LWP
381 Van Ness Ave. Suite 1507
Torrance CA, 90501
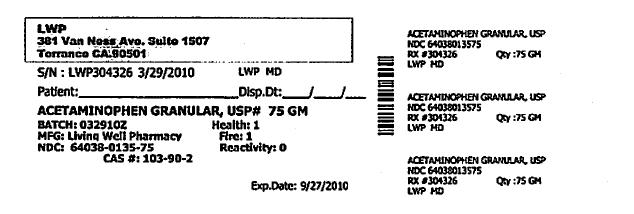
S/N: LWP304326 3/29/2010 LWP,MD
ACETAMINOPHEN GRANULAR, USP #75 GM
BATCH: 03292010Z HEALTH: 1
MFG: LIVING WELL PHARMACY FIRE: 1
NDC: 64038-0135-75 REACTIVITY: 0
CAS #: 103-90-2LWP
381 Van Ness Ave. Suite 1507
Torrance CA, 90501
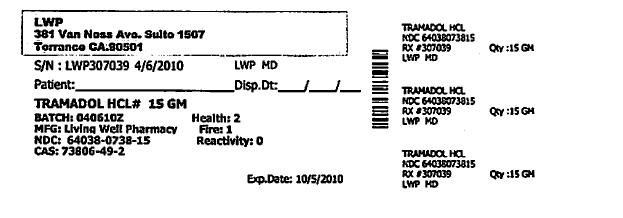
S/N: LWP307039 4/6/2010 LWP,MD
TRAMADOL HCL #15GM
BATCH: 04062010Z HEALTH: 2
MFG: LIVING WELL PHARMACY FIRE: 1
NDC: 64038-0738-15 REACTIVITY: 0
CAS #: 73806-49-2- RX Only
For Prescription Compounding Only
Tramapap Kit
Tramadol HCl and Acetaminophen Compound Kit
Description
Each Tramapap Kit is comprised of 15 grams of tramadol hydrochloride powder and 75 grams of acetaminophen powder.
Certificate of Analysis on File
Tramapap Kit also contains 23.25 grams of Lactose Monohydrate (Spray dried) powder and 1.5 grams of
riboflavin USP powder. When compounded, the final product provides a homogeneous product of 300
capsules each capsule containing 50mg of tramadol and 250mg of acetaminophen.
Equipment
Required supplies needed to compound this kit
| Item | Quantity |
|---|---|
| Tramadol HCl (Included) | 15 grams |
| Acetaminophen (Included) | 75 grams |
| Lactose Monohydrate Spray Dried Powder ( Included) | 23.25 grams |
| Riboflavin Powder (Included) | 1.5 grams |
| Red/Blue #1 Capsules ( Required Not Included) | 300 Capsules |
|
|
|
Equipment
Recommended supplies not included in this kit
| Item | Quantity |
|---|---|
| 16 oz Glass Mortar and Pestle | 1 each |
| 300 Capsule Machine Number 2 Notch | 1 |
| Scraper ( Recommended Not Included) | 1 |
| Tamper (Recommended Not Included) | 1 |
| Capsule Locker (Recommended Not Included) | 1 |
|
|
|
Directions
1. Using a 16 oz mortar and pestle triturate powders well to reduce particle size until uniform.
2. Using a 300 capsule machine encapsulate triturate powder mixture into number 1 red/blue capsules
Prior to compounding, store Tramapap Kit at room temperature. Store the final product at room temperature.