FULL PRESCRIBING INFORMATION
WARNING: SEVERE BRONCHOCONSTRICTION
WARNING: SEVERE BRONCHOCONSTRICTION
Severe bronchoconstriction can result from Provocholine administration (including the lowest dose). The use of Provocholine is contraindicated in pediatric and adult patients with baseline FEV1 < 60% predicted or adults with FEV1 < 1.5 L. Because of the potential for severe bronchoconstriction, the use of Provocholine in patients with clinically apparent asthma or wheezing is not recommended [see Warnings and Precautions (5.1)].
Emergency equipment and medication should be immediately available to treat acute respiratory distress. If severe bronchoconstriction occurs, reverse immediately with a rapid-acting inhaled bronchodilator agent (β-agonist) [see Warnings and Precautions (5.1)].
If baseline spirometry is not performed or is measured inaccurately, the initial FEV1 may be underestimated. In this situation, decreases in FEV1 may not be detected after administration of escalating Provocholine doses, which may result in administration of unnecessary higher doses and an increased risk for excessive bronchoconstriction [see Warnings and Precautions (5.1)].
1 INDICATIONS AND USAGE
Provocholine, used in a methacholine challenge test, is indicated for the diagnosis of bronchial airway hyperreactivity in adults and pediatric patients five years of age and older who do not have clinically apparent asthma.
2 DOSAGE AND ADMINISTRATION
2.1 Methacholine Challenge Test Overview
- Provocholine should be administered in a methacholine challenge test in a pulmonary function laboratory or clinic, by adequately trained personnel, for safety and accuracy, and should be performed only under the responsibility of a healthcare practitioner trained in and thoroughly familiar with all aspects of the technique of the test and the management of respiratory distress. Emergency medication and equipment should be immediately available to treat acute respiratory distress.
- Only consider Provocholine use in patients on chronic asthma drugs if the accuracy of the asthma diagnosis is in doubt. In these patients, only administer Provocholine if spirometry is normal after supervised withdrawal of the asthma drugs.
- Provocholine is not recommended for use in patients with clinically apparent asthma or wheezing.
- Before starting a methacholine challenge test, baseline spirometry must be performed. For a patient to be able to undergo the test, he or she must present with baseline FEV1 (Forced Expiratory Volume in 1 second) greater than or equal to 60% of the predicted value (in adults and children) and greater than or equal to 1.5 L (in adults).
- Do not use Provocholine in pediatric and adult patients with baseline FEV1 < 60% predicted or in adults with FEV1 < 1.5 L [see Contraindications (4) and Warnings and Precautions (5.1)].
- At commencement of the methacholine challenge test and prior to nebulization with Provocholine dose(s), FEV1 must be measured following exposure to nebulized diluent or base solution (contains no methacholine chloride) to obtain the post-diluent FEV1 [see Dosage and Administration (2.4, 2.5)].
- Provocholine powder for inhalation solution requires reconstitution and dilution before use (see Reconstitution and Dilution Prior to Administration (2.3)).
- Provocholine inhalation solution (in a ready-to-use kit) does not require reconstitution and/or dilution.
- Administer Provocholine by oral inhalation using either the 5-Breath Dosimeter Dosing Method or the 2-Minute Tidal Breathing Dosing Method with the doubling or quadrupling stepwise protocols [see Dosage and Administration (2.4, 2.5)].
- Discard any unused solution from the nebulizer after each administration.
- May use Provocholine with or without meals.
- The methacholine challenge test is considered positive if there is a reduction in FEV1 of 20% or more from post-diluent FEV1. The test should be stopped at this point. The reduction value must be calculated and recorded before starting the test with Provocholine (see Calculation and Interpretation of Methacholine Challenge Test Results (2.6)).
- An inhaled β agonist must be administered after a methacholine challenge test with Provocholine to expedite the return of the FEV1 to baseline and to relieve any discomfort of the subject. Most patients revert to normal pulmonary function within 10 to 20 minutes following administration of a β agonist.
2.2 Recommended Dosage
The recommended dosage of Provocholine powder for inhalation solution (require reconstitution and dilution) or inhalation solution (in a ready-to-use kit) used in the methacholine challenge test administered via nebulization in adult and pediatric patients (5 years or older) is increasing concentrations of methacholine chloride solutions using either doubling or quadrupling dosing concentrations. Please refer to Table 3 for the doubling or quadrupling dosing concentrations.
