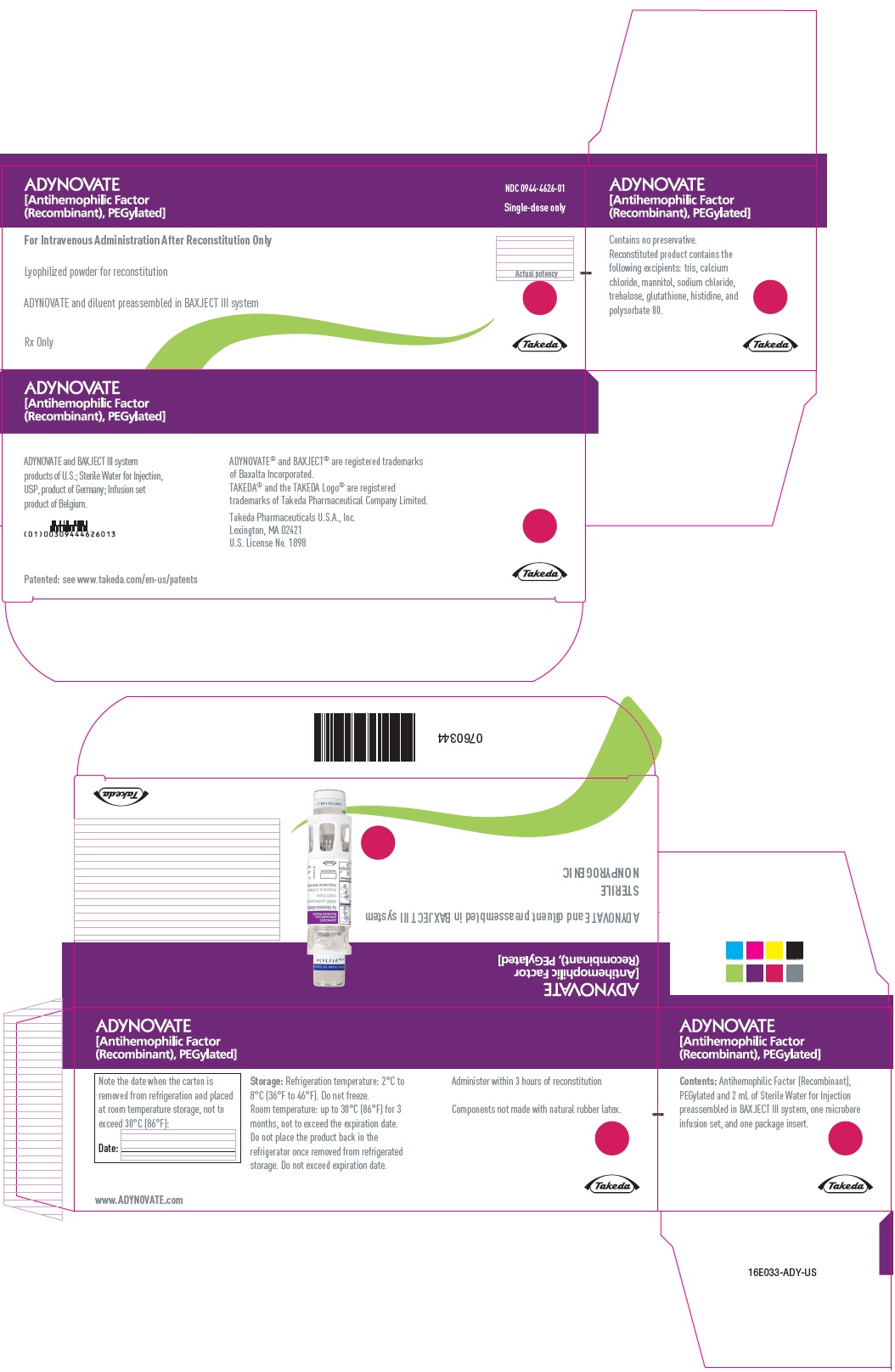
PRINCIPAL DISPLAY PANEL - Kit Carton - 750 IU
ADYNOVATE
[Antihemophilic Factor
(Recombinant), PEGylated]
NDC 0944-4626-01
Single-dose only
For Intravenous Administration After Reconstitution Only
Lyophilized powder for reconstitution
ADYNOVATE and diluent preassembled in BAXJECT III system
Rx Only
Actual potency
Takeda
PRINCIPAL DISPLAY PANEL - Barrel Label - 750 IU
ADYNOVATE
[Antihemophilic Factor
(Recombinant), PEGylated]
For Intravenous Administration Only
ADYNOVATE and diluent preassembled
in BAXJECT III system
Directions for use: see package insert
Rx Only
IU:
LOT:
EXP:
Takeda
0754182

