FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
1.1 Treatment of Influenza
XOFLUZA is indicated for the treatment of acute uncomplicated influenza in patients 12 years of age and older who have been symptomatic for no more than 48 hours and who are:
- otherwise healthy, or
- at high risk of developing influenza-related complications1 [see Clinical Studies (14.2)].
1.2 Post-Exposure Prophylaxis of Influenza
XOFLUZA is indicated for post-exposure prophylaxis of influenza in persons 12 years of age and older following contact with an individual who has influenza [see Clinical Studies (14.3)].
1.3 Limitations of Use
Influenza viruses change over time, and factors such as the virus type or subtype, emergence of resistance, or changes in viral virulence could diminish the clinical benefit of antiviral drugs. Consider available information on drug susceptibility patterns for circulating influenza virus strains when deciding whether to use XOFLUZA [see Microbiology (12.4) and Clinical Studies (14)].
2 DOSAGE AND ADMINISTRATION
2.1 Dosage and Administration Overview
XOFLUZA is available in two dosage forms:
- XOFLUZA tablets
- XOFLUZA for oral suspension. This granule formulation is intended for patients who are unable to or have difficulty swallowing tablets, or those who require enteral administration [see Dosage and Administration (2.3)].
XOFLUZA should be taken as soon as possible after influenza symptom onset or exposure to influenza and may be taken with or without food. However, coadministration of XOFLUZA with dairy products, calcium-fortified beverages, polyvalent cation-containing laxatives, antacids, or oral supplements (e.g., calcium, iron, magnesium, selenium, or zinc) should be avoided [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)].
2.2 Recommended Dosage
Treatment of Acute Uncomplicated Influenza or Post-Exposure Prophylaxis in Adults and Adolescents (12 Years of Age and Older)
XOFLUZA should be taken as a single dose as soon as possible and within 48 hours of influenza symptom onset for treatment of acute uncomplicated influenza or following contact with an individual who has influenza. The recommended dosage of XOFLUZA in patients 12 years of age or older is a single weight-based dose displayed in Tables 1 and 2.
| Patient Body Weight (kg) | Recommended Single Oral Dose (Tablets) |
|---|---|
| Less than 80 kg | One 40 mg tablet (blister card contains one 40 mg tablet) |
| At least 80 kg | One 80 mg tablet (blister card contains one 80 mg tablet) |
| Patient Body Weight (kg) | Recommended Single Oral Dose (Suspension) |
|---|---|
| Less than 80 kg | 40 mg/20 mL (1 bottle) taken as a single dose |
| At least 80 kg | 80 mg/40 mL (2 bottles) taken as a single dose |
2.3 Preparation of XOFLUZA for Oral Suspension by Healthcare Provider
Prior to dispensing to the patient, constitute XOFLUZA for oral suspension with 20 mL of drinking water or sterile water. After constitution, each bottle of XOFLUZA suspension contains 40 mg of baloxavir marboxil per 20 mL of volume for a final concentration of 2 mg/mL. This dosage form can be used for oral or enteral use.
Constituting XOFLUZA for Oral Suspension
Prepare the suspension at the time of dispensing. Administration must occur within 10 hours after constitution because the product does not contain a preservative.
- Gently tap the bottom of the bottle to loosen the granules.
- Constitute XOFLUZA for oral suspension with 20 mL of drinking water or sterile water.
- Gently swirl the suspension to ensure that the granules are evenly suspended. Do not shake.
- Write the expiration time and date on the bottle label in the space provided (10 hours from constitution time).
Important Information for the Healthcare Provider
- Provide caregiver or patient with a measuring device (e.g., oral syringe, measuring cup) to deliver the prescribed dose of the suspension for oral use. For enteral administration (i.e., feeding tube), draw up suspension with an enteral syringe. Flush with 1 mL of water before and after enteral administration.
- Instruct the caregiver or patient that the total prescribed dose of XOFLUZA for oral suspension may require more than one bottle (e.g., for adults and adolescents weighing at least 80 kg).
3 DOSAGE FORMS AND STRENGTHS
XOFLUZA Tablets:
XOFLUZA 40 mg tablets are white to light yellow, oblong-shaped, film-coated tablets debossed with "BXM40" on one side.
XOFLUZA 80 mg tablets are white to light yellow, oblong shaped, film-coated tablets debossed with "BXM80" on one side.
XOFLUZA for Oral Suspension:
XOFLUZA for oral suspension contains 40 mg/20 mL or 2 mg/mL baloxavir marboxil after constitution with 20 mL of drinking water or sterile water. The granules are white to light yellow. The constituted product is a white to light yellow opaque suspension with strawberry flavor.
4 CONTRAINDICATIONS
XOFLUZA is contraindicated in patients with a history of hypersensitivity to baloxavir marboxil or any of its ingredients. Serious allergic reactions have included anaphylaxis, angioedema, urticaria, and erythema multiforme [see Warnings and Precautions (5.1)].
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity
Cases of anaphylaxis, urticaria, angioedema, and erythema multiforme have been reported in postmarketing experience with XOFLUZA. Appropriate treatment should be instituted if an allergic-like reaction occurs or is suspected. The use of XOFLUZA is contraindicated in patients with known hypersensitivity to XOFLUZA [see Contraindications (4) and Adverse Reactions (6.2)].
5.2 Risk of Bacterial Infections
There is no evidence of efficacy of XOFLUZA in any illness caused by pathogens other than influenza viruses. Serious bacterial infections may begin with influenza-like symptoms or may coexist with, or occur as, a complication of influenza. XOFLUZA has not been shown to prevent such complications. Prescribers should be alert to potential secondary bacterial infections and treat them as appropriate.