2.3 Reconstitution and Dilution Prior to Administration
1. Provocholine Powder for Inhalation Solution requires reconstitution before use (see Tables 1 and 2):
Add 6.25 mL of 0.9% Sodium Chloride Injection (0.9% saline) or 0.9% Sodium Chloride Injection with 0.4% phenol (0.9% saline with 0.4% phenol) to the supplied vials containing 100 mg of Provocholine powder. Shake the vial to obtain a clear solution.
2. Dilute the reconstituted Provocholine solution:
Using sterile, empty USP Type 1 borosilicate glass vials, dilute the reconstituted Provocholine solution with 0.9% saline or 0.9% saline with 0.4% phenol either by doubling the concentration (see Table 1) or quadrupling the concentration (see Table 2). After adding the diluent, shake each vial to obtain a clear solution. Use the same diluent to prepare all concentrations
3. Use a sterile bacterial-retentive filter (porosity 0.22 µm) when transferring the reconstituted or diluted solution from each vial (at least 2 mL) to a nebulizer.
4. Refrigerate the reconstituted and diluted solutions at 36oF to 46oF (2oC to 8oC) for up to 2 weeks. Since the temperature of the solution affects nebulizer output, solutions should be taken out of the refrigerator and allowed to equilibrate to room temperature (approximately 30 minutes) before use.
Table 1: Reconstitution and Dilution of Supplied Provocholine powder for inhalation solution: Doubling Concentrations
|
TAKE |
ADD 0.9% Saline or 0.9% Saline with 0.4% Phenol |
Concentration (Total Volume) after reconstitution or dilution |
|
100 mg of Provocholine Powder in one supplied vial |
6.25 mL |
16 mg/mL (6.25 mL) (Solution A) |
|
3 mL of Solution A |
3 mL |
8 mg/mL (6 mL) (Solution B) |
|
3 mL of Solution B |
3 mL |
4 mg/mL (6 mL) (Solution C) |
|
3 mL of Solution C |
3 mL |
2 mg/mL (6 mL) (Solution D) |
|
3 mL of Solution D |
3 mL |
1 mg/mL (6 mL) (Solution E) |
|
3 mL of Solution E |
3 mL |
0.5 mg/mL (6 mL) (Solution F) |
|
3 mL of Solution F |
3 mL |
0.25 mg/mL (6 mL) (Solution G) |
|
3 mL of Solution G |
3 mL |
0.125 mg/mL (6 mL) (Solution H) |
|
3 mL of Solution H |
3 mL |
0.0625 mg/mL (6 mL) (Solution I) |
Table 2: Reconstitution and Dilution of Supplied Provocholine powder for inhalation solution: Quadrupling Concentrations
|
TAKE |
ADD 0.9% Saline or 0.9% Saline with 0.4% Phenol |
Concentration (Total Volume) after reconstitution or dilution |
|
100 mg of Provocholine Powder in one supplied vial |
6.25 mL |
16 mg/mL (6.25 mL) (Solution 1) |
|
3 mL of Solution 1 |
9 mL |
4 mg/mL (12 mL) (Solution 2) |
|
3 mL of Solution 2 |
9 mL |
1 mg/mL (12 mL) (Solution 3) |
|
3 mL of Solution 3 |
9 mL |
0.25 mg/mL (12 mL) (Solution 4) |
|
3 mL of Solution 4 |
9 mL |
0.0625 mg/mL (12 mL) (Solution 5) |
2.4 Administration with the Five (5)-Breath Dosimeter Dosing Method in Patients 5 Years of Age and Older
Prior to administering the Provocholine dose(s), determine the post-diluent FEV1 value required for the methacholine challenge test.
Administration of the Diluent or Base to Obtain Post-Diluent FEV1 Value
1. For the Provocholine powder for inhalation solution:
Using a 3 mL syringe and needle, draw up 2 to 3 mL of the same diluent used to reconstitute the Provocholine Powder (0.9% saline or 0.9% saline with 0.4% phenol) and dispense into the nebulizer using a sterile bacterial-retentive filter (porosity 0.22 µm).
For the Provocholine inhalation solution:
Dispense the contents of a vial containing the base solution (contains no methacholine chloride) into the nebulizer.
