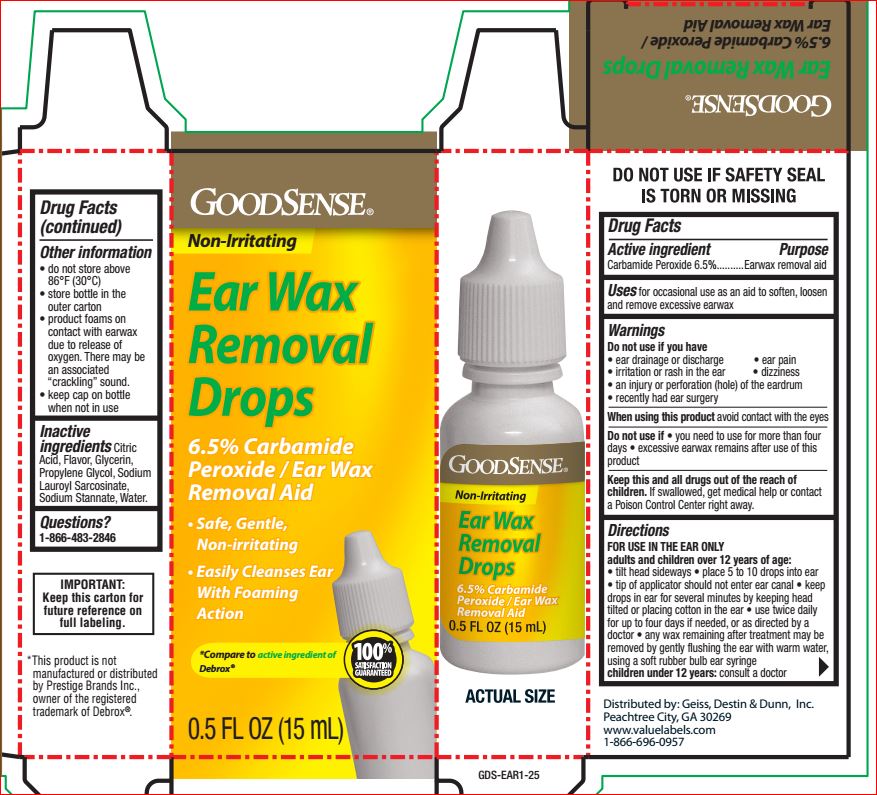
Warnings
Do not use if you have • ear drainage or discharge • ear pain • irritation or rash in ear • dizziness • an injury or perforation (hole) of the eardrum • recently had ear surgery
DirectionsFor use in the ear only.
Adults and children over 12 years of age:
• tilt head sideways and place 5 to 10 drops into ear
• tip of applicator should not enter ear canal
• keep drops in ear for several minutes by keeping head tilted or placing cotton in the ear
• use twice daily for up to 4 days if needed, or as directed by a doctor
• any wax remaining after treatment may be removed by gently flushing the ear with warm water, using a soft rubber bulb ear syringe
Children under 12 years of age: consult a doctor.