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The overall safety profile of XOFLUZA is based on data from 1,943 subjects 12 years of age and older in 4 controlled clinical trials who received XOFLUZA [see Clinical Studies (14)].
Treatment of Acute Uncomplicated Influenza
Adult and Adolescent Subjects:
The safety of XOFLUZA in adult and adolescent subjects is based on data from 3 placebo-controlled trials in which a total of 1,640 subjects received XOFLUZA: 1,334 (81%) subjects were 18 to 64 years of age, 209 (13%) subjects were adults 65 years of age or older, and 97 (6%) subjects were adolescents 12 to 17 years of age. These trials included otherwise healthy adults and adolescents (N=910) and subjects at high risk of developing complications associated with influenza (N=730). Of these, 1,440 subjects received XOFLUZA at the recommended dose [see Clinical Studies (14.1, 14.2)]. Trial 1 was a phase 2 dose-finding placebo-controlled trial where otherwise healthy adult subjects 20 to 64 years of age received single oral dose of XOFLUZA or placebo. Trial 2 was a placebo- and active-controlled trial in otherwise healthy adults and adolescents 12 to 64 years of age; subjects received weight-based XOFLUZA or placebo as a single oral dose on Day 1 or oseltamivir twice a day for 5 days. Trial 3 was a randomized, double-blind, placebo- and active-controlled trial where adults and adolescents at high risk of influenza complications 12 years of age and older received either XOFLUZA, placebo or oseltamivir.
Table 3 displays the most common adverse events (regardless of causality assessment) reported in at least 1% of adult and adolescent subjects who received XOFLUZA at the recommended dose in Trials 1, 2, and 3.
| Adverse Event | XOFLUZA (N=1,440) | Placebo (N=1,136) |
|---|---|---|
| Diarrhea | 3% | 4% |
| Bronchitis | 3% | 4% |
| Nausea | 2% | 3% |
| Sinusitis | 2% | 3% |
| Headache | 1% | 1% |
Post-Exposure Prophylaxis of Influenza
The safety of XOFLUZA in adult and adolescent subjects is based on data from one placebo-controlled clinical trial in which 374 subjects, of which 303 were adult and adolescent subjects ≥ 12 years, received XOFLUZA: eight (3%) subjects were adults 65 years of age or older, and 12 (4%) subjects were adolescents 12 to 17 years of age. The most frequently reported AE in the total study population was nasopharyngitis which occurred in 6% of subjects who received XOFLUZA and 7% on placebo [see Clinical Studies (14.3)].
6.2 Postmarketing Experience
The following adverse reactions have been identified during postmarketing use of XOFLUZA. Because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to XOFLUZA exposure.
Immune System Disorders: Anaphylactic reactions, anaphylactic shock, anaphylactoid reactions, hypersensitivity reactions, angioedema (swelling of face, eyelids, tongue and lips)
Skin and Subcutaneous Tissue Disorders: Rash, urticaria, erythema multiforme
Gastrointestinal Disorders: Vomiting, hematochezia, melena, colitis
Psychiatric Disorders: Delirium, abnormal behavior, hallucinations
7 DRUG INTERACTIONS
7.1 Effect of Other Drugs on XOFLUZA
Baloxavir may form a chelate with polyvalent cations such as calcium, aluminum, or magnesium. Coadministration with polyvalent cation-containing products may decrease plasma concentrations of baloxavir [see Clinical Pharmacology (12.3)], which may reduce XOFLUZA efficacy. Avoid coadministration of XOFLUZA with dairy products, calcium-fortified beverages, polyvalent cation-containing laxatives, antacids, or oral supplements (e.g., calcium, iron, magnesium, selenium, or zinc).
7.2 Vaccines
The concurrent use of XOFLUZA with intranasal live attenuated influenza vaccine (LAIV) has not been evaluated. Concurrent administration of antiviral drugs may inhibit viral replication of LAIV and thereby decrease the effectiveness of LAIV vaccination. Interactions between inactivated influenza vaccines and XOFLUZA have not been evaluated.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no adequate and well-controlled studies with XOFLUZA in pregnant women to inform a drug-associated risk of adverse developmental outcomes. There are risks to the mother and fetus associated with influenza virus infection in pregnancy [see Clinical Considerations]. In animal reproduction studies, no adverse developmental effects were observed in rats or rabbits with oral administration of baloxavir marboxil at exposures approximately 5 (rats) and 7 (rabbits) times the systemic baloxavir exposure at the maximum recommended human dose (MRHD) [see Data].
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defects, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2%–4% and 15%–20%, respectively.
Clinical Considerations
Disease-Associated Maternal and/or Embryo/Fetal Risk
Pregnant women are at higher risk of severe complications from influenza, which may lead to adverse pregnancy and/or fetal outcomes, including maternal death, stillbirth, birth defects, preterm delivery, low birth weight, and small for gestational age.
Data
Animal Data
Baloxavir marboxil was administered orally to pregnant rats (20, 200, or 1,000 mg/kg/day from gestation day 6 to 17) and rabbits (30, 100, or 1,000 mg/kg/day from gestation day 7 to 19). No adverse embryo-fetal effects were observed in rats up to the highest dose of baloxavir marboxil (1,000 mg/kg/day), resulting in systemic baloxavir exposure (AUC) of approximately 5 times the exposure at the MRHD. In rabbits, fetal skeletal variations occurred at a maternally toxic dose (1,000 mg/kg/day) resulting in 2 abortions out of 19 pregnancies. No adverse maternal or embryo-fetal effects were observed in rabbits at the middle dose (100 mg/kg/day) resulting in systemic baloxavir exposure (AUC) approximately 7 times the exposure at the MRHD.