2. Instruct the patient to hold the nebulizer upright with the mouthpiece in his/her mouth. The patient should wear a nose clip while inhaling from the nebulizer.
3. At the end of exhalation during tidal breathing (functional residual capacity), instruct the patient to inhale slowly and deeply through the mouthpiece. Trigger the dosimeter soon after oral inhalation begins. Encourage the patient to continue inhaling slowly (about 5 seconds to complete the inhalation) and to hold the breath at total lung capacity (TLC) for another 5 seconds.
4. Repeat Step 3 for a total of five inspiratory capacity inhalations. Take no more than 2 minutes to perform these 5 inhalations.
5. Perform spirometry and measure the FEV1 30 and 90 seconds after the fifth inhalation from the nebulizer to obtain the post-diluent FEV1 value. These values may be left at ambient (spirometer) temperature pressure saturated (ATPS). If the FEV1 value is not of acceptable quality, repeat the procedure. If the post-diluent FEV1 falls by ≥ 20% from baseline FEV1, do not give further inhalations and proceed to Step 8. If the post-diluent FEV1 falls by < 20% from baseline FEV1, continue to Step 6.
Administration of Provocholine in a Methacholine Challenge Test
6. For the Provocholine powder for inhalation:
Using a 3 mL syringe and needle, draw up the recommended Provocholine concentration (see Table 3) that was prepared using either the doubling or quadrupling dose method and dispense into the nebulizer using a sterile bacterial-retentive filter (porosity 0.22 µm). See Tables 1 and 2 for preparation of the Provocholine powder for inhalation solution.
Table 3: Recommended Provocholine Dose(s) By Nebulization [Doubling Dose(s) or Quadrupling Dose(s)]
| Doubling Dose Increments | |
| Provocholine Concentration | Provocholine Dose* |
| 0.0625 mg/mL (Solution I) | 1.484 mcg |
| 0.125 mg/mL (Solution H) | 2.969 mcg |
| 0.25 mg/mL (Solution G) | 5.938 mcg |
| 0.5 mg/mL (Solution F) | 11.875 mcg |
| 1 mg/mL (Solution E) | 23.75 mcg |
| 2 mg/mL (Solution D) | 47.5 mcg |
| 4 mg/mL (Solution C) | 95 mcg |
| 8 mg/mL (Solution B) | 190 mcg |
| 16 mg/mL (Solution A) | 380 mcg |
| Quadrupling Dose Increments | |
| Provocholine Concentration | Provocholine Dose* |
| 0.0625 mg/mL (Solution 5) | 1.484 mcg |
| 0.25 mg/mL (Solution 4) | 5.938 mcg |
| 1 mg/mL (Solution 3) | 23.75 mcg |
| 4 mg/mL (Solution 2) | 95 mcg |
| 16 mg/mL (Solution 1) | 380 mcg |
* Dose delivered based on the drug output of the English Wright Nebulizer and the duration of inhalation (2 minutes).
For the Provocholine inhalation solution:
Dispense the contents of a vial of the appropriate Provocholine concentration, starting with the lowest dose, into the nebulizer. The Provocholine solution concentrations, 0.0625 mg/mL, 0.25 mg/mL, 1 mg/mL, 4 mg/mL, and 16 mg/mL, provided in the kit are ready-to-use. No further dilution is required.
7. Repeat steps 2 through 5 for each Provocholine concentration, emptying the nebulizer between each concentration. To keep the cumulative effect of Provocholine relatively constant, the time interval between the commencement of two subsequent concentrations should be kept to 5 minutes.
8. Stop dosing if the FEV1 has fallen by ≥ 20% from the post-diluent FEV1, or the highest Provocholine concentration (16 mg/mL) has been administered (whichever comes first). Do not administer additional Provocholine concentrations if severe bronchoconstriction occurs [see Warnings and Precautions (5.1)].
9. After the test is completed, administer an inhaled β-agonist to the patient to expedite the return of the FEV1 to within 90% of baseline and to relieve any discomfort (the majority of patients revert to normal pulmonary function within 5 minutes after β-agonist administration; in contrast the majority of patients revert to normal pulmonary function within 30-45 minutes without β-agonist administration). Wait 10 minutes and measure the FEV1 and Vital Capacity. Patients should not be allowed to leave the laboratory until their FEV1 has returned to within 90% of baseline.
