FULL PRESCRIBING INFORMATION
WARNING
THROMBOSIS, RENAL DYSFUNCTION AND ACUTE RENAL FAILURE
- Thrombosis may occur with immune globulin intravenous (IGIV) products, including Octagam 10%. Risk factors may include: advanced age, prolonged immobilization, hypercoagulable conditions, history of venous or arterial thrombosis, use of estrogens, indwelling central vascular catheters, hyperviscosity, and cardiovascular risk factors. Thrombosis may occur in the absence of known risk factors. (See WARNING and PRECAUTIONS [ 5.5 ], PATIENT COUNSELING INFORMATION [ 17 ]
- Renal dysfunction, acute renal failure, osmotic nephrosis, and death may occur in predisposed patients who receive IGIV products, including Octagam 10%. Patients predisposed to renal dysfunction include those with a degree of pre-existing renal insufficiency, diabetes mellitus, age greater than 65, volume depletion, sepsis, paraproteinemia, or patients receiving known nephrotoxic drugs. Renal dysfunction and acute renal failure occur more commonly in patients receiving IGIV product containing sucrose. Octagam 10% does not contain sucrose.
- For patients at risk of thrombosis, renal dysfunction or acute renal failure, administer Octagam 10% at the minimum dose and infusion rate practicable. Ensure adequate hydration in patients before administration. Monitor for signs and symptoms of thrombosis and assess blood viscosity in patients at risk for hyperviscosity. (See DOSAGE and ADMINISTRATION [ 2.3 ], WARNINGS and PRECAUTIONS [ 5.5 ])
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
For intravenous use only.
2.1 Dosage
| Indication | Dose | Initial Infusion rate | Maintenance Infusion Rate (if tolerated) |
| Chronic ITP | 2 g/kg divided in equal doses given on 2 consecutive days | 1.0 mg/kg/min(0.01 mL/kg/min) | Up to 12.0 mg/kg/min(Up to 0.12 mL/kg/min) |
| Dermatomyositis (DM) | 2 g/kg divided in equal doses given over 2-5 consecutive days every 4 weeks | 1.0 mg/kg/min(0.01 mL/kg/min) | Up to 4.0 mg/kg/min(Up to 0.04 mL/kg/min) |
2.2 Preparation and Handling
- Parenteral products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not use if Octagam 10% is turbid and/or if discoloration is observed.
- Do not mix Octagam 10% with other medicinal products or administer simultaneously with other intravenous preparation in the same infusion set. Do not mix Octagam 10% with immune globulin intravenous (IGIV) products from other manufacturers.
- Do not freeze Octagam 10%. Do not use solutions that have been frozen.
- Octagam 10% contains no preservative. Octagam 10% bottle is for single use only. Use promptly any bottle that has been entered or opened, and discard partially used bottles.
- The content of Octagam 10% bottles may be pooled under aseptic conditions into sterile infusion bags and infused within 8 hours after pooling.
- Do not use after expiration date.
- Do not dilute Octagam 10%.
- The infusion line may be flushed before and after administration of Octagam 10% with either normal saline or 5% dextrose in water.
2.3 Administration
- Administer Octagam 10%, which is to be at room temperature, only by the intravenous route.
- Octagam 10% is not supplied with an infusion set. If an in-line filter is used the pore size should be 0.2 – 200 microns.
- Do not use a needle of larger than 16 gauge to prevent the possibility of coring. Insert needle only once, within the stopper area delineated (by the raised ring for penetration). Penetrate the stopper perpendicular to its plane and within the ring.
For rate of administration, see Table 1.
Table 1
| Rate of Administration | mg/kg/min (mg/kg/hour) | mL/kg/min |
| first 30 min | 1 (60) | 0.01 |
| next 30 min, if above is tolerated | 2 (120) | 0.02 |
| next 30 min, if above is tolerated | 4 (240) | 0.04* |
| next 30 min, if above is tolerated | 8 (480) | 0.08 |
| Maximum | ≤ 12 (≤720) | ≤0.12 |
*Patients with dermatomyositis are at increased risk for thromboembolic events; monitor carefully and do not exceed an infusion rate of 0.04 ml/kg/min.
- Monitor the patient carefully throughout the infusion. Certain adverse drug reactions may be related to the rate of infusion. Slowing or stopping the infusion usually allows the symptoms to disappear promptly. Once the symptoms subside, the infusion may then be resumed at a lower rate.
- Ensure that patients with pre-existing renal insufficiency are not volume depleted. For patients at risk of renal dysfunction or thromboembolic events, administer Octagam 10% at the minimum infusion rate practicable, not to exceed 3.3 mg/kg/min (0.03 mL/kg/min). Discontinue Octagam 10% if renal function deteriorates.
3 DOSAGE FORMS AND STRENGTHS
Solution containing 10% IgG (100 mg/mL) (See How Supplied/Storage and Handling ( 16 )).
4 CONTRAINDICATIONS
- Octagam 10% is contraindicated in patients who have a history of severe systemic hypersensitivity reactions, such as anaphylaxis, to human immunoglobulin.
