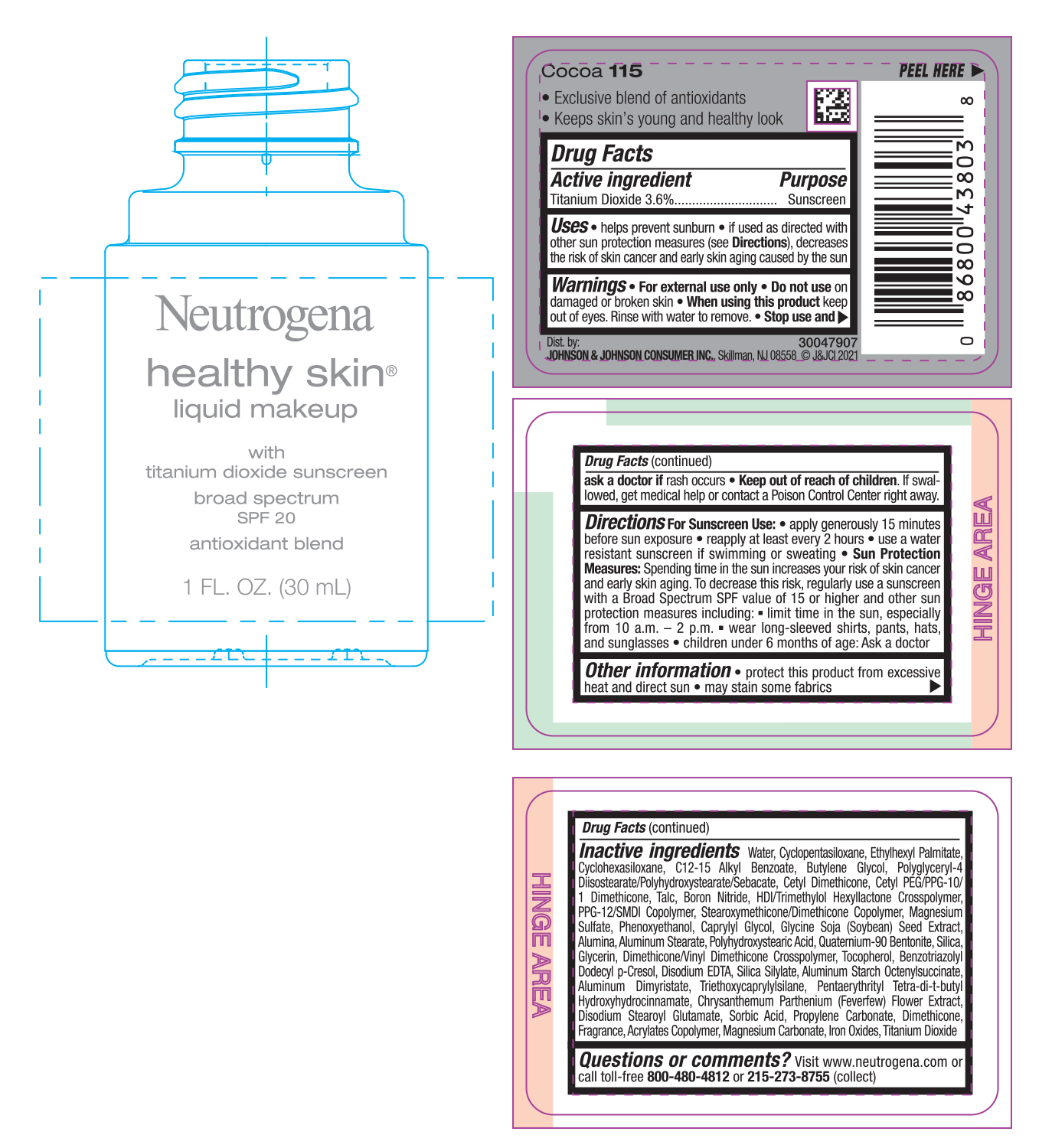
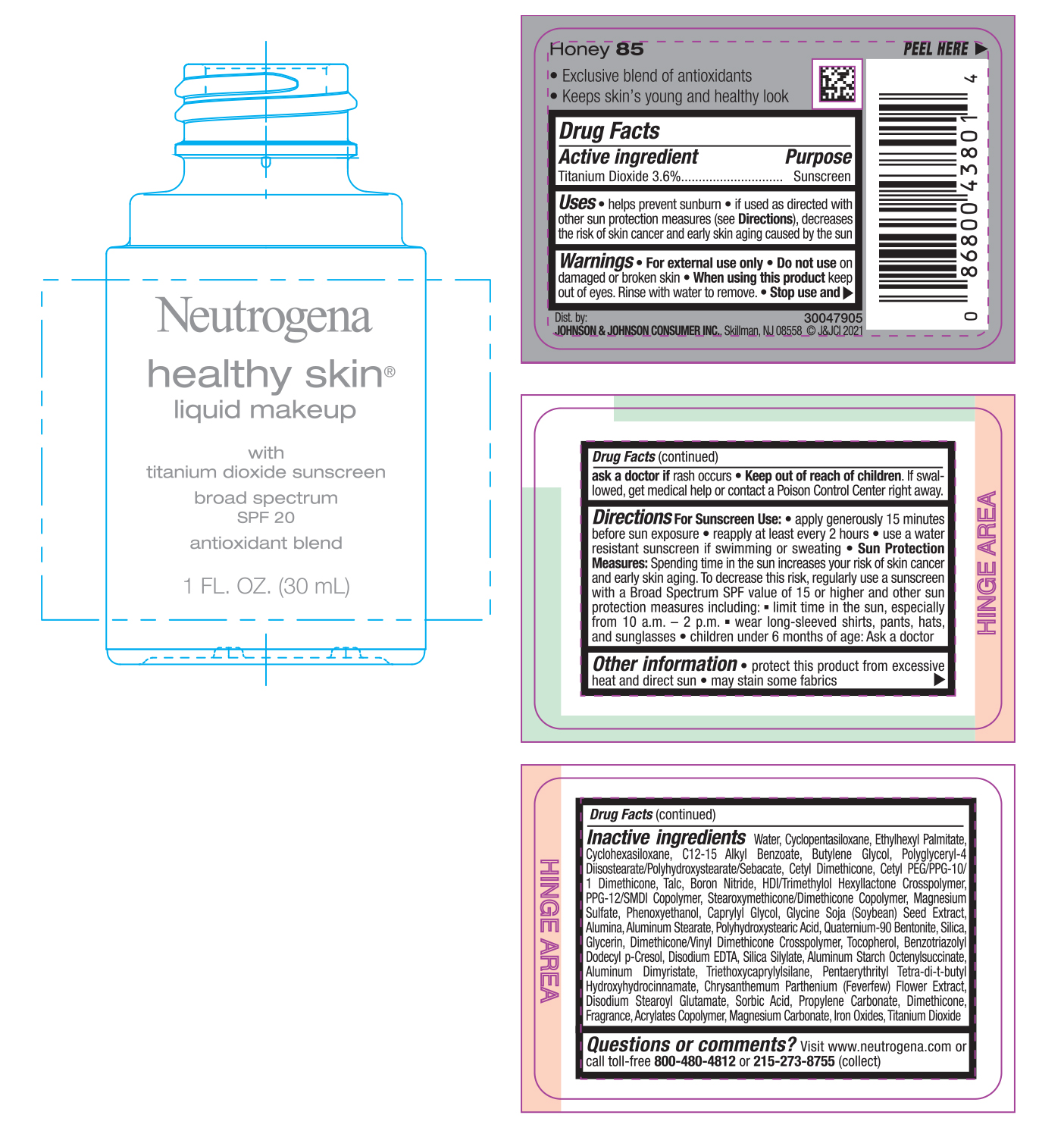
Use
- helps prevent sunburn
- if used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun
Directions
For sunscreen use:
- apply generously 15 minutes before sun exposure
- reapply at least every 2 hours
- use a water resistant sunscreen if swimming or sweating
Sun Protection Measures: Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. – 2 p.m.
- wear long-sleeved shirts, pants, hats, and sunglasses
- children under 6 months of age: Ask a doctor
Inactive ingredients
Water, Cyclopentasiloxane, Ethylhexyl Palmitate, Cyclohexasiloxane, C12-15 Alkyl Benzoate, Butylene Glycol, Polyglyceryl-4 Diisostearate/Polyhydroxystearate/Sebacate, Cetyl Dimethicone, Cetyl PEG/PPG-10/1 Dimethicone, Talc, Boron Nitride, HDI/Trimethylol Hexyllactone Crosspolymer, PPG-12/SMDI Copolymer, Stearoxymethicone/Dimethicone Copolymer, Magnesium