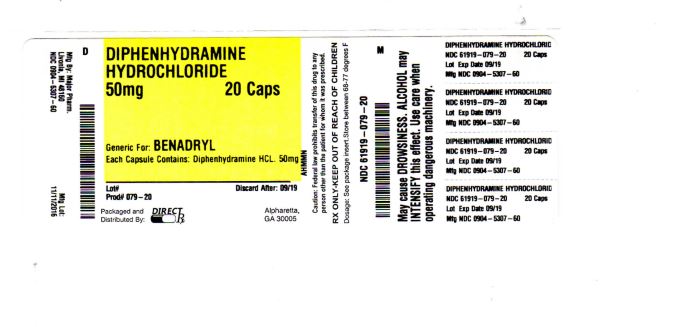
DIPHENHYDRAMINE HYDROCHLORIDE- diphenhydramine hydrochloride capsule
Direct Rx
----------
DIPHENHYDRAMINE HYDROCHLORIDE
Uses
temporarily relieves these symptoms of hay fever or other upper respiratory allergies:
- runny nose
- itchy nose or throat
- sneezing
- itchy, watery eyes
WARNINGS
Do not use with any other product containing diphenhydramine, even one used on skin
Ask a doctor before use if you have
- glaucoma
- trouble urinating due to an enlarged prostate gland
- a breathing problem such as emphysema or chronic bronchitis
Ask a doctor or pharmacist before use if you are taking sedatives or tranquilizers
When using this product
- you may get very drowsy
- avoid alcoholic drink
- salcohol, sedatives & tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
- excitability may occur, especially in children
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away.
DIRECTIONS
- adults and children 12 years and over:take 1 to 2 capsules every 4-6 hours; not more than 6 doses in 24 hours
- children under 12 years: ask a doctor
Other Information
- store at 15-30 °C (59-86 °F)
- protect from moisture
- For 1000 Count: This is a bulk package. Dispense contents in a tight, light-resistant container with a child-resistant closure as defined in the USP
Inactive Ingredients
benzyl alcohol, butylparaben, D&C red# 28, edible black ink, FD&C bule #1, FD&C red# 40, gelatin, lactose, magnesium stearate, methylparaben, polysorbate 80, propylparaben, sodium laurel sulfate
Questions or Comments
TAMPER EVIDENT: DO NOT USE IF IMPRINTED SAFETY SEAL UNDER CAP OR BAND AROUND ANY CAPSULE IS MISSING OR DAMAGED
Distributed by: Qualitest Pharmaceuticals, Inc.
OTHER INFORMATION
- store at 15-30 °C (59-86 °F)
- protect from moisture
- For 1000 Count: This is a bulk package. Dispense contents in a tight, light-resistant container with a child-resistant closure as defined in the USP
OTHER INFORMATION
benzyl alcohol, butylparaben, D&C red# 28, edible black ink, FD&C bule #1, FD&C red# 40, gelatin, lactose, magnesium stearate, methylparaben, polysorbate 80, propylparaben, sodium laurel sulfate
| DIPHENHYDRAMINE HYDROCHLORIDE
diphenhydramine hydrochloride capsule |
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| Labeler - Direct Rx (079254320) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Direct Rx | 079254320 | repack(61919-079) , relabel(61919-079) | |