- Octagam 10% contains trace amounts of IgA (average 106 µg/mL in a 10% solution). It is contraindicated in IgA-deficient patients with antibodies against IgA and history of hypersensitivity .
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity
Severe hypersensitivity reactions may occur (See Contraindications ( 4 ) ) .[ 1 ] In case of hypersensitivity, discontinue Octagam 10% infusion immediately and institute appropriate treatment. Epinephrine should be immediately available for treatment of severe acute hypersensitivity reactions.
Octagam 10% contains trace amounts of IgA (average 106 µg/mL in a 10% solution). IgA-deficient patients with antibodies against IgA are at greater risk of developing severe hypersensitivity and anaphylactoid reactions when administered Octagam 10% (See Contraindications ( 4 )) .
Octagam 10% may cause hypersensitivity reactions in patients with corn allergy. Octagam 10% contains maltose, a disaccharide sugar which is derived from corn.
5.2 Renal Dysfunction/Failure
Renal dysfunction, acute renal failure, osmotic nephropathy, and death may occur upon use of Octagam 10% in predisposed patients.
Ensure that patients are not volume depleted prior to the initiation of the infusion of Octagam 10%.
For patients at risk of renal dysfunction because of pre-existing renal insufficiency or predisposition to acute renal failure (such as individuals with diabetes mellitus, age greater than 65, volume depletion, sepsis, paraproteinemia, or those receiving known nephrotoxic drugs), administer Octagam 10% at the minimum infusion rate practicable, not to exceed 3.3mg/kg/min (0.03mL/kg/min) (See Boxed Warning, and Dosage and Administration ( 2 )).
Periodic monitoring of renal function tests and urine output is particularly important in patients judged to be at risk of developing acute renal failure. Assess renal function, including a measurement of blood urea nitrogen (BUN)/serum creatinine, prior to the initial infusion of Octagam 10% and again at appropriate intervals thereafter. If renal function deteriorates, consider discontinuation of the product.
5.3 Blood Glucose Monitoring
Some glucose monitoring systems give falsely elevated glucose readings because they interpret the maltose in Octagam 10% as glucose.[ 2 ] This occurs in systems that use glucose dehydrogenase pyrroloquinoline quinone (GDH-PQQ) or glucose-dye-oxidoreductase methods for detecting blood glucose. Inaccurate glucose readings can cause inappropriate administration of insulin and resultant potentially life-threatening hypoglycemia. Also, true hypoglycemia may go undetected when masked by a falsely elevated glucose reading.
Monitor glucose levels in diabetic patients with a glucose-specific method only. Review the product information of blood glucose testing systems, including the test strips, to determine if the system is appropriate to use with maltose-containing parenteral products. Contact the manufacturer if there is any uncertainty about the system.
5.4 Hyperproteinemia, Increased Serum Viscosity and Hyponatremia
Hyperproteinemia, increased serum viscosity and hyponatremia may occur in patients receiving Octagam 10% therapy. It is clinically critical to distinguish true hyponatremia from pseudohyponatremia related to hyperproteinemia with concomitant decreased calculated serum osmolality or elevated osmolar gap, because treatment aimed at decreasing serum free water in patients with pseudohyponatremia may lead to volume depletion, a further increase in serum viscosity and a higher risk of thromboembolic events.[ 3 ]
5.5 Thrombosis
Thrombosis may occur following treatment with immune globulin products, including Octagam 10%. Risk factors may include: advanced age, prolonged immobilization, hypercoagulable conditions, history of venous or arterial thrombosis, use of estrogens, indwelling central vascular catheters, hyperviscosity, and cardiovascular risk factors. Thrombosis may occur in the absence of known risk factors.
Consider baseline assessment of blood viscosity in patients at risk for hyperviscosity, including those with cryoglobulins, fasting chylomicronemia / markedly high triacylglycerols (triglycerides), or monoclonal gammopathies. For patients at risk of thrombotic events, administer Octagam 10% at the minimum dose and infusion rate practicable. Ensure adequate hydration in patients before administration. Monitor for signs and symptoms of thrombosis and assess blood viscosity in patients at risk for hyperviscosity. [ 4-6 ] (See BOXED WARNING, DOSAGE and ADMINSTRATION [ 2.3 ], PATIENT COUNSELING INFORMATION [ 17 ])
5.6 Hemolysis
Octagam 10% may contain blood group antibodies that can act as hemolysins and induce in vivo coating of red blood cells (RBCs) with immunoglobulin, causing a positive direct antiglobulin test (DAT) (Coombs’ test) result and hemolysis. Delayed hemolytic anemia can develop subsequent to IGIV therapy due to enhanced RBC sequestration and acute hemolysis, consistent with intravascular hemolysis, has been reported. Cases of severe hemolysis-related renal dysfunction/failure or disseminated intravascular coagulation have occurred following infusion of IGIV.