10. After the test, wash and clean reusable nebulizers thoroughly according to manufacturer’s recommendations.
2.5 Administration with the Two (2)-Minute Tidal Breathing Dosing Method in Patients 5 Years of Age and Older
Administer the diluent and the Provocholine dose(s) using the English Wright nebulizer or other suitable nebulizer as long as the device output and particle size are characterized.
Prior to administering the Provocholine dose(s), determine the post-diluent FEV1 required for the methacholine challenge test.
Administration of the Diluent or Base Solution to Obtain Post-Diluent FEV1 Value
1. For the Provocholine powder for inhalation solution:
Using a 3 mL syringe and needle, draw up 2 to 3 mL of the same diluent used to reconstitute the Provocholine powder (0.9% saline or 0.9% saline with 0.4% phenol) and dispense into the nebulizer using a sterile bacterial-retentive filter (porosity 0.22 µm).
For the Provocholine inhalation solution:
Dispense the contents of a vial containing the base solution (contains no methacholine chloride) into the nebulizer.
2. Instruct the patient to relax and breathe the aerosol quietly (tidal breathing) for 2 minutes of inhalation time.
3. Place the face mask loosely over the nose and mouth or the mouthpiece in the mouth (with a nose clip) of the patient. The patient should hold the nebulizer to avoid warming the solution. Nebulizer should be kept upright and vertical.
4. Start the nebulizer by adjusting the flow meter so that the nebulizer is operating at the calibrated output (0.13 mL/minute for the English Wright nebulizer). Start the stopwatch immediately.
5. After exactly 2 minutes, turn off the flow meter, remove the face mask (or the mouthpiece from the mouth), and discard any remaining solution.
6. Perform spirometry and measure the FEV1 30 and 90 seconds after the end of the inhalation to obtain the post-diluent FEV1. These values may be left at ambient (spirometer) temperature pressure saturated (ATPS). If the FEV1 value is not of acceptable quality, repeat the procedure. If the post-diluent FEV1 falls by ≥ 20% from baseline FEV1, do not give further inhalations and proceed to Step 9. If the post-diluent FEV1 falls by < 20% from baseline FEV1, continue to Step 7.
Administration of Provocholine in a Methacholine Challenge Test
7. For the Provocholine powder for inhalation solution:
Using a 3 mL syringe and needle, draw up the recommended Provocholine dose (see Table 3) using either the doubling or quadrupling dose method and dispense into the nebulizer using a sterile bacterial-retentive filter (porosity 0.22 µm). See Tables 1 and 2 for preparation of the Provocholine powder for inhalation.
For the Provocholine inhalation solution:
Dispense the contents of a vial of the appropriate Provocholine concentration, starting with the lowest dose, into the nebulizer. The Provocholine solution concentrations, 0.0625 mg/mL, 0.25 mg/mL, 1 mg/mL, 4 mg/mL, and 16 mg /mL, provided in the kit are ready-to-use. No further dilution is required.
8. Repeat steps 2 through 6 for each Provocholine dose, emptying the nebulizer between each dose. However, stop dosing if the FEV1 has fallen by ≥ 20% from the post-diluent FEV1 or the highest Provocholine concentration (16 mg/mL) has been administered (whichever comes first). Do not administer additional Provocholine doses if severe bronchoconstriction occurs [see Warnings and Precautions (5.1)].
9. After the test is completed, administer an inhaled β-agonist to the patient to expedite the return of the FEV1 to within 90% of baseline and to relieve any discomfort (the majority of patients revert to normal pulmonary function within 5 minutes after β-agonist administration; in contrast the majority of patients revert to normal pulmonary function within 30-45 minutes without β-agonist administration). Wait 10 minutes and measure the FEV1 and Vital Capacity. Patients should not be allowed to leave the laboratory until their FEV1 has returned to within 90% of baseline.
10. After the test, wash and clean reusable nebulizers thoroughly according to manufacturer’s recommendations and discard disposable nebulizers appropriately.