PRINCIPAL DISPLAY PANEL - Blister Label - 750 IU
ADYNOVATE
[Antihemophilic Factor
(Recombinant), PEGylated]
NDC 0944-4626-02
Single-dose only
0760343
For Intravenous Administration Only
ADYNOVATE and diluent preassembled in BAXJECT III system
Do not open until time of use
Do not use if packaging is opened or damaged
Rx Only
Takeda

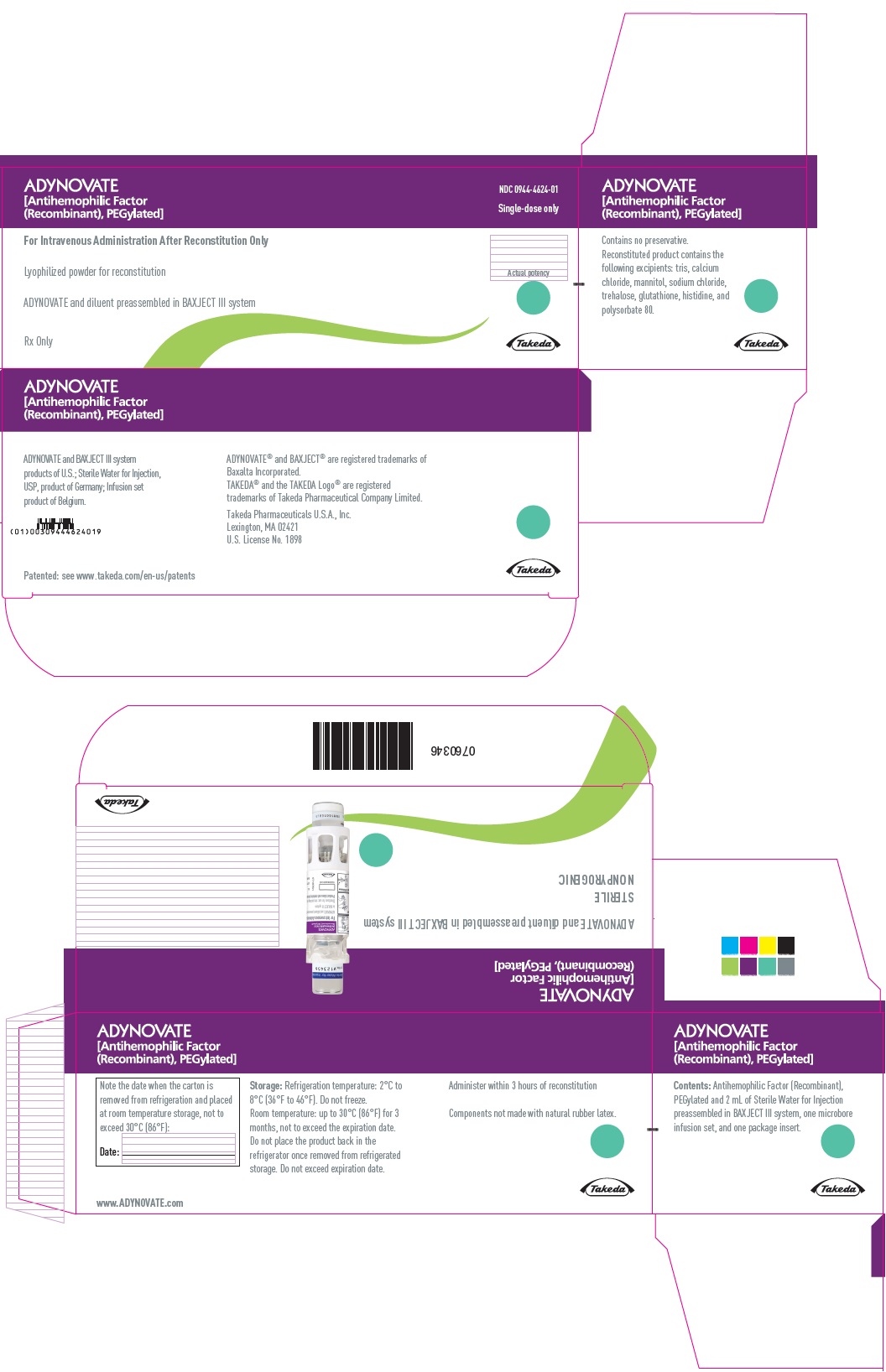
PRINCIPAL DISPLAY PANEL - Kit Carton - 1000 IU
ADYNOVATE
[Antihemophilic Factor
(Recombinant), PEGylated]
NDC 0944-4624-01
Single-dose only
For Intravenous Administration After Reconstitution Only
Lyophilized powder for reconstitution
ADYNOVATE and diluent preassembled in BAXJECT III system
Rx Only
Actual potency
Takeda
PRINCIPAL DISPLAY PANEL - Barrel Label - 1000 IU
ADYNOVATE
[Antihemophilic Factor
(Recombinant), PEGylated]
For Intravenous Administration Only
ADYNOVATE and diluent preassembled
in BAXJECT III system
Directions for use: see package insert
Rx Only
IU:
LOT:
EXP:
Takeda
0754170