The following risk factors may be associated with the development of hemolysis following IGIV administration: high doses (e.g. ≥ 2 g/kg), given either as a single administration or divided over several days, and non-O blood group.[ 7 ]. Other individual patient factors, such as an underlying inflammatory state (as may be reflected by, for example, elevated C-reactive protein or erythrocyte sedimentation rate), have been hypothesized to increase the risk of hemolysis following administration of IGIV.[ 8 ], but their role is uncertain. Hemolysis has been reported following administration of IGIV for a variety of indications, including ITP.
Closely monitor patients for clinical signs and symptoms of hemolysis, particularly patients with risk factors noted above. Consider appropriate laboratory testing in higher risk patients, including measurement of hemoglobin or hematocrit prior to infusion and within approximately 36 to 96 hours post infusion. If clinical signs and symptoms of hemolysis or a significant drop in hemoglobin or hematocrit have been observed, perform confirmatory laboratory testing. If transfusion is indicated for patients who develop hemolysis with clinically compromising anemia after receiving IGIV, perform adequate cross-matching to avoid exacerbating on-going hemolysis.
5.7 Aseptic Meningitis Syndrome
Aseptic meningitis syndrome (AMS) may occur with Octagam treatment. Discontinuation of treatment has resulted in remission of AMS within several days without sequelae. The syndrome usually begins within several hours to two days following rapid infusion with Octagam treatment. It is characterized by symptoms and signs including severe headache, nuchal rigidity, drowsiness, fever, photophobia, painful eye movements, nausea and vomiting. Cerebrospinal fluid (CSF) studies frequently reveal pleocytosis up to several thousand cells per cubic millimeter, predominantly from the granulocytic series, and elevated protein levels up to several hundred mg/dl, but negative culture results. Conduct a thorough neurological examination in patients exhibiting such symptoms and signs, including CSF studies, to rule out other causes of meningitis. It appears that patients with a history of migraine may be more susceptible.[ 9 ]
AMS may occur more frequently following high doses (≥2 g/kg) and/or rapid infusion of IGIV.
5.8 Transfusion-Related Acute Lung Injury (TRALI)
Noncardiogenic pulmonary edema [Transfusion-Related Acute Lung Injury (TRALI)] may occur in patients administered IGIV.[ 10 ] TRALI is characterized by severe respiratory distress, pulmonary edema, hypoxemia, normal left ventricular function, and fever. Signs and symptoms typically appear within 1 to 6 hours after transfusion. Patients with TRALI may be managed using oxygen therapy with adequate ventilatory support.
Monitor recipients of Octagam 10% for pulmonary adverse reactions. If TRALI is suspected, perform appropriate tests for the presence of anti-HLA and anti-neutrophil antibodies in the product.
5.9 Transmission of Infectious Agents
Because Octagam 10% is made from human blood, it may carry a risk of transmitting infectious agents, e.g., viruses and theoretically, the Creutzfeldt-Jakob disease (CJD) agent. Report all infections thought by a physician or other healthcare provider to have been possibly transmitted by this product to Pfizer Inc. at 1-800-438-1985. Discuss the risks and benefits of Octagam 10% with the patient before prescribing or administering this product.
5.10 Monitoring Laboratory Tests
- After infusion of immunoglobulin, the transitory rise of the various passively transferred antibodies in the patient’s blood may yield positive serological testing results, with the potential for misleading interpretation.
- Passive transmission of antibodies to erythrocyte antigens (e.g. A, B, and D) may cause a positive direct or indirect antiglobulin (Coombs’) test. Clinically assess patients with known renal dysfunction, diabetes mellitus, age greater than 65, volume depletion, sepsis, paraproteinemia, or those receiving nephrotic agents, and monitor as appropriate (BUN; serum creatinine, urine output) during therapy with Octagam 10%.
- Consider baseline assessment of blood viscosity in patients at risk for hyperviscosity, including those with polycythemia, cryoglobulins, fasting chylomicronemia/markedly high triglycerides, or monoclonal gammopathies.
- Consider measuring hemoglobin or hematocrit at baseline and approximately 36 to 96 hours post infusion in patients at higher risk of hemolysis. If signs and/or symptoms of hemolysis are present after an infusion of Octagam 10%, perform appropriate laboratory testing for confirmation.
- If TRALI is suspected, perform appropriate tests for the presence of anti-neutrophil antibodies in both the product and patient’s serum.
6 ADVERSE REACTIONS
Chronic Immune Thrombocytopenic Purpura (ITP)
The most common adverse reactions observed in > 5% of clinical study subjects with ITP were headache, fever, and increased heart rate.
Dermatomyositis (DM)
The most common adverse reactions observed in > 5% of clinical study subjects with DM were headache, fever, nausea, vomiting, increased blood pressure, chills, musculoskeletal pain, increased heart rate, dyspnea, and infusions site reactions.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a product cannot be directly compared to rates in the clinical trials of another product and may not reflect the rates observed in clinical practice.
Treatment of Chronic Immune Thrombocytopenic Purpura (ITP) in Adults
In a prospective, open-label, single-arm, multicenter study 116 subjects with newly diagnosed or chronic ITP received Octagam 10% at a dose of 2 g/kg, administered daily as 1 g/kg intravenous infusions on 2 consecutive days. In the initial study phase, the infusion rate allowed for Octagam 10% administration was up to 6 mg/kg/min [0.06 mL/kg/min], which was achieved in 24 of 26 subjects (92%). Subsequently, the maximum infusion rate allowed was 12 mg/kg/min [0.12mL/kg/min], and 54 (60%) of 90 subjects received Octagam 10% at this rate in the second phase of the trial.
