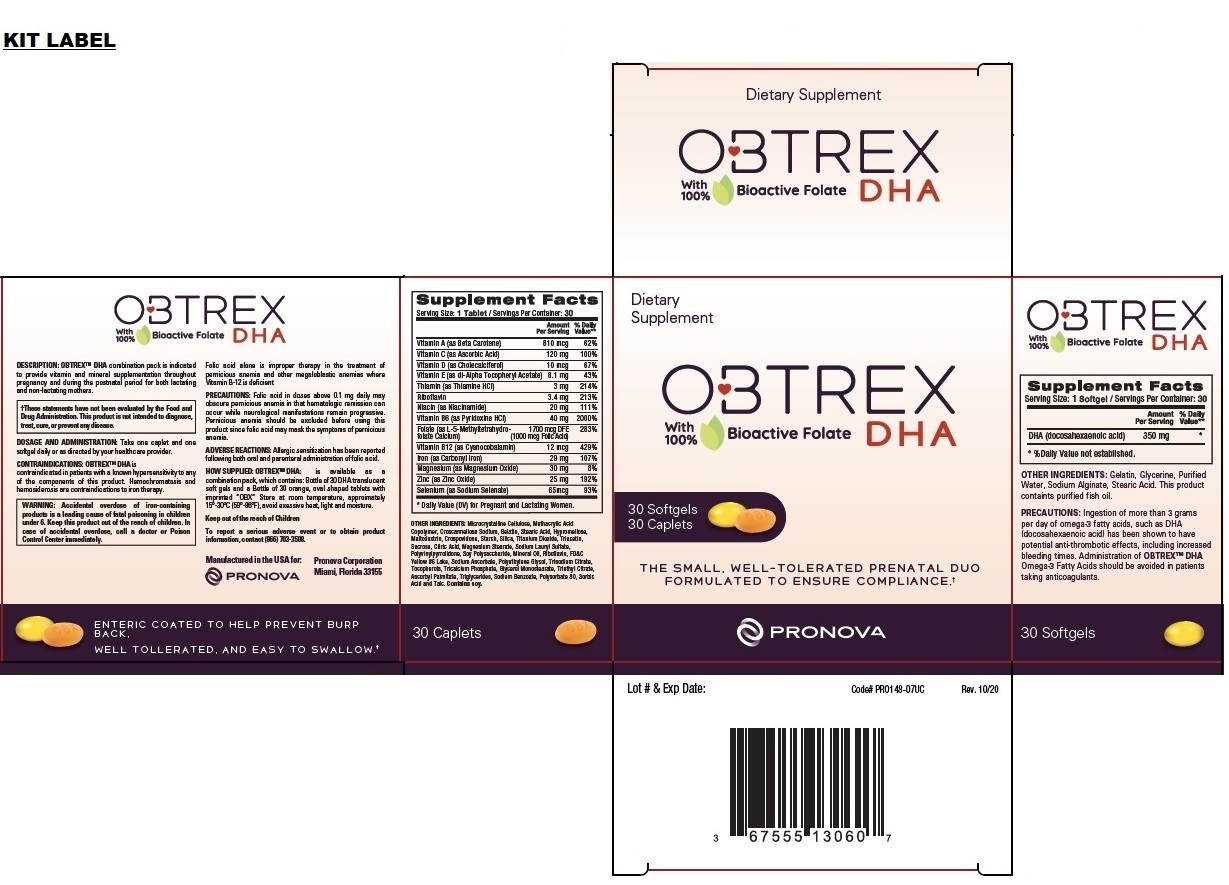
OBTREX DHA Softgels
| Supplement Facts | ||
| Serving Size: 1 Softgel / Servings Per Container: 30 | ||
| Amount | % Daily | |
| Per Serving | Value | |
| DHA (docosahexaenoic acid) | 350 mg | * |
| * % Daily Value not established. |
OTHER INGREDIENTS:
Gelatin, Glycerine, Purified Water, Sodium Alginate, Stearic Acid. This product contains purified fish oil.
OBTREX™ DHA Omega-3 Fatty Acids are manufactured under cGMP, CFIA and HACCP standards and are also in compliance with the Global Organization of DHA Omega-3 standards.
INDICATIONS:
OBTREX™ DHA combination pack is indicated to provide vitamin and mineral supplementation throughout pregnancy and during the postnatal period for both lactating and non-lactating mothers. OBTREX™ DHA combination pack is also useful in improving nutritional status prior to conception.†
| †These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease. |
DOSAGE AND ADMINISTRATION:
One softgel daily; before, during and after pregnancy, with or without food, or as directed by a physician.
PRECAUTIONS:
Ingestion of more than 3 grams per day of omega-3 fatty acids, such as DHA (docosahexaenoic acid) has been shown to have potential anti-thrombotic effects, including increased bleeding times. Administration of OBTREX™ DHA Omega-3 Fatty Acids should be avoided in patients taking anticoagulants.