In the prenatal and postnatal development study in rats, baloxavir marboxil was administered orally at 20, 200, or 1,000 mg/kg/day from gestation day 6 to postpartum/lactation day 20. No significant effects were observed in the offspring at maternal systemic baloxavir exposure (AUC) approximately 5 times the exposure at the MRHD.
8.2 Lactation
Risk Summary
There are no data on the presence of baloxavir marboxil in human milk, the effects on the breastfed infant, or the effects on milk production. Baloxavir and its related metabolites were present in the milk of lactating rats [see Data]. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for XOFLUZA and any potential adverse effects on the breastfed child from the drug or from the underlying maternal condition.
Data
In a lactation study, baloxavir and its related metabolites were excreted in the milk of lactating rats administered baloxavir marboxil (1 mg/kg) on postpartum/lactation day 11, with peak milk concentration approximately 5 times that of maternal plasma concentrations occurring 2 hours post-dose. No effects of baloxavir marboxil on growth and postnatal development were observed in nursing pups at the highest oral dose tested in rats. Maternal systemic exposure was approximately 5 times the baloxavir exposure in humans at the MRHD.
8.4 Pediatric Use
Treatment of Acute Uncomplicated Influenza in Pediatric Subjects
The safety and effectiveness of XOFLUZA for the treatment of acute uncomplicated influenza in pediatric subjects 12 years of age and older weighing at least 40 kg is supported by one randomized, double-blind, controlled trial in otherwise healthy subjects (Trial 2) and one trial in subjects at high risk of developing influenza-related complications (Trial 3) [see Clinical Studies (14.1)]. A total of 117 otherwise healthy adolescents 12-17 years old were randomized and received either XOFLUZA (N=76) or placebo (N=41) in Trial 2; 38 adolescents aged 12 to 17 years at high risk for influenza complications were randomized and received either XOFLUZA (N=21) or placebo (N=17) in Trial 3. The median time to alleviation of symptoms in influenza-infected adolescent subjects aged 12 to 17 years in Trial 2 was comparable to that observed in adults. In Trial 3, the median time to improvement of symptoms in the limited number of influenza-infected adolescent subjects aged 12 to 17 years was similar in the XOFLUZA and placebo arms [see Clinical Studies (14.1)]. Adverse events reported in adolescents in both trials were similar to those reported in adults [see Adverse Reactions (6.1)].
The safety and efficacy of XOFLUZA in pediatric subjects less than 12 years of age have not been established for the treatment of acute uncomplicated influenza. For information on baloxavir resistance in subjects less than 12 years of age [see Microbiology (12.4)].
Post-Exposure Prophylaxis of Influenza in Pediatric Subjects
The safety and effectiveness of XOFLUZA for post-exposure prophylaxis in adolescents (12 years to < 18 years) is supported by one randomized, double-blind, controlled trial conducted in Japan (Trial 4) [see Clinical Studies (14.3)]. Subjects in this trial were randomized in a 1:1 ratio to receive XOFLUZA or placebo. A total of 12 subjects from ≥ 12 to < 18 years of age received XOFLUZA. The incidence of RT-PCR-confirmed symptomatic influenza was similar in the XOFLUZA and placebo arms in the limited number of adolescent subjects aged 12 to 17 years of age [see Clinical Studies (14.3)]. Adverse events reported in adolescent subjects were similar to those reported in adults in the same trial [see Adverse Reactions (6.1)].
The safety and efficacy of XOFLUZA for post-exposure prophylaxis of influenza in pediatric subjects less than 12 years of age have not been established.
8.5 Geriatric Use
The safety and effectiveness of XOFLUZA in subjects 65 years of age and older has been established and is supported by one randomized, double-blind, controlled trial [see Clinical Studies (14.2)]. In Trial 3, of 730 XOFLUZA-treated subjects at high risk of influenza-related complications, 209 (29%) subjects were 65 years of age and older. The median time to improvement of influenza symptoms in subjects 65 years of age and older was 70 hours in subjects who received XOFLUZA (N=112) and 88 hours in those who received placebo (N=102). The safety profile observed for this population was similar to that reported in the overall trial population except for nausea, which was reported in 6% of elderly subjects compared to 1% of subjects from 18 to 64 years of age.
10 OVERDOSAGE
Treatment of an overdose of XOFLUZA should consist of general supportive measures, including monitoring of vital signs and observation of the clinical status of the patient. There is no specific antidote for overdose with XOFLUZA.
Baloxavir is unlikely to be significantly removed by dialysis due to high serum protein binding [see Clinical Pharmacology (12.3)].
11 DESCRIPTION
XOFLUZA (baloxavir marboxil) is an antiviral PA endonuclease inhibitor.
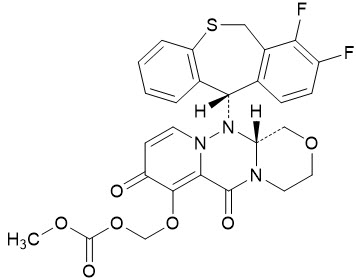
The active component of XOFLUZA is baloxavir marboxil. The chemical name of baloxavir marboxil is ({(12aR)-12-[(11S)-7,8-Difluoro-6,11-dihydrodibenzo[b,e]thiepin-11-yl]-6,8-dioxo-3,4,6,8,12,12a-hexahydro-1H-[1,4]oxazino[3,4-c]pyrido[2,1-f][1,2,4]triazin-7-yl}oxy)methyl methyl carbonate. The empirical formula of baloxavir marboxil is C27H23F2N3O7S, and the chemical structure is shown below.