Pre-medication to alleviate potential adverse drug reactions was allowed, and was only administered to 1 (0.9%) of the 116 patients.
The investigators judged 129 (54%) of the temporally associated ("infusional") adverse reactions (AR) in 62 patients to be at least possibly related to the infusion of Octagam 10%. The most common causally related ARs observed in > 5% of subjects are shown in Table 2.
Table 2: Drug-related Adverse Reactions Experienced by >5% of Subjects
| Reactions | No. of Subjects (% of Subjects [n=116]) |
| Headache | 29 (25) |
| Pyrexia | 17 (15) |
| Heart Rate Increased | 13 (11) |
Table 3: ARs in >5% Subjects During and Within 72 Hours After the End of 2-Days Treatment Cycle, Irrespective of Causality
| Reactions | No. of Subjects (% of Subjects [n=116]) |
| Headache | 35 (30) |
| Heart Rate Increased | 25 (22) |
| Pyrexia | 20 (17) |
| Heart Rate Decreased | 18 (16) |
| Hypertension | 7 (6) |
One subject experienced a serious adverse reaction (headache).
Treatment of Dermatomyositis (DM) in Adults
In a prospective, double-blind, randomized, placebo-controlled, multicenter study with two study periods, 95 adults with DM were enrolled. In the First Period (16 weeks), 47 subjects received 2 g/kg Octagam 10% and 48 subjects received placebo every 4 weeks for 4 infusion cycles. In the following 6-month open-label Extension Period, 91 subjects received 6 infusion cycles of Octagam 10% (one infusion cycle every 4 weeks). One infusion cycle comprised of all infusions administered over 2-5 days.
Table 4 and Table 5 summarize the most frequent adverse reactions (ARs) that occurred in >5% of subjects with DM in the clinical study. Premedication for infusions was given to 21.3% of patients in the Octagam group.
During the study, no subject met the criteria for a hemolytic transfusion reaction (HTR). No deaths were reported during the study.
The following serious adverse reactions were observed in the study: muscle spasms and dyspnea in one subject, loss of consciousness in one subject, and thromboembolic events (TEEs) in five subjects, including deep vein thrombosis and pulmonary embolism in one subject, cerebrovascular accident in one subject, cerebral infarction in one subject, hypoesthesia in one subject and pulmonary embolism in one subject. For risk factors for TEE see also [ 5.5 ]).
Table 4: Drug-related Adverse Reactions Experienced by >5% of Subjects
| Reactions | No. of Subjects (% of Subjects [n=95]) |
| Headache | 40 (42) |
| Pyrexia | 18 (19) |
| Nausea | 15 (16) |
| Vomiting | 8 (8) |
| Chills | 7 (7) |
| Musculoskeletal pain | 7 (7) |
| Blood pressure increased | 6 (6) |
Table 5: ARs in >5% of Subjects During and Within 72 Hours After End of Infusion Cycle, Irrespective of Causality
| Reactions | No. of Subjects (% of Subjects [n=95]) |
| Headache | 44 (46) |
| Pyrexia | 19 (20) |
| Nausea | 16 (17) |
| Vomiting | 8 (8) |
| Blood pressure increased | 8 (8) |
| Chills | 7 (7) |
| Musculoskeletal pain | 5 (5) |
| Heart Rate Increased | 5 (5) |
| Dyspnea | 5 (5) |
| Infusion site reactions | 5 (5) |
6.2 Postmarketing Experience
Because postmarketing reporting of adverse reactions is voluntary and from a population of uncertain size, it is not always possible to reliably estimate the frequency of these reactions or establish a causal relationship to product exposure.
The following adverse reactions have been identified during post-approval use of Octagam.