Sulfate, Phenoxyethanol, Caprylyl Glycol, Glycine Soja (Soybean) Seed Extract, Alumina, Aluminum Stearate, Polyhydroxystearic Acid, Quaternium-90 Bentonite, Silica, Glycerin, Dimethicone/Vinyl Dimethicone Crosspolymer, Tocopherol, Benzotriazolyl Dodecyl p-Cresol, Disodium EDTA, Silica Silylate, Aluminum Starch Octenylsuccinate, Aluminum Dimyristate, Triethoxycaprylylsilane, Pentaerythrityl Tetra-di-t-butyl Hydroxyhydrocinnamate, Chrysanthemum Parthenium (Feverfew) Flower Extract, Disodium Stearoyl Glutamate, Sorbic Acid, Propylene Carbonate, Dimethicone, Fragrance, Acrylates Copolymer, Magnesium Carbonate, Iron Oxides, Titanium Dioxide
Questions or Comments?
Visit www.neutrogena.com or call toll-free 800-480-4812 or 215-273-8755 (collect)
PRINCIPAL DISPLAY PANEL - 30 mL Bottle Label - Classic Ivory 10
Neutrogena
healthy skin®
liquid makeup
with
titanium dioxide sunscreen
broad spectrum
SPF 20
antioxidant blend
1 FL. OZ. (30mL)