PRINCIPAL DISPLAY PANEL - Blister Label - 1000 IU
ADYNOVATE
[Antihemophilic Factor
(Recombinant), PEGylated]
NDC 0944-4624-02
Single-dose only
0760345
For Intravenous Administration Only
ADYNOVATE and diluent preassembled in BAXJECT III system
Do not open until time of use
Do not use if packaging is opened or damaged
Rx Only
Takeda

PRINCIPAL DISPLAY PANEL - Kit Carton - 1500 IU
ADYNOVATE
[Antihemophilic Factor
(Recombinant), PEGylated]
NDC 0944-4627-01
Single-dose only
For Intravenous Administration After Reconstitution Only
Lyophilized powder for reconstitution
ADYNOVATE and diluent preassembled in BAXJECT III system
Rx Only
Actual potency
Takeda
PRINCIPAL DISPLAY PANEL - Barrel Label - 1500 IU
ADYNOVATE
[Antihemophilic Factor
(Recombinant), PEGylated]
For Intravenous Administration Only
ADYNOVATE and diluent preassembled
in BAXJECT III system
Directions for use: see package insert
Rx Only
IU:
LOT:
EXP:
Takeda
0754173

PRINCIPAL DISPLAY PANEL - Blister Label - 1500 IU
ADYNOVATE
[Antihemophilic Factor
(Recombinant), PEGylated]
NDC 0944-4627-02
Single-dose only
0760353
For Intravenous Administration Only
ADYNOVATE and diluent preassembled in BAXJECT III system
Do not open until time of use
Do not use if packaging is opened or damaged
Rx Only
Takeda