2.6 Calculation and Interpretation of Methacholine Challenge Test Results
A positive methacholine challenge test is a ≥ 20% reduction in the FEV1 (after Provocholine oral inhalation) compared with the mean post-diluent FEV1. Calculate and record post-diluent FEV1 value before the methacholine challenge test is started. Express airway hyperreactivity as the provocative Provocholine concentration (mg/mL) providing a fall in FEV1 of ≥ 20% (PC20) when the methacholine challenge test is dosed using either the 5-breath dosimeter method or the 2-minute tidal breathing method, or as the provocative Provocholine dose (mcg) providing a fall in FEV1 of ≥ 20% (PD20) when using the 2-minute tidal breathing method.
Calculation of PC20
Calculate PC20 using one of the following methods. Determine the percent decrease in FEV1 using the mean post-diluent FEV1 and the lowest FEV1 post-dose, as shown below:
% fall in FEV1 = mean post-diluent FEV1 - lowest FEV1 post-Provocholine x 100
mean post-diluent FEV1
Method #1
Plot the percent decrease in FEV1 against the increasing methacholine concentration using a log scale and obtain the PC20 by linear interpolation between the last two points, as shown in Figure 1.
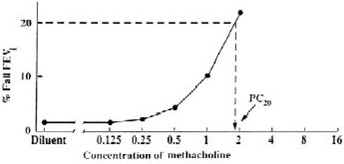
Method #2
Alternatively, calculate the PC20 as follows:
PC20 = antilog [log C1+ (log C2 - log C1)(20 - R1)]
(R2- R1)
Where:
• C1 = second last methacholine concentration (< 20% FEV1 decrease)
• C2 = last methacholine concentration (≥ 20% FEV1 decrease)
• R1 = % fall FEV1 after C1
• R2 = % fall FEV1 after C2
Calculation of PD20 (2-minute tidal breathing method only)
Calculate the PD20 as follows:
PD20 = antilog [log D1+ (log D2 - log D1)(20 - R1)]
(R2- R1)
Where:
• D1 = second last Provocholine dose (< 20% FEV1 decrease)
• D2 = last Provocholine dose (≥ 20% FEV1 decrease)
• R1 = % FEV1 decrease after D1
• R2 = % FEV1 decrease after D2
When using the English Wright nebulizer, refer to Table 2 for D1 and D2.
Interpretation of Results
A negative (normal) methacholine challenge result is defined as FEV1 reduction of < 20% after all the doses (doubling or quadrupling dose increments) in Table 1 (for 5-breath dosimeter method) or Table 2 (for the 2-minute tidal breathing method) have been administered.
If asthma drugs are discontinued prior to the methacholine challenge test, consider the possibility of rebound airway hyperreactivity in the interpretation of the test results. The methacholine challenge test may occasionally be falsely positive after an influenza infection or upper respiratory infection, immunizations, in very young or very old patients, in patients with chronic lung disease (e.g., cystic fibrosis, sarcoidosis, tuberculosis, chronic obstructive pulmonary disease), in patients with allergic rhinitis without asthma symptoms, in smokers, or in patients after exposure to air pollutants.
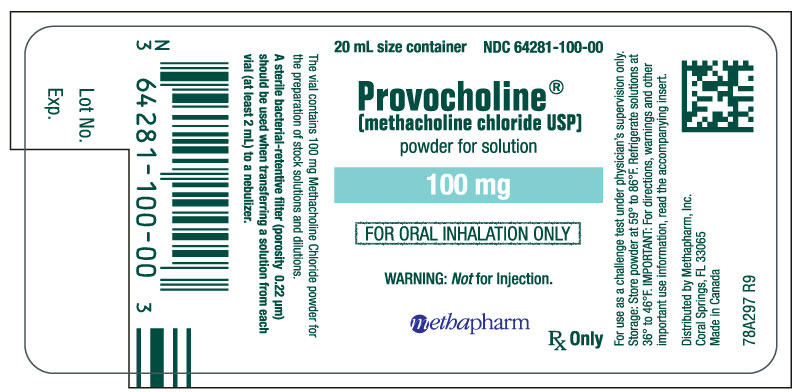
3 DOSAGE FORMS AND STRENGTHS
• For inhalation solution: 100 mg of white to off-white crystalline powder in amber glass vials (powder is reconstituted and then diluted prior to administration)
• Inhalation solution:
o base solution (contains no methacholine chloride/3 mL)
o 0.0625 mg/mL (0.1875 mg/3 mL)
o 0.25 mg/mL (0.75 mg/3 mL)
o 1 mg/mL (3 mg/3 mL)
o 4 mg/mL (12 mg/3 mL)
o 16 mg/mL (48 mg/3 mL)
4 CONTRAINDICATIONS
Provocholine is contraindicated in the following:
- Hypersensitivity to methacholine or other parasympathomimetic agents. Reactions have included rash, itching/swelling (especially of the face/tongue/throat), severe dizziness, trouble breathing.