Sold in combination with OBTREX™ DHA Prenatals
STORAGE:
Store at room temperature, USP
Keep out of the reach of Children
To report a serious adverse event or to obtain product information, contact (866) 703-3508.
Rev 10/20
Dietary Supplement
ENTERIC COATED TO HELP PREVENT BURP BACK
DO NOT USE IF IMPRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING.
OBTREX DHA Caplets
| Supplement Facts | ||
| Serving Size: 1 Caplet / Servings Per Container: 30 | ||
| Amount
Per Serving | % Daily
Value** |
|
| Vitamin A (as Beta Carotene) | 810 mcg | 62% |
| Vitamin C (as Ascorbic Acid) | 120 mg | 100% |
| Vitamin D (as Cholecalciferol) | 10 mcg | 67% |
| Vitamin E (as dl-Alpha Tocopheryl Acetate) | 8.1 mg | 43% |
| Thiamin (as Thiamine HCl) | 3 mg | 214% |
| Riboflavin | 3.4 mg | 213% |
| Niacin (as Niacinamide) | 20 mg | 111% |
| Vitamin B6 (as Pyridoxine HCl) | 40 mg | 2000% |
| Folate (as L-5-Methyltetrahydrofolate Calcium) | 1700 mcg DFE | 283% |
| (1000 mcg Folic Acid) | ||
| Vitamin B12 (as Cyanocobalamin) | 12 mcg | 429% |
| Iron (as Carbonyl Iron) | 29 mg | 107% |
| Magnesium (as Magnesium Oxide) | 30 mg | 8% |
| Zinc (as Zinc Oxide) | 25 mg | 192% |
| Selenium (as Sodium Selenate) | 65mcg | 93% |
| * Daily Value (DV) for Pregnant and Lactating Women. | ||
OTHER INGREDIENTS:
Microcrystalline Cellulose, Methacrylic Acid Copolymer, Croscarmellose Sodium, Gelatin, Stearic Acid, Hypromellose, Maltodextrin, Crospovidone, Starch, Silica, Titanium Dioxide, Triacetin, Sucrose, Citric Acid, Magnesium Stearate, Sodium Lauryl Sulfate, Polyvinylpyrrolidone, Soy Polysaccharide, Mineral Oil, Riboflavin, FD&C Yellow #6 Lake, Sodium Ascorbate, Polyethylene Glycol, Trisodium Citrate, Tocopherols, Tricalcium Phosphate, Glycerol Monostearate, Triethyl Citrate, Ascorbyl Palmitate, Triglycerides, Sodium Benzoate, Polysorbate 80, Sorbic Acid and Talc. Contains soy.
DESCRIPTION:
OBTREX™ DHA combination pack is indicated to provide vitamin and mineral supplementation throughout pregnancy and during the postnatal period for both lactating and non-lactating mothers.
OBTREX™ DHA combination pack is also useful in improving nutritional status prior to conception.†
| †These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease. |
DOSAGE AND ADMINISTRATION:
One caplet daily or as directed by a physician.
CONTRAINDICATIONS:
OBTREX™ DHA is contraindicated in patients with a known hypersensitivity to any of the components of this product. Hemochromatosis and hemosiderosis are contraindications to iron therapy.
| WARNING: Accidental overdose of iron-containing products is a leading cause of fatal poisoning in children under 6. Keep this product out of the reach of children. In case of accidental overdose, call a doctor or Poison Control Center immediately. |
Folic acid alone is improper therapy in the treatment of pernicious anemia and other megaloblastic anemias where Vitamin B-12 is deficient
PRECAUTIONS:
Folic acid in doses above 0.1 mg daily may obscure pernicious anemia in that hematologic remission can occur while neurological manifestations remain progressive. Pernicious anemia should be excluded before using this product since folic acid may mask the symptoms of pernicious anemia.
ADVERSE REACTIONS:
Allergic sensitization has been reported following both oral and parenteral administration of folic acid.
STORAGE:
Store at room temperature, USP.
Keep out of the reach of Children
To report a serious adverse event or to obtain product information, contact (866) 703-3508.
Rev 10/20
Dietary Supplement
with 100% Bioactive Folate
ENTERIC COATED, WELL TOLERATED, AND EASY TO SWALLOW.
DO NOT USE IF IMPRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING.
DOSAGE AND ADMINISTRATION:
Take one caplet and one softgel daily or as directed by your healthcare provider.
HOW SUPPLIED:
OBTREX™ DHA: is available as a combination pack, which contains: Bottle of 30 DHA translucent soft gels and a Bottle of 30 orange, oval shaped tablets with imprinted “OBX” Store at room temperature, approximately 15°-30°C (59°-86°F), avoid excessive heat, light and moisture.