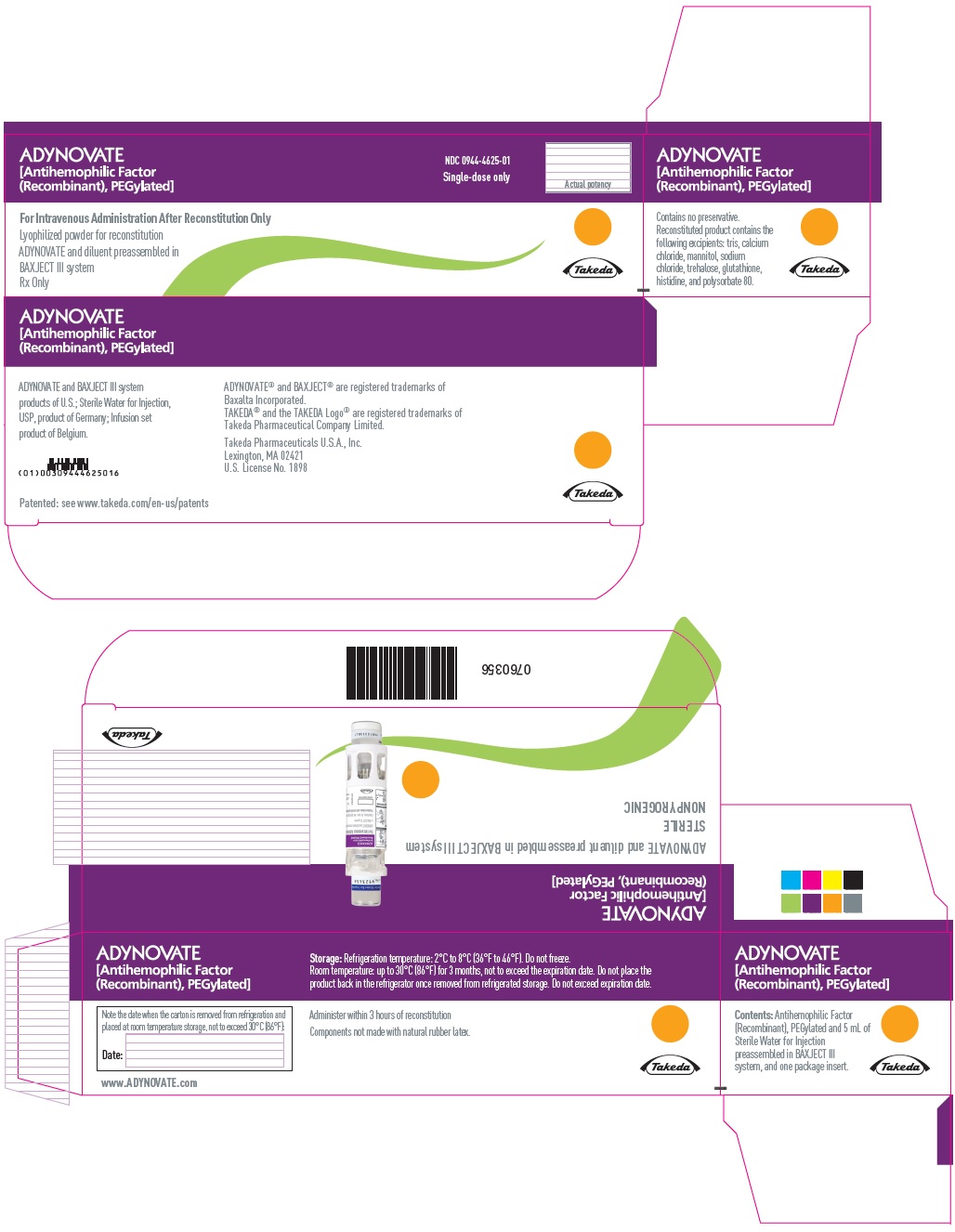
PRINCIPAL DISPLAY PANEL - Kit Carton - 2000 IU
ADYNOVATE
[Antihemophilic Factor
(Recombinant), PEGylated]
NDC 0944-4625-01
Single-dose only
Actual potency
For Intravenous Administration After Reconstitution Only
Lyophilized powder for reconstitution
ADYNOVATE and diluent preassembled in
BAXJECT III system
Rx Only
Takeda
PRINCIPAL DISPLAY PANEL - Barrel Label - 2000 IU
ADYNOVATE
[Antihemophilic Factor
(Recombinant), PEGylated]
For Intravenous Administration Only
ADYNOVATE and diluent preassembled
in BAXJECT III system
Directions for use: see package insert
Rx Only
IU:
LOT:
EXP:
Takeda
0754176

PRINCIPAL DISPLAY PANEL - Blister Label - 2000 IU
ADYNOVATE
[Antihemophilic Factor
(Recombinant), PEGylated]
NDC 0944-4625-02
Single-dose only
0760355
For Intravenous Administration Only
ADYNOVATE and diluent preassembled in BAXJECT III system
Do not open until time of use
Do not use if packaging is opened or damaged
Rx Only
Takeda