- Baseline FEV1 < 60% predicted (adults or pediatric patients) or <1.5 L (adults)
5 WARNINGS AND PRECAUTIONS
5.1 Risk of Severe Bronchoconstriction
Severe bronchoconstriction can result from Provocholine administration (including the lowest dose). The use of Provocholine is contraindicated in pediatric and adult patients with baseline FEV1 < 60% predicted or adults with FEV1 < 1.5 L. Emergency equipment and medication should be immediately available to treat acute respiratory distress. Because of the potential for severe bronchoconstriction, the use of Provocholine in patients with clinically apparent asthma or wheezing is not recommended. If severe bronchoconstriction occurs, reverse immediately by the administration of a rapid-acting inhaled β-agonist.
If baseline spirometry is not performed or is measured inaccurately, the initial FEV1 may be underestimated. In this situation, decreases in FEV1 may not be detected after escalating Provocholine doses, which may result in administration of unnecessary higher doses and an increase in the risk for excessive bronchoconstriction.
5.2 Risks to Healthcare Providers Administering Provocholine
The supplied Provocholine powder or the Provocholine nebulized aerosol may cause bronchoconstriction in healthcare providers administering Provocholine in a methacholine challenge test. Healthcare providers and any other personnel involved in the administration of Provocholine should take the following precautionary steps:
- Do not inhale the supplied Provocholine powder
- Do not handle the Provocholine powder if you have asthma or hay fever.
- Apply a low resistance filter to expiratory ports of dosing apparatus, as necessary, to prevent Provocholine release in the room air
5.3 Coexisting Diseases and Conditions
Provocholine is not recommended for patients with uncontrolled hypertension, aortic aneurysm, or history of myocardial infarction or stroke diseases. Patients with epilepsy, vagotonia, peptic ulcer disease, thyroid disease, urinary tract obstruction or other condition that could be adversely affected by a cholinergic agent should undergo methacholine challenge only if the healthcare practitioner considers the benefit to the individual outweighs the potential risks.
6 ADVERSE REACTIONS
The following adverse reactions associated with the use of Provocholine were identified in clinical studies or post marketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Bronchospasm (includes symptoms such as chest tightness, cough or wheezing).
Adverse reactions less commonly associated with Provocholine include headache, throat irritation, light-headedness and itching.
7 DRUG INTERACTIONS
Beta-Adrenergic Blockers
The use of beta-adrenergic blockers may impair reversal of Provocholine-caused bronchoconstriction.
Beta-Agonists, Anticholinergics, and Theophylline
Beta-agonists, anticholinergics, and theophylline inhibit the response of airways to Provocholine; therefore, hold these drugs before Provocholine use for the following duration:
• Short-acting β-agonists (e.g., albuterol): 6 hours
• Long-acting β-agonists (e.g., salmeterol): 36 hours
• Short-acting anti-cholinergics (e.g., ipratropium): 12 hours
• Long-acting anti-cholinergics (e.g., tiotropium): ≥168 hours
• Oral theophylline: 12-48 hours
Oral or Inhaled Corticosteroids, and Inhaled Cromoglycate
Regular use of oral or inhaled corticosteroids and inhaled cromoglycate may acutely decrease bronchial responsiveness to Provocholine. However, these drugs may be continued with Provocholine use.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
The available data from published literature on Provocholine use in pregnant women are insufficient to evaluate for a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. Animal reproduction studies evaluating effects of methacholine chloride on embryofetal development have not been conducted. Diagnosis of bronchial airway hyperreactivity with bronchoprovocation challenge is not recommended for pregnant women because of the potential for hypoxia in the fetus. If bronchial airway hyperreactivity is suspected, consider trial of empiric treatment.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the United States general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
8.2 Lactation
Risk Summary
There are no available data on the presence of methacholine chloride in human milk, the effect on the breastfed infant, or the effect on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Provocholine and any potential adverse effects on the breastfed infant from Provocholine or from the underlying maternal condition.