Baloxavir marboxil has a molecular mass of 571.55 grams per mole and a partition coefficient (log P) of 2.26. It is freely soluble in dimethylsulfoxide, soluble in acetonitrile, slightly soluble in methanol and ethanol, and practically insoluble in water.
XOFLUZA is supplied as tablets and as granules for oral suspension:
XOFLUZA tablets are white to light yellow, film-coated tablets for oral administration. The inactive ingredients of XOFLUZA tablets are: croscarmellose sodium, hypromellose, lactose monohydrate, microcrystalline cellulose, povidone, sodium stearyl fumarate, talc, and titanium dioxide.
XOFLUZA for oral suspension is supplied as white to light yellow granules in an amber glass bottle. Each bottle contains 40 mg (nominal) of baloxavir marboxil. The granules must be constituted with 20 mL of drinking water or sterile water to yield a 2 mg/mL suspension with strawberry flavor. The inactive ingredients are: colloidal silicon dioxide, hypromellose, maltitol, mannitol, povidone K25, sodium chloride, strawberry flavor, sucralose and talc.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Baloxavir marboxil is an antiviral drug with activity against influenza virus [see Microbiology (12.4)].
12.2 Pharmacodynamics
Cardiac Electrophysiology
At twice the expected exposure from recommended dosing, XOFLUZA did not prolong the QTc interval.
Exposure-Response Relationships
When XOFLUZA is dosed by weight, as recommended (40 mg in patients weighing 40–80 kg; 80 mg in patients weighing at least 80 kg), no difference in baloxavir exposure-response (time to alleviation of influenza symptoms in the otherwise healthy population or time to improvement of influenza symptoms in the high risk population) relationship has been observed.
12.3 Pharmacokinetics
Baloxavir marboxil is a prodrug that is almost completely converted to its active metabolite, baloxavir, following oral administration.
Baloxavir pharmacokinetic parameters are presented for healthy adults and adolescents as the mean [% coefficient of variation (%CV)], unless otherwise specified, in Table 4.
| Pharmacokinetic Parameters of Plasma Baloxavir in Adults and Adolescents* | XOFLUZA dose 40 mg | XOFLUZA dose 80 mg |
|---|---|---|
|
||
| AUC (ng∙hr/mL) | 5520 (46.3%) | 6930 (48.6%) |
| Cmax (ng/mL) | 68.9 (44.9%) | 82.5 (43.0%) |
| C24 (ng/mL) | 50.9 (45.8%) | 62.6 (45.9%) |
| C72 (ng/mL) | 24.2 (45.5%) | 30.8 (47.0%) |
| Absorption | |
| Tmax (hr)* | 4 |
| Effect of food (relative to fasting)† | Cmax: ↓48%, AUC0-inf: ↓36% |
| Distribution | |
| % bound to human serum proteins‡ | 92.9–93.9 |
| Ratio of blood cell to blood | 48.5%–54.4% |
| Volume of distribution (V/F, L)§ | 1180 (20.8%) |
| Elimination | |
| Clearance (CL/F, L/hr) | 10.3 (22.5%) |
| Apparent terminal elimination half-life (hr) | 79.1 (22.4%) |
| Metabolism | |
| Metabolic pathways | Primary: UGT1A3 Secondary: CYP3A4 |
| Excretion | |
| % of dose excreted¶ | Urine: 14.7 (total radioactivity); 3.3 (baloxavir) Feces: 80.1 (total radioactivity) |
No clinically significant differences in the pharmacokinetics of baloxavir were observed based on age, sex, creatinine clearance (CrCl: 50 mL/min and above), or moderate hepatic impairment (Child-Pugh class B). The effect of severe renal or hepatic impairment on baloxavir pharmacokinetics has not been evaluated.
Body Weight
Baloxavir exposure decreases as body weight increases. No clinically significant difference in exposure was observed between body weight groups following the approved recommended dosage.
Race/Ethnicity
Based on a population pharmacokinetic analysis, baloxavir exposure is approximately 35% lower in non-Asians as compared to Asians; this difference is not considered clinically significant when the recommended dose was administered.
Drug Interaction Studies
Clinical Studies
No clinically significant changes in the pharmacokinetics of baloxavir marboxil and its active metabolite, baloxavir, were observed when coadministered with itraconazole (combined strong CYP3A and P-gp inhibitor), probenecid (UGT inhibitor), or oseltamivir.
No clinically significant changes in the pharmacokinetics of the following drugs were observed when coadministered with baloxavir marboxil: midazolam (CYP3A4 substrate), digoxin (P-gp substrate), rosuvastatin (BCRP substrate), or oseltamivir.
In Vitro Studies
Cytochrome P450 (CYP) Enzymes: Both baloxavir marboxil and baloxavir do not inhibit CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, or CYP2D6 and do not induce CYP1A2, CYP2B6, or CYP3A4.
Uridine diphosphate (UDP)-glucuronosyl transferase (UGT) Enzymes: Both baloxavir marboxil and baloxavir do not inhibit UGT1A1, UGT1A3, UGT1A4, UGT1A6, UGT1A9, UGT2B7, or UGT2B15.
Transporter Systems: Both baloxavir marboxil and baloxavir are substrates of P-glycoprotein (P-gp). Baloxavir does not inhibit organic anion-transporting polypeptides (OATP) 1B1, OATP1B3, organic cation transporter (OCT) 1, OCT2, organic anion transporter (OAT) 1, OAT3, multidrug and toxin extrusion (MATE) 1, or MATE2K.
