VISINE ORIGINAL REDNESS RELIEF- tetrahydrozoline hydrochloride solution/ drops
Johnson & Johnson Consumer Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
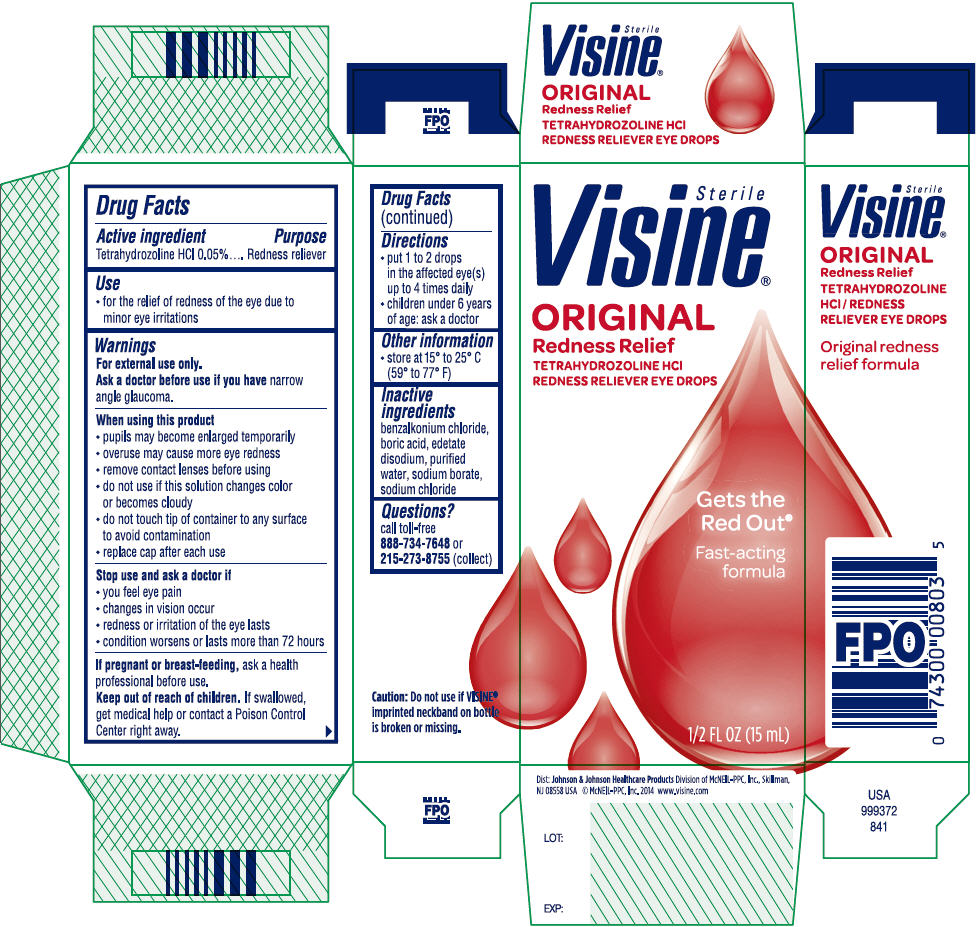
Visine® Original Redness Relief
Eye Drops
Warnings
For external use only.
When using this product
- pupils may become enlarged temporarily
- overuse may cause more eye redness
- remove contact lenses before using
- do not use if this solution changes color or becomes cloudy
- do not touch tip of container to any surface to avoid contamination
- replace cap after each use
Directions
- put 1 to 2 drops in the affected eye(s) up to 4 times daily
- children under 6 years of age: ask a doctor
| VISINE ORIGINAL
REDNESS RELIEF
tetrahydrozoline hydrochloride solution/ drops |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Johnson & Johnson Consumer Inc. (002347102) |
Revised: 9/2020
Document Id: 3a707756-04d2-46e0-a2f6-c1425fecb2aa
Set id: e3c41a58-fc93-4e6c-99be-0529edfb54a4
Version: 9
Effective Time: 20200922
Johnson & Johnson Consumer Inc.