8.4 Pediatric Use
Provocholine is used in a methacholine challenge test for the diagnosis of bronchial airway hyperreactivity in pediatric patients 5 years of age and older who do not have clinically apparent asthma. The safety and effectiveness of Provocholine have not been established in pediatric patients below the age of 5 years.
11 DESCRIPTION
Methacholine chloride, the active ingredient of Provocholine, is a parasympathomimetic (cholinergic) bronchoconstrictor agent. Provocholine (methacholine chloride) powder for solution is administered by oral inhalation.
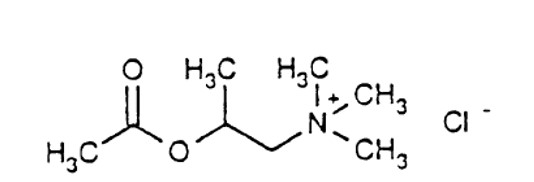
Chemically, methacholine chloride (the active ingredient) is 1-propanaminium, 2-(acetyloxy)-N,N,N,-trimethyl-, chloride. It is a white to practically white deliquescent compound, soluble in water, alcohol and chloroform and insoluble in ether. Aqueous solutions are neutral to litmus.
Methacholine chloride has an empirical formula of C8H18ClNO2, a molecular weight of 195.69, and the following structural formula:
Provocholine Powder for Solution: Each vial of Provocholine contains 100 mg of methacholine chloride powder.
Provocholine Solution for Inhalation Kit (Sterile):
Each kit is packaged in a carton with six plastic vials with twist-off cap each containing 3 mL of the following concentrations of methacholine chloride solution. Each solution also contains: Sodium Acetate Trihydrate, Sodium Chloride, glacial acetic acid as pH adjuster.
a) base solution (contains no methacholine chloride)
b) 0.0625 mg/mL (0.1875 mg/3 mL)
c) 0.25 mg/mL (0.75 mg/3 mL)
d) 1 mg/mL (3 mg/3 mL)
e) 4 mg/mL (12 mg/3 mL)
f) 16 mg/mL (48 mg/3 mL)
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Methacholine chloride is a cholinergic agonist. Bronchial smooth muscle contains significant parasympathetic (cholinergic) innervation. Methacholine chloride agonizes the muscarinic receptors which eventually induce bronchoconstriction.