12.4 Microbiology
Mechanism of Action
Baloxavir marboxil is a prodrug that is converted by hydrolysis to baloxavir, the active form that exerts anti-influenza virus activity. Baloxavir inhibits the endonuclease activity of the polymerase acidic (PA) protein, an influenza virus-specific enzyme in the viral RNA polymerase complex required for viral gene transcription, resulting in inhibition of influenza virus replication. The 50% inhibitory concentration (IC50) values of baloxavir ranged from 1.4 to 3.1 nM (n=4) for influenza A viruses and 4.5 to 8.9 nM (n=3) for influenza B viruses in a PA endonuclease assay. Viruses with reduced susceptibility to baloxavir have amino acid substitutions in the PA protein.
Antiviral Activity
The antiviral activity of baloxavir against laboratory strains and clinical isolates of influenza A and B viruses was determined in an MDCK cell-based plaque reduction assay. The median 50% effective concentration (EC50) values of baloxavir were 0.73 nM (n=31; range: 0.20–1.85 nM) for subtype A/H1N1 strains, 0.83 nM (n=33; range: 0.35–2.63 nM) for subtype A/H3N2 strains, and 5.97 nM (n=30; range: 2.67–14.23 nM) for type B strains. In an MDCK cell-based virus titer reduction assay, the 90% effective concentration (EC90) values of baloxavir against avian subtypes A/H5N1 and A/H7N9 were in the range of 0.80 to 3.16 nM. The relationship between antiviral activity in cell culture and clinical response to treatment in humans has not been established.
Resistance
Cell culture: Influenza A virus isolates with reduced susceptibility to baloxavir were selected by serial passage of virus in cell culture in the presence of increasing concentrations of baloxavir. Reduced susceptibility of influenza A virus to baloxavir was conferred by amino acid substitutions I38T (A/H1N1 and A/H3N2) and E199G (A/H3N2) in the PA protein of the viral RNA polymerase complex.
Clinical studies: Influenza A and B viruses with treatment-emergent amino acid substitutions at positions associated with reduced susceptibility to baloxavir in cell culture were observed in clinical studies (Table 6). In adult and adolescent subjects who had a confirmed influenza virus infection, the overall frequencies of treatment-emergent amino acid substitutions associated with reduced susceptibility to baloxavir were 4.5% (6/134), 10.9% (53/485), and 0.9% (2/224) in influenza A/H1N1, A/H3N2, and B virus infections, respectively, in pooled data from Trials 1, 2, and 3 [see Clinical Studies (14)]. In Trial 4, of 374 subjects, including 71 subjects <12 years of age, who received XOFLUZA post-exposure prophylaxis, 49 were viral RNA-positive post-baseline, including 31 subjects who were evaluated for resistance. Of these 31 subjects, influenza virus with substitutions associated with reduced susceptibility to baloxavir was identified in 7/7 subjects who developed clinical influenza (as described for the primary endpoint) and 8/24 other subjects evaluated who did not meet the primary endpoint definition for clinical influenza [see Clinical Studies (14)]. Selection of influenza viruses with reduced susceptibility to baloxavir has occurred at higher frequencies in pediatric subjects, and such viruses were detected with overall frequencies of 20% (4/20), 27.9% (34/122), and 0% (0/21) in influenza A/H1N1, A/H3N2, and B virus infections, respectively, in pooled data from 3 pediatric treatment trials in subjects <12 years of age.
| Influenza Type/Subtype | A/H1N1 | A/H3N2 | B |
|---|---|---|---|
| Amino Acid Substitution | E23K/R, I38F/N/S/T | E23G/K, A37T, I38M/T, E199G | I38T |
None of the treatment-emergent substitutions associated with reduced susceptibility to baloxavir were identified in virus from pretreatment respiratory specimens in the clinical studies. Strains containing substitutions known to be associated with reduced susceptibility to baloxavir were identified in approximately 0.05% of PA sequences in the National Center for Biotechnology Information/GenBank database (queried August 2018).
Prescribers should consider currently available surveillance information on influenza virus drug susceptibility patterns and treatment effects when deciding whether to use XOFLUZA.
Cross-Resistance
Cross-resistance between baloxavir and neuraminidase (NA) inhibitors, or between baloxavir and M2 proton pump inhibitors (adamantanes), is not expected because these drugs target different viral proteins. Baloxavir is active against NA inhibitor-resistant strains, including A/H1N1 and A/H5N1 viruses with the NA substitution H275Y (A/H1N1 numbering), A/H3N2 virus with the NA substitutions E119V or R292K, A/H7N9 virus with the NA substitution R292K (A/H3N2 numbering), and type B virus with the NA substitutions R152K or D198E (A/H3N2 numbering). The NA inhibitor oseltamivir is active against viruses with reduced susceptibility to baloxavir, including A/H1N1 virus with PA substitutions E23K or I38F/T, A/H3N2 virus with PA substitutions E23G/K, A37T, I38M/T, or E199G, and type B virus with the PA substitution I38T. Influenza virus may carry amino acid substitutions in PA that reduce susceptibility to baloxavir and at the same time carry resistance-associated substitutions for NA inhibitors and M2 proton pump inhibitors. The clinical relevance of phenotypic cross-resistance evaluations has not been established.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Mutagenesis
Baloxavir marboxil and the active metabolite, baloxavir, were not mutagenic in in vitro and in in vivo genotoxicity assays, which included bacterial mutation assays in S. typhimurium and E. coli, micronucleus tests with cultured mammalian cells, and in the rodent micronucleus assay.