|
Blood and lymphatic system disorders
Leucopenia, hemolytic anemia |
|
Immune system disorders
Hypersensitivity, anaphylactic shock, anaphylactic reaction, anaphylactoid reaction, angioedema, face edema |
|
Metabolic and nutritional disorders
Fluid overload, (pseudo)hyponatremia |
|
Psychiatric disorders
Agitation, confusional state, anxiety, nervousness |
|
Nervous system disorders
Cerebrovascular accident, loss of consciousness, speech disorder, aseptic meningitis, migraine, dizziness, paresthesia, hypoesthesia, tremor, photophobia, |
|
Eye disorders
Visual impairment |
|
Cardiac disorders
Myocardial infarction, angina pectoris, bradycardia, tachycardia, palpitations, cyanosis |
|
Vascular disorders
Hypotension, thrombosis, circulatory collapse, peripheral circulatory failure, hypertension, phlebitis, pallor |
|
Respiratory, thoracic and mediastinal disorders
Respiratory failure, pulmonary embolism, pulmonary edema, bronchospasm, dyspnea, cough, hypoxia |
|
Gastrointestinal disorders
Nausea, vomiting, diarrhea, abdominal pain |
|
Skin and subcutaneous tissue disorders
Eczema, skin exfoliation, urticaria, rash, rash erythematous, dermatitis, erythema, pruritus, alopecia |
|
Musculoskeletal and connective tissue disorders
Back pain, arthralgia, myalgia, pain in extremity, neck pain, muscle spasms, muscular weakness, musculoskeletal stiffness |
|
Renal and urinary disorders
Acute renal failure, renal pain |
|
General disorders and administration site conditions
Fatigue, oedema, injection site reaction, pyrexia, influenza-like illness, chills, chest pain, chest discomfort, hot flush, flushing, feeling hot, feeling cold, hyperhidrosis, malaise, lethargy, asthenia, burning sensation |
|
Investigations
Hepatic enzymes increased, falsely elevated blood glucose level |
|
Injury, poisoning and procedural complications
Transfusion-related acute lung injury |
7 DRUG INTERACTIONS
Admixtures of Octagam 10% with other drugs and intravenous solutions have not been evaluated. It is recommended that Octagam 10% be administered separately from other drugs or medications which the patient may be receiving. Do not mix the product.
Do not mix Octagam 10% with IGIVs from other manufacturers.
Passively transferred antibodies in immunoglobulin preparations can confound the results of serological testing.
Antibodies in Octagam 10% may interfere with the response to live viral vaccines, such as measles, mumps, and rubella. Inform physicians of recent therapy with Octagam 10%, so that administration of live viral vaccines, if indicated, can be appropriately delayed for 3 or more months from the time of Octagam 10% administration.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
No human data are available to indicate the presence or absence of drug-associated risk. Animal reproduction studies have not been conducted with Octagam 10%. It is not known whether Octagam 10% can cause foetal harm when administered to a pregnant woman or can affect reproduction capacity. Immune globulins cross the placenta from maternal circulation increasingly after 30 weeks of gestation. Octagam 10% should be given to pregnant women only if clearly needed. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
8.2 Lactation
Risk Summary
No human data are available to assess the presence or absence of Octagam 10% in human milk, the effects of Octagam 10% on the breastfed child, and the effects of Octagam 10% on milk production/excretion. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Octagam 10% liquid and any potential adverse effects on the breastfed infant from Octagam 10% liquid or from the underlying maternal condition. Immunoglobulins are excreted into the milk and may contribute to the transfer of protective antibodies to the neonate.
8.4 Pediatric Use
The safety and effectiveness of Octagam 10% has not been established in pediatric patients with ITP or DM.
8.5 Geriatric Use
Patients > 65 years of age may be at increased risk for developing certain adverse reactions such as thromboembolic events and acute renal failure (See Boxed Warnings and Thrombotic Events ( 5.5 ) and Renal Failure ( 5.2 ) . Do not exceed recommended doses in this population, and the applied infusion rate should be the minimum practicable. Clinical studies of Octagam 10% did not include sufficient numbers of subjects > 65 years to determine whether they respond differently from younger subjects. During the First Period (placebo-controlled) of the DM trial, 11 patients over 65 years were treated with Octagam.
10 OVERDOSAGE
With intravenous administration, overdose of Octagam 10% may lead to fluid overload and hyperviscosity. Patients at risk of complications of fluid overload and hyperviscosity include elderly patients and those with cardiac or renal impairment.
11 DESCRIPTION
Immune Globulin Intravenous (Human), Octagam 10%, is a solvent/detergent (S/D)-treated, sterile preparation of highly purified immunoglobulin G (IgG) derived from large pools of human plasma. Octagam 10% is a solution for infusion to be administered intravenously.
This preparation contains approximately 100 mg of protein per mL (10%) of which not less than 96% is normal human immunoglobulin G. Octagam 10% contains not more than 3% aggregates, not less than 94% monomers and dimers and not more than 3% fragments. On average, the product contains 106 µg/mL of IgA and even lower amounts of IgM.
The sodium content of the final solution is not more than 30 mmol/L and the pH is between 4.5 and 5.0. The osmolality is 310 - 380 mosmol/kg.
The manufacturing process for Octagam 10% isolates IgG without additional chemical or enzymatic modification, and the Fc portion is maintained intact Octagam 10% contains the IgG antibody activities present in the donor population. IgG subclasses are fully represented with the following approximate percents of total IgG: IgG 1 is 65%, IgG 2 is 30%, IgG 3 is 3% and IgG 4 is 2%.
Octagam 10% contains a broad spectrum of IgG antibodies against bacterial and viral agents that are capable of opsonization and neutralization of microbes and toxins. It contains maltose (90 mg/mL), but no preservatives or sucrose.
All units of human plasma used in the manufacture of Octagam 10% are provided by FDA-approved blood establishments, and are tested by FDA-licensed serological tests for HBsAg, antibodies to HCV and HIV and Nucleic Acid Test (NAT) for HCV and HIV-1 and found to be non-reactive (negative).