16 HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
- Provocholine (methacholine chloride) Powder for Inhalation Solution: in amber glass vials that contain 100 mg of methacholine chloride powder, white to off-white in color. Cartons have 6 vials (NDC 64281-100-06).
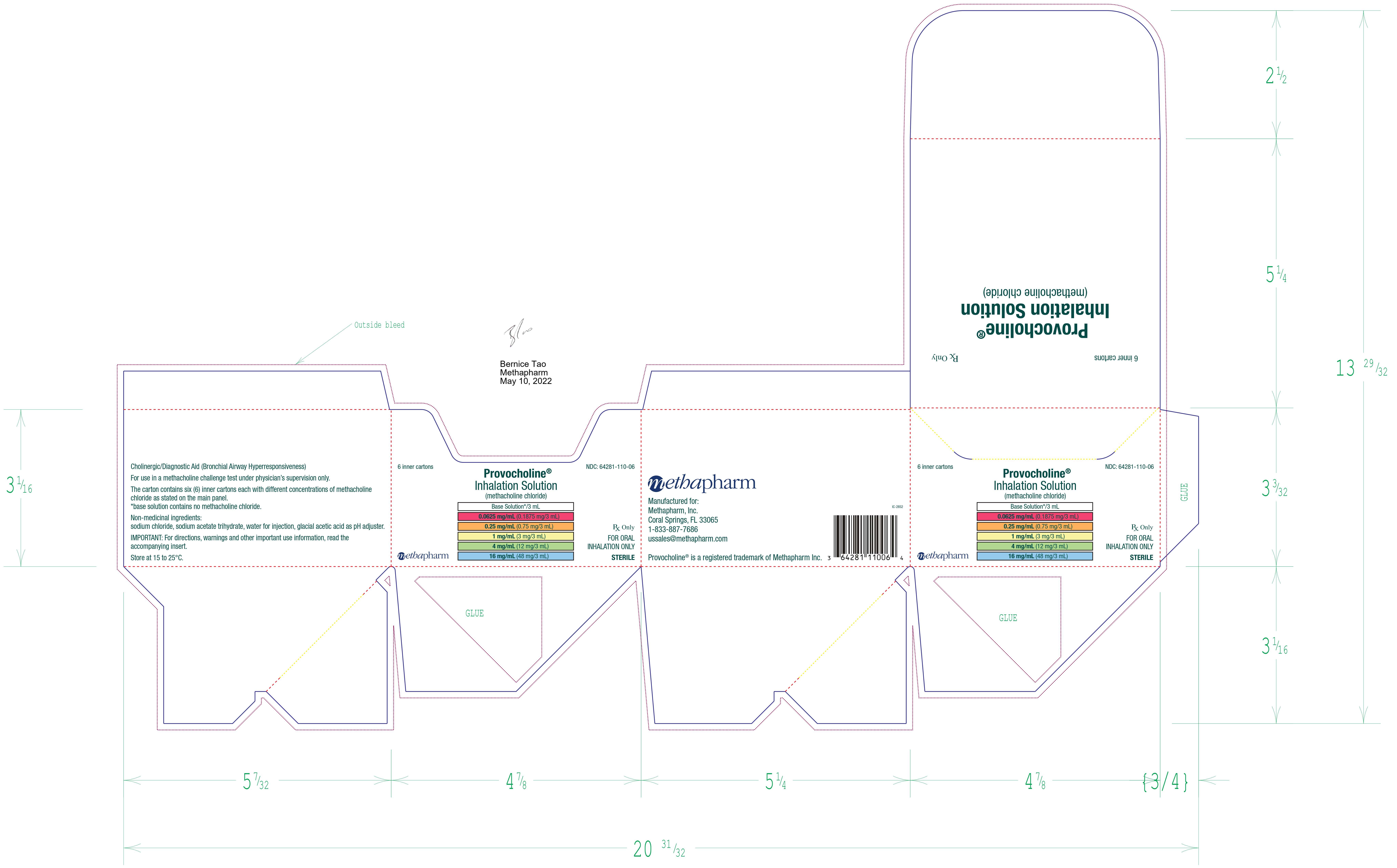
- Provocholine (methacholine chloride) Inhalation Solution Kit (Sterile):
Each kit (NDC: 64281-110-05) contains six (6) plastic vials with twist-off cap each containing 3 mL of different concentrations of methacholine chloride as shown below. Cartons have six (6) kits (NDC 64281-110-06).
a) base solution (contains no methacholine chloride) (NDC: 64281-111-00)
b) 0.0625 mg/mL (0.1875 mg/3 mL) (NDC:64281-112-00)
c) 0.25 mg/mL (0.75 mg/3 mL) (NDC:64281-113-00)
d) 1 mg/mL (3 mg/3 mL) (NDC:64281-114-00)
e) 4 mg/mL (12 mg/3 mL) (NDC:64281-115-00)
f) 16 mg/mL (48 mg/3 mL) (NDC:64281-116-00)
Storage and Handling
- Powder for inhalation solution: Store the supplied powder at 59oF to 86oF (15oC to 30oC). Refrigerate the reconstituted and diluted solutions at 36oF to 46oF (2oC to 8oC) for up to 2 weeks [see Dosage and Administration (2.3)].
- Provocholine inhalation solution kit: Store between 59oF to 86oF (15°C to 30°C). Use immediately upon opening the vial.
17 PATIENT COUNSELING INFORMATION
Risk of Severe Bronchoconstriction
Inform the patient or caregiver that severe bronchoconstriction can result from Provocholine administration [see Warnings and Precautions (5.1)].
Cough, Chest Tightness, or Shortness of Breath
Inform the patient or caregiver that Provocholine may produce mild cough, chest tightness or shortness of breath.

Manufactured for:
Methapharm, Inc.
11772 West Sample Road, Suite 101
Coral Springs, FL,
USA 33065
For more information visit www.methapharmrespiratory.com,
email ussales@methapharm.com
or call 1-833-887-7686.
®Provocholine is a registered trademark of Methapharm Inc.
Revision: December 2022