Impairment of Fertility
In a fertility and early embryonic development study in rats, doses of baloxavir marboxil at 20, 200, or 1,000 mg/kg/day were administered to females for 2 weeks before mating, during mating, and until day 7 of pregnancy. Males were dosed for 4 weeks before mating and throughout mating. There were no effects on fertility, mating performance, or early embryonic development at any dose level, resulting in systemic drug exposure (AUC) approximately 5 times the MRHD.
14 CLINICAL STUDIES
14.1 Treatment of Acute Uncomplicated Influenza—Otherwise Healthy Subjects
Adults and Adolescents (Aged 12 Years and Older)
Two randomized, controlled, double-blinded clinical trials conducted in two different influenza seasons evaluated efficacy and safety of XOFLUZA in otherwise healthy subjects with acute uncomplicated influenza.
In Trial 1, a placebo-controlled phase 2 dose-finding trial, a single oral dose of XOFLUZA was compared with placebo in 400 adult subjects 20 to 64 years of age in Japan. All subjects in Trial 1 were Asian, the majority of subjects were male (62%), and the mean age was 38 years. In this trial, among subjects who received XOFLUZA and had influenza virus typed, influenza A/H1N1 was the predominant strain (63%), followed by influenza B (25%), and influenza A/H3N2 (12%).
In Trial 2 (NCT02954354), a phase 3, randomized, double-blind, active- and placebo-controlled trial, XOFLUZA was studied in 1,436 otherwise healthy adults and adolescents with signs and symptoms of influenza in the U.S. and Japan. Subjects were 12 to 64 years of age and weighed at least 40 kg. Adults ages 20 to 64 years received weight-based XOFLUZA (subjects who weighed 40 to less than 80 kg received 40 mg and subjects who weighed 80 kg and above received 80 mg) (N=612) or placebo as a single oral dose on day 1 (N=310) or oseltamivir twice a day for 5 days (N=514). Subjects in the XOFLUZA and placebo arms received a placebo for the duration of oseltamivir dosing after XOFLUZA or placebo dosing in that arm. Adolescent subjects 12 to less than 20 years of age received weight-based XOFLUZA or placebo as a single oral dose.
Seventy-eight percent of subjects in Trial 2 were Asian, 17% were White, and 4% were Black or African American. The mean age was 34 years, and 11% of subjects were less than 20 years of age; 54% of subjects were male and 46% female. In Trial 2, 1,062 of 1,436 enrolled subjects had influenza confirmed by RT-PCR and were included in the efficacy analysis (XOFLUZA N=455, placebo N=230, or oseltamivir N=377). Among subjects who received XOFLUZA and had influenza virus typed, influenza A/H3N2 was the predominant strain (90%), followed by influenza B (9%), and influenza A/H1N1 (2%).
In both Trials 1 and 2, eligible subjects had an axillary temperature of at least 38˚C, at least one moderate or severe respiratory symptom (cough, nasal congestion, or sore throat), and at least one moderate or severe systemic symptom (headache, feverishness or chills, muscle or joint pain, or fatigue), and all were treated within 48 hours of symptom onset. Subjects participating in the trial were required to self-assess their influenza symptoms as "none," "mild," "moderate," or "severe" twice daily. The primary efficacy population was defined as those with a positive rapid influenza diagnostic test (Trial 1) or positive influenza reverse transcription polymerase chain reaction (RT-PCR) (Trial 2) at trial entry.
The primary endpoint of both trials, time to alleviation of symptoms, was defined as the time when all seven symptoms (cough, sore throat, nasal congestion, headache, feverishness, myalgia, and fatigue) had been assessed by the subject as none or mild for a duration of at least 21.5 hours.
In both trials, XOFLUZA treatment at the recommended dose resulted in a statistically significant shorter time to alleviation of symptoms compared with placebo in the primary efficacy population (Tables 7 and 8).
| XOFLUZA 40 mg (95% CI*) N=100 | Placebo (95% CI*) N=100 |
|
|---|---|---|
|
||
| Adults (20 to 64 Years of Age) | 50 hours†
(45, 64) | 78 hours (68, 89) |
| XOFLUZA 40 mg or 80 mg (95% CI*) N=455 | Placebo (95% CI*) N=230 |
|
|---|---|---|
| Subjects (≥ 12 Years of Age) | 54 hours†
(50, 59) | 80 hours (73, 87) |
In Trial 2, there was no difference in the time to alleviation of symptoms between subjects (age ≥ 20 years) who received XOFLUZA (54 hours) and those who received oseltamivir (54 hours). For adolescent subjects (12 to 17 years of age) in Trial 2, the median time to alleviation of symptoms for subjects infected with influenza and who received XOFLUZA (N=63) was 54 hours (95% CI of 43, 81) compared to 93 hours (95% CI of 64, 118) in the placebo arm (N=27).
The number of subjects who received XOFLUZA at the recommended dose and who were infected with influenza type B virus was limited, including 24 subjects in Trial 1 and 38 subjects in Trial 2. In the influenza B subset in Trial 1, the median time to alleviation of symptoms in subjects who received 40 mg XOFLUZA was 63 hours (95% CI of 43, 70) compared to 83 hours (95% CI of 58, 93) in subjects who received placebo. In the influenza B subset in Trial 2, the median time to alleviation of symptoms in subjects who received 40 mg or 80 mg XOFLUZA was 93 hours (95% CI of 53, 135) compared to 77 hours (95% CI of 47, 189) in subjects who received placebo.