The product is manufactured by the cold ethanol fractionation process followed by ultrafiltration and chromatography. The manufacturing process includes treatment with an organic S/D mixture composed of tri-n-butyl phosphate (TNBP) and Triton X-100 (Octoxynol). The Octagam 10% manufacturing process shows significant viral reduction in in vitro studies (Table 6). These reductions are achieved through a combination of process steps including cold ethanol fractionation, S/D treatment and pH 4 treatment.
Table 6: In vitro reduction factor during Octagam 10% manufacturing
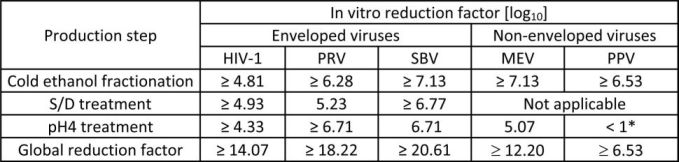
*not calculated for global LRF
HIV-1: Human Immunodeficiency Virus - 1
PRV: Pseudorabies Virus
SBV: Sindbis Virus
MEV: Mouse Encephalomyelitis Virus
PPV: Porcine Parvovirus
12 CLINICAL PHARMACOLOGY
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No studies were conducted on carcinogenesis, mutagenesis, or impairment of fertility with Octagam 10%.
Repeated-dose toxicity studies and genotoxic studies gave no evidence of carcinogenic properties of TNBP and Octoxynol.[ 11,12 ].
The results of in vitro and in vivo genotoxicity studies for TNBP and Octoxynol were negative. The results of studies on the embryotoxic and teratogenic properties of TNBP and Octoxynol in rats and rabbits at a wide range of i.v. doses were also negative.
13.2 Animal Toxicology and/or Pharmacology
No studies were conducted on pharmacology and pharmacokinetics with Octagam 10% in animals.
A variety of single-dose toxicity studies were performed for TNBP and Octoxynol alone or in combination: the lowest toxic dose of TNBP + Octoxynol (in a ratio of 1:5) was 10,000 mcg/kg BW in rats after intravenous administration. Studies on 13-week toxicity were performed for intravenous combinations of TNBP + Octoxynol in a broad dose range in dogs and rats: the lowest toxic dose for rats was local 60 mcg TNBP/kg + 300 mcg Octoxynol/kg BW (concentration: 0.0006% and 0.003%, respectively) and systemic 300 mcg TNBP/kg + 1,500 mcg Octoxynol/kg BW (concentration: 0.003% and 0.015%, respectively); the lowest toxic dose for dogs was local 50 mcg TNBP/kg + 250 mcg Octoxynol/kg BW (concentration: 0.005% and 0.025%, respectively) and systemic 500 mcg TNBP/kg + 2,500 mcg Octoxynol/kg BW (concentration: 0.05% and 0.25%, respectively).
A pharmacokinetic study was carried out in rats given 300 mcg of TNBP/kg and 1,500 mcg Octoxynol/kg BW i.v. The plasma half-life for TNBP was approximately 20 minutes. Octoxynol was not detected.
14 CLINICAL STUDIES
14.1 Treatment of Chronic Immune Thrombocytopenic Purpura
A prospective, open-label, single-arm, multicenter study assessed the efficacy, safety, and tolerability of Octagam 10% in 116 subjects with tentative newly diagnosed or chronic ITP and a platelet count of 20 x 10 9 /L or less. Subjects ranged in age from 17 to 88; 64% were female and 36% were male.
Subjects received a 2 g/kg dose of Octagam 10% administered as two daily 1 g/kg intravenous doses, given on 2 consecutive days. Pre-medication to alleviate potential adverse drug reactions was allowed but only given to 1 (0.9%) of 116 patients. In the initial study phase, the infusion rate allowed for Octagam 10% administration was up to 6 mg/kg/min [0.06 mL/kg/min], which was achieved in 24 of 26 subjects (92%). Subsequently, the maximum infusion rate allowed was 12 mg/kg/min [0.12mL/kg/min], which was achieved in 54 of 90 subjects (60%). Platelet counts were measured on Days 1 to 7, 14, 21, and 63.
The study was designed to determine the response rate defined as the percentage of subjects with an increase in platelet count to at least 50 x 10 9 /L within 7 days after the first infusion (responders). Additionally, maximum platelet count, the time to reach a platelet count of at least 50 x 10 9 /L within the first 7 days, the duration of that response (i.e. the number of days the platelet count remained in excess of 50 x 10 9 /L), and the regression of hemorrhages in subjects who had bleeding at baseline were observed.
Of the 66 subjects with chronic ITP in the efficacy analysis, 54 (82%) responded to Octagam 10% with a rise in platelet counts to at least 50 x 10 9 /L within 7 days after the first infusion. The lower bound of the overall 95% confidence interval for the response rate (73%) is above the predefined response rate of 70%.
The mean maximum platelet count achieved in the 66 subjects with chronic ITP was 227 x 10 9 /L.