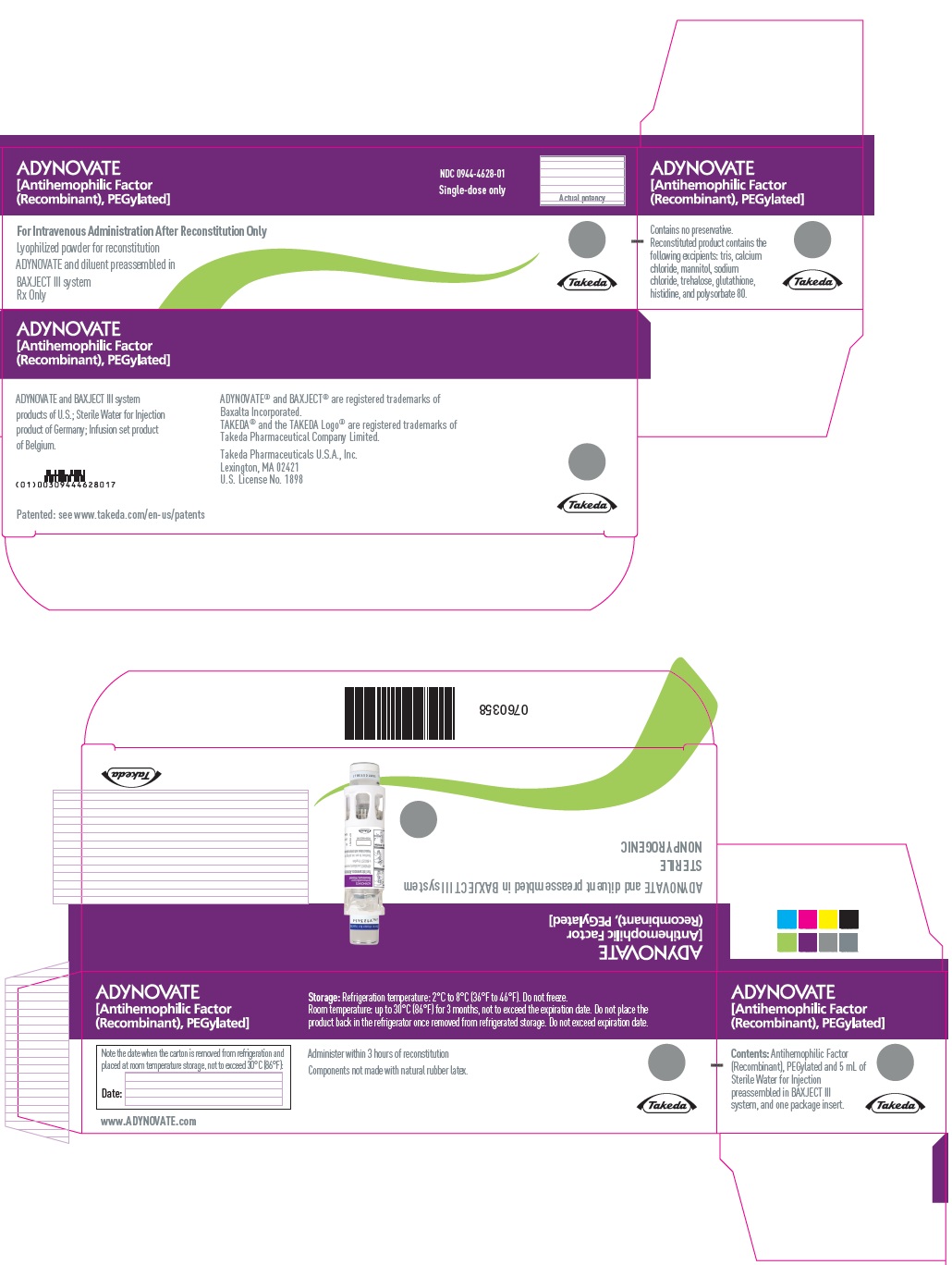
PRINCIPAL DISPLAY PANEL - Kit Carton - 3000 IU
ADYNOVATE
[Antihemophilic Factor
(Recombinant), PEGylated]
NDC 0944-4628-01
Single-dose only
Actual potency
For Intravenous Administration After Reconstitution Only
Lyophilized powder for reconstitution
ADYNOVATE and diluent preassembled in
BAXJECT III system
Rx Only
Takeda
PRINCIPAL DISPLAY PANEL - Barrel Label - 3000 IU
ADYNOVATE
[Antihemophilic Factor
(Recombinant), PEGylated]
For Intravenous Administration Only
ADYNOVATE and diluent preassembled
in BAXJECT III system
Directions for use: see package insert
Rx Only
IU:
LOT:
EXP:
Takeda
0754179

PRINCIPAL DISPLAY PANEL - Blister Label - 3000 IU
ADYNOVATE
[Antihemophilic Factor
(Recombinant), PEGylated]
NDC 0944-4628-02
Single-dose only
0760357
For Intravenous Administration Only
ADYNOVATE and diluent preassembled in BAXJECT III system
Do not open until time of use
Do not use if packaging is opened or damaged
Rx Only
Takeda