14.2 Treatment of Acute Uncomplicated Influenza—High Risk Subjects
Trial 3 (NCT02949011) was a randomized, double-blind, placebo- and active-controlled trial to evaluate the efficacy and safety of a single oral dose of XOFLUZA compared with placebo or oseltamivir in adult and adolescent subjects 12 years of age or older with influenza who were at high risk of developing influenza-related complications.
A total of 2,182 subjects with signs and symptoms of influenza were randomized to receive a single oral dose of 40 mg or 80 mg of XOFLUZA according to body weight (subjects who weighed 40 to less than 80 kg received 40 mg and subjects who weighed 80 kg and above received 80 mg) (N=729), oseltamivir 75 mg twice daily for 5 days (N=725), or placebo (N=728). Twenty-eight percent of subjects were Asian, 59% were White, and 10% were Black or African American. The mean age was 52 years, and 3% of subjects were less than 18 years of age; 43% of subjects were male and 57% female.
High risk factors were based on the Centers for Disease Control and Prevention definition1 of health factors known to increase the risk of developing serious complications from influenza. The majority of subjects had underlying asthma or chronic lung disease, diabetes, heart disease, morbid obesity, or were 65 years of age or older.
In Trial 3, 1,158 of the 2,182 enrolled subjects had influenza confirmed by RT-PCR and were included in the efficacy analysis (XOFLUZA N=385, placebo N=385, or oseltamivir N=388). Among subjects in whom only one type/subtype of influenza virus was identified, 50% were infected with subtype A/H3N2, 43% were infected with type B, and 7% were infected with subtype A/H1N1.
Eligible subjects had an axillary temperature of at least 38°C, at least one moderate or severe respiratory symptom (cough, nasal congestion, or sore throat), and at least one moderate or severe systemic symptom (headache, feverishness or chills, muscle or joint pain, or fatigue), and all were treated within 48 hours of symptom onset. Subjects participating in the trial were required to self-assess their influenza symptoms as "none," "mild," "moderate," or "severe" twice daily. A total of 215 subjects (19%) had preexisting symptoms (cough, muscle or joint pain, or fatigue) associated with their underlying high risk condition that were worsened due to influenza infection. The primary efficacy endpoint was time to improvement of influenza symptoms (cough, sore throat, headache, nasal congestion, feverishness or chills, muscle or joint pain, and fatigue). This endpoint included alleviation of new symptoms and improvement of any preexisting symptoms that had worsened due to influenza. A statistically significant improvement in the primary endpoint was observed for XOFLUZA when compared with placebo (see Table 9).
| XOFLUZA 40 mg or 80 mg*
(95% CI†) N=385 | Placebo (95% CI†) N=385 |
|---|---|
| 73 hours‡
(67, 85) | 102 hours‡
(93, 113) |
There was no statistically significant difference in the median time to improvement of influenza symptoms in the subjects who received XOFLUZA (73 hours) and those who received oseltamivir (81 hours). The median time to improvement of influenza symptoms in the limited number of adolescent subjects aged 12 to 17 years infected with influenza virus was similar for subjects who received XOFLUZA (188 hours) or placebo (191 hours) (N=13 and N=12, respectively).
For subjects infected with type B virus, the median time to improvement of influenza symptoms was 75 hours in the XOFLUZA group (95% CI of 67, 90) compared to 101 hours in the placebo group (95% CI of 83, 116).
14.3 Post-Exposure Prophylaxis of Influenza
Trial 4 was a phase 3, randomized, double-blind, multicenter, placebo-controlled study designed to evaluate the efficacy of a single oral dose of XOFLUZA compared with placebo in the prevention of influenza in subjects who were household contacts of influenza-infected patients in Japan. Influenza-infected index patients were required to have onset of symptoms for ≤ 48 hours, and subjects (household contacts) were required to have lived with the influenza-infected index patient for ≥ 48 hours.
A total of 607 subjects (XOFLUZA N=303, placebo N=304) ≥ 12 years of age were randomized and received a single oral dose of XOFLUZA according to body weight and age, or placebo, on day 1. Subjects received 40 mg or 80 mg of XOFLUZA according to body weight (40 to < 80 kg or ≥ 80 kg, respectively). The primary efficacy endpoint was the proportion of household subjects who were infected with influenza virus and presented with fever and at least one respiratory symptom from day 1 to day 10. Influenza infection was confirmed by RT-PCR, fever was defined as a body temperature (axillary) ≥ 37.5°C, and respiratory symptoms were defined as having a symptom of "cough" or "nasal discharge/nasal congestion" with a severity of moderate or severe as assessed by the subject.
The mean age of subjects that were ≥ 12 years of age in Trial 4 was 40 years; 33 (5%) were ≥ 12 to < 18 years of age, 551 (91%) were ≥ 18 to < 65 years of age, and 23 (4%) were ≥ 65 years of age. All subjects were Asian, 84% were female, and 16% were male. The predominant influenza virus strains in the index patients of this study were the A/H3N2 subtype (49%) and the A/H1N1 subtype (46%), followed by the B subtype (1%).
In subjects that were ≥12 years of age, there was a statistically significant reduction in the proportion of household contacts (subjects) with laboratory-confirmed clinical influenza from 13% in the placebo group to 1% in the XOFLUZA group (see Table 10).
| XOFLUZA (95% CI*) N=303 | Placebo (95% CI*) N=304 |
|---|---|
|
|
| 1% (0, 3) | 13% (10, 17) |
15 REFERENCES
- "People at High Risk For Flu Complications." CDC. https://www.cdc.gov/flu/highrisk/index.htm.