Table 7. Maximum Platelet Count (x10 9 /L)
| Chronic ITP subjects (n=66) | |
| Median and range | 212.0 (18 to 686) |
| Mean ± s.d. | 227.0 ± 155.63 |
The median time to reach a platelet response of at least 50 x 10 9 /L was 2 days after the first infusion. The duration of platelet response was analyzed for the 54 subjects with chronic ITP who achieved a response within 7 days after the first infusion: the median duration of platelet response in these subjects was 12 days (range: 1 to 79 days).
Table 8. Time to and Duration of Platelet Response (Responders Only).
| Time to platelet response (days) | Duration of platelet response (days) | |
| Chronic ITP subjects (n=54) | Chronic ITP subjects (n=54) | |
| Median and range | 2 (1 to 5) | 12 (1 to 79) |
| Mean ± s.d. | 1.8 ± 0.91 | 20.9 ± 21.76 |
In 35 of the 45 subjects with chronic ITP (78%) who had bleeding at baseline, the hemorrhages had completely resolved by Day 7. A decrease in the severity of hemorrhage from baseline to Day 7 was observed in 38 of 45 subjects (84%) with chronic ITP.
Table 9. Assessment of Bleeding at Day 7 in Subjects with Baseline Bleeding
| Number of Subjects with Baseline Bleeding | Severity at Day 7 | ||||
| None | Minor | Mild | Moderate | Missing | |
| Chronic ITP (N=45) | 35 (78%) | 8 (18%) | 0 | 0 | 2 (4%) |
14.2 Treatment of Dermatomyositis
In a prospective, double-blind, randomized, placebo-controlled, multicenter study, 95 adults with dermatomyositis were enrolled and randomized. Forty-seven subjects received Octagam and 48 subjects received placebo in an initial 16week, double-blind First Period, followed by a 6-month, open-label Extension Period, during which all subjects who were eligible to continue, received Octagam 10% every 4 weeks. Forty-five subjects in the initial Octagam group and 46 subjects in the placebo group entered the Extension Period.
Efficacy in this study was based on the proportion of responders at Week 16. A responder was defined as a subject with a minimal improvement of ≥20 points on the Total Improvement Score (TIS). TIS is based on six Core Set Measures (CSM) which include: Manual Muscle Testing MMT-8, Physician Global Disease Activity (GDA) as part of Myositis Disease Activity Assessment Tool (MDAAT) assessed by the Investigator on a Visual Analog Scale (VAS), Extramuscular Activity as part of MDAAT assessed by the Investigator on a VAS, Patient GDA assessed by the Patient on a VAS, Health Assessment Questionnaire (HAQ) assessed by the Patient, and Muscle Enzymes: aldolase, creatine kinase, alanine aminotransferase (ALT), aspartate aminotransferase (AST), lactate dehydrogenase (LDH).
Table 10 shows the proportion of responders in the Octagam 10% and the placebo groups, and the difference in responder rate between the two groups at Week 16. The median time to response was 35 days in the Octagam 10% group. In addition, there was a greater proportion of subjects in the Octagam 10% group compared to placebo with at least moderate improvement defined as ≥40 points on the TIS and major improvement defined as ≥60 points on the TIS (Table 10).
Table 10. Total Improvement Score – Proportion of Responders by Improvement Category at Week 16
|
Total Improvement Score (TIS)
Response Category |
Octagam 10%
N=47 Number of Subjects (%) |
Placebo
N=48 Number of Subjects (%) |
Difference in responder proportion
Octagam 10% – placebo Point Estimate [95% CI], p-value* |
|
At Least Minimal Improvement (TIS ≥ 20) (Primary Efficacy Endpoint) | 37 (78.7%) | 21 (43.8%) |
35.0% [16.7%, 53.2%], 0.0008 |
|
At Least Moderate Improvement (TIS ≥ 40) | 32 (68.1%) | 11 (22.9%) |
45.2% [27.3%, 63.0%] |
|
At Least Major Improvement (TIS ≥ 60) | 15 (32.0%) | 4 (8.3%) |
23.6% [8.1%, 39.0%] |
*Cochran-Mantel-Haenszel Test; CI = Confidence interval
Efficacy was further supported by an improvement in the Cutaneous Dermatomyositis Disease Area and Severity Index (CDASI) total activity score, with a mean decrease of 9.4 (SD: 10.5) points from baseline to Week 16 in the Octagam 10% group versus 1.2 (SD: 7.0) point in the placebo group.
The Octagam 10% group maintained their improvement in TIS (32/45, 71.1%) during the 6-month Extension Period. Among the 46 subjects who switched from placebo to Octagam 10% in the Extension Period, 69.6% (32/46) were classified as responders at the end of the 6-month Extension Period.
15 REFERENCES
- Duhem C, Dicato MA, Ries F: Side-effects of intravenous immune globulins. Clin.Exp.Immunol. 1994;97 Suppl 1:79-83.
- Kannan S, Rowland CH, Hockings GI, et al: Intragam can interfere with blood glucose monitoring. Med J Aust. 2004;180:251-252.
- Steinberger BA, Ford SM, Coleman TA: Intravenous immunoglobulin therapy results in post-infusional hyperproteinemia, increased serum viscosity, and pseudohyponatremia. Am.J Hematol. 2003;73:97-100.
- Dalakas MC: High-dose intravenous immunoglobulin and serum viscosity: risk of precipitating thromboembolic events. Neurology 1994;44:223-226.
- Go RS, Call TG: Deep venous thrombosis of the arm after intravenous immunoglobulin infusion: case report and literature review of intravenous immunoglobulin-related thrombotic complications. Mayo Clin Proc 2000;75:83-85.
- Wolberg AS, Kon RH, Monroe DM, et al: Coagulation factor XI is a contaminant in intravenous immunoglobulin preparations. Am.J.Hematol. 2000;65:30-34.
- Kahwaji J, Barker E, Pepkowitz S, et al: Acute Hemolysis After High-Dose Intravenous Immunoglobulin Therapy in Highly HLA Sensitized Patients. Clinical journal of the American Society of Nephrology : CJASN 2009;DOI:10.2215/CJN.04540709.
- Daw Z, Padmore R, Neurath D, et al: Hemolytic transfusion reactions after administration of intravenous immune (gamma) globulin: a case series analysis. Transfusion 2008;48:1598-1601.
- Sekul EA, Cupler EJ, Dalakas MC: Aseptic meningitis associated with high-dose intravenous immunoglobulin therapy: frequency and risk factors. Ann Intern.Med 1994;121:259-262.
- Rizk A, Gorson KC, Kenney L, et al: Transfusion-related acute lung injury after the infusion of IVIG. Transfusion 2001;41:264-268.
- Auletta CS, Kotkoskie LA, Saulog T, et al: A dietary oncogenicity study of tributyl phosphate in the CD-1 mouse. Toxicology 1998;128:135-141.
- Auletta CS, Weiner ML, Richter WR: A dietary toxicity/oncogenicity study of tributyl phosphate in the rat. Toxicology 1998;128:125-134
16 HOW SUPPLIED/STORAGE AND HANDLING
Octagam 10% is supplied in 2 g, 5 g, 10 g, 20 g or 30 g single use bottles.
| Octagam 10% | |||
| Carton NDC Number | Container NDC Number | Size | Grams Protein |
| 0069-6002-02 | 0069-6002-01 | 20 mL | 2.0 |
| 0069-6550-02 | 0069-6550-01 | 50 mL | 5.0 |
| 0069-6111-02 | 0069-6111-01 | 100 mL | 10.0 |
| 0069-6237-02 | 0069-6237-01 | 200 mL | 20.0 |
| 0069-6339-02 | 0069-6339-01 | 300 mL | 30.0 |
Octagam 10% is not supplied with an infusion set. If a filtered infusion set is used (not mandatory), choose a filter size of 0.2 – 200 microns.
Components used in the packaging of Octagam 10% are not made with natural rubber latex.
Store Octagam 10% for 36 months at +2°C to + 8°C (36°F to 46°F) from the date of manufacture. Within this shelf-life, the product may be stored up to 9 months at ≤ +25°C (77°F). After storage at ≤ +25°C (77°F) the product must be used or discarded.
Do not use after expiration date.
Do not freeze. Do not use frozen product.
Dispose of any unused product or waste material in accordance with local requirements.
17 PATIENT COUNSELING INFORMATION
Inform patients of the signs and symptoms of hypersensitivity reactions including urticaria, generalized urticaria, tightness of the chest, wheezing, hypotension, and anaphylaxis, and to contact their physicians immediately if allergic symptoms occur,
Inform patients to immediately report the signs and symptoms of the following conditions to their physician:
• renal failure, such as decreased urine output, sudden weight gain, fluid retention/edema, and/or shortness of breath
• aseptic meningitis, such as headache, neck stiffness, drowsiness, fever, sensitivity to light, painful eye movements, nausea, and vomiting
• hemolysis, such as fatigue, increased heart rate, yellowing of the skin or eyes, and dark-colored urine
• TRALI, such as troubled breathing, chest pain, blue lips or extremities, and fever. TRALI typically occurs within 1 to 6 hours following transfusion
Thrombosis
Instruct patients to immediately report symptoms of thrombosis. These symptoms may include: pain and/or swelling of an arm or leg with warmth over the affected area, discoloration of an arm or leg, unexplained shortness of breath, chest pain or discomfort that worsens on deep breathing, unexplained rapid pulse, numbness or weakness on one side of the body.
Inform patients that Octagam 10% is made from human plasma and may contain infectious agents that can cause disease (e.g., viruses, and, theoretically, the CJD agent), and that the risk of infectious agent transmission has been reduced by (a) screening plasma donors for prior exposure to certain viruses, (b) testing the donated plasma for certain viral infections and (c) inactivating and/or removing certain viruses during manufacture.
Inform patients that administration of Octagam 10% may interfere with the response to live viral vaccines such as measles, mumps and rubella, and to notify their immunizing physician of their therapy with Octagam 10%.
Manufactured by:
Octapharma Pharmazeutika Produktionsges.m.b.H.
Oberlaaer Strasse 235
A-1100 Vienna, Austria
Octapharma AB
Lars Forssells gata 23
SE- 112 75, Sweden
U.S. License No. 1646