16 HOW SUPPLIED/STORAGE AND HANDLING
Product: 50090-4102
NDC: 50090-4102-0 2 TABLET, FILM COATED in a BLISTER PACK / 1 in a CARTON
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Important Dosing Information
Instruct patients to begin treatment with XOFLUZA as soon as possible at the first appearance of influenza symptoms, within 48 hours of onset of symptoms. Instruct patients to start taking XOFLUZA for prevention as soon as possible after exposure [see Dosage and Administration (2)].
XOFLUZA can be taken with or without food, but advise patients not to take with dairy products, calcium-fortified beverages, polyvalent cation-containing laxatives, antacids, or oral supplements (e.g., calcium, iron, magnesium, selenium, or zinc) [see Dosage and Administration (2) and Drug Interactions (7.1)].
Advise patients to follow the healthcare provider's dosing recommendation for a single, one-time dose of XOFLUZA. XOFLUZA is dosed based on weight and is available in tablet form or as for oral suspension for oral or enteral use.
Tablets: Provided as a blister card containing either one tablet of 40 mg, or one tablet of 80 mg as a single dose [see Dosage and Administration (2.2)].
For oral suspension: Provided in a bottle containing 40 mg/20 mL (2 mg/mL) after constitution by the healthcare provider [see How Supplied/Storage and Handling (16)]. The total prescribed dose of XOFLUZA for oral suspension may require more than one bottle. The suspension should be taken as soon as possible but no later than 10 hours after constitution by the healthcare provider because the product does not contain a preservative [see Dosage and Administration (2.3)].
Hypersensitivity
Advise patients and/or caregivers of the risk of severe allergic reactions such as anaphylaxis, angioedema, urticaria, and erythema multiforme. Instruct patients and/or caregivers to seek immediate medical attention if an allergic-like reaction occurs or is suspected [see Contraindications (4) and Warnings and Precautions (5.1)].
Influenza Vaccines
Because of the potential for antivirals to decrease the effectiveness of live attenuated influenza vaccines, advise patients to consult their healthcare provider prior to receiving a live attenuated influenza vaccine after taking XOFLUZA [see Drug Interactions (7.2)].
Distributed by:
Genentech USA, Inc.
A Member of the Roche Group
1 DNA Way
South San Francisco, CA 94080-4990
XOFLUZA® is a registered trademark of Genentech, Inc.
© 2021 Genentech USA, Inc.
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 3/2021 | |||
| PATIENT INFORMATION | ||||
| XOFLUZA® (zoh-FLEW-zuh) (baloxavir marboxil) tablets | XOFLUZA® (zoh-FLEW-zuh) (baloxavir marboxil) for oral suspension |
|||
| What is XOFLUZA?
XOFLUZA is a prescription medicine used to:
XOFLUZA does not treat or prevent illness that is caused by infections other than the influenza virus. XOFLUZA does not prevent bacterial infections that may happen with the flu. |
||||
| Do not take XOFLUZA if you are allergic to baloxavir marboxil or any of the ingredients in XOFLUZA. See the end of this leaflet for a complete list of ingredients in XOFLUZA. | ||||
Before you take XOFLUZA, tell your healthcare provider about all of your medical conditions, including if you:
Talk to your healthcare provider before you receive a live flu vaccine after taking XOFLUZA. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
||||
How should I take XOFLUZA?
Step 1. Swirl the XOFLUZA for oral suspension bottle well before each use. Do not shake. Step 2. Open the bottle by pushing downward on the child resistant bottle cap and twisting it in the direction of the arrow. Step 3. Measure the oral suspension with the measuring device provided by the pharmacist to be sure you give the prescribed dose. Step 4. The patient should sit in an upright position when taking XOFLUZA.
Step 6. Close the bottle. Throw away any remaining oral suspension and the measuring device. |
||||
| What are the possible side effects of XOFLUZA? XOFLUZA may cause serious side effects, including:
|
||||
|
|
|||
| The most common side effects of XOFLUZA for treatment of the flu in adults and adolescents include: | ||||
|
|
|||
| XOFLUZA is not effective in treating or preventing infections other than influenza. Other kinds of infections can appear like flu or occur along with flu and may need different kinds of treatment. Tell your healthcare provider if you feel worse or develop new symptoms during or after treatment with XOFLUZA or if your flu symptoms do not start to get better. These are not all the possible side effects of XOFLUZA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||
| How should I store XOFLUZA? XOFLUZA tablets:
|
||||
| General information about the safe and effective use of XOFLUZA.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use XOFLUZA for a condition for which it was not prescribed. Do not give XOFLUZA to other people, even if they have the same symptoms that you have. It may harm them. You can ask for information about XOFLUZA that is written for health professionals. |
||||
| What are the ingredients in XOFLUZA? Active ingredient: baloxavir marboxil XOFLUZA tablets inactive ingredients: croscarmellose sodium, hypromellose, lactose monohydrate, microcrystalline cellulose, povidone, sodium stearyl fumarate, talc, and titanium dioxide. XOFLUZA for oral suspension inactive ingredients: colloidal silicon dioxide, hypromellose, maltitol, mannitol, povidone K25, sodium chloride, strawberry flavor, sucralose and talc. Distributed by: Genentech USA, Inc., A Member of the Roche Group, 1 DNA Way, South San Francisco, CA 94080-4990 XOFLUZA® is a registered trademark of Genentech, Inc. © 2021 Genentech USA, Inc. For more information, go to www.XOFLUZA.com or call 1-855-XOFLUZA (1-855-963-5892). |
||||
Representative sample of labeling (see the HOW SUPPLIED section for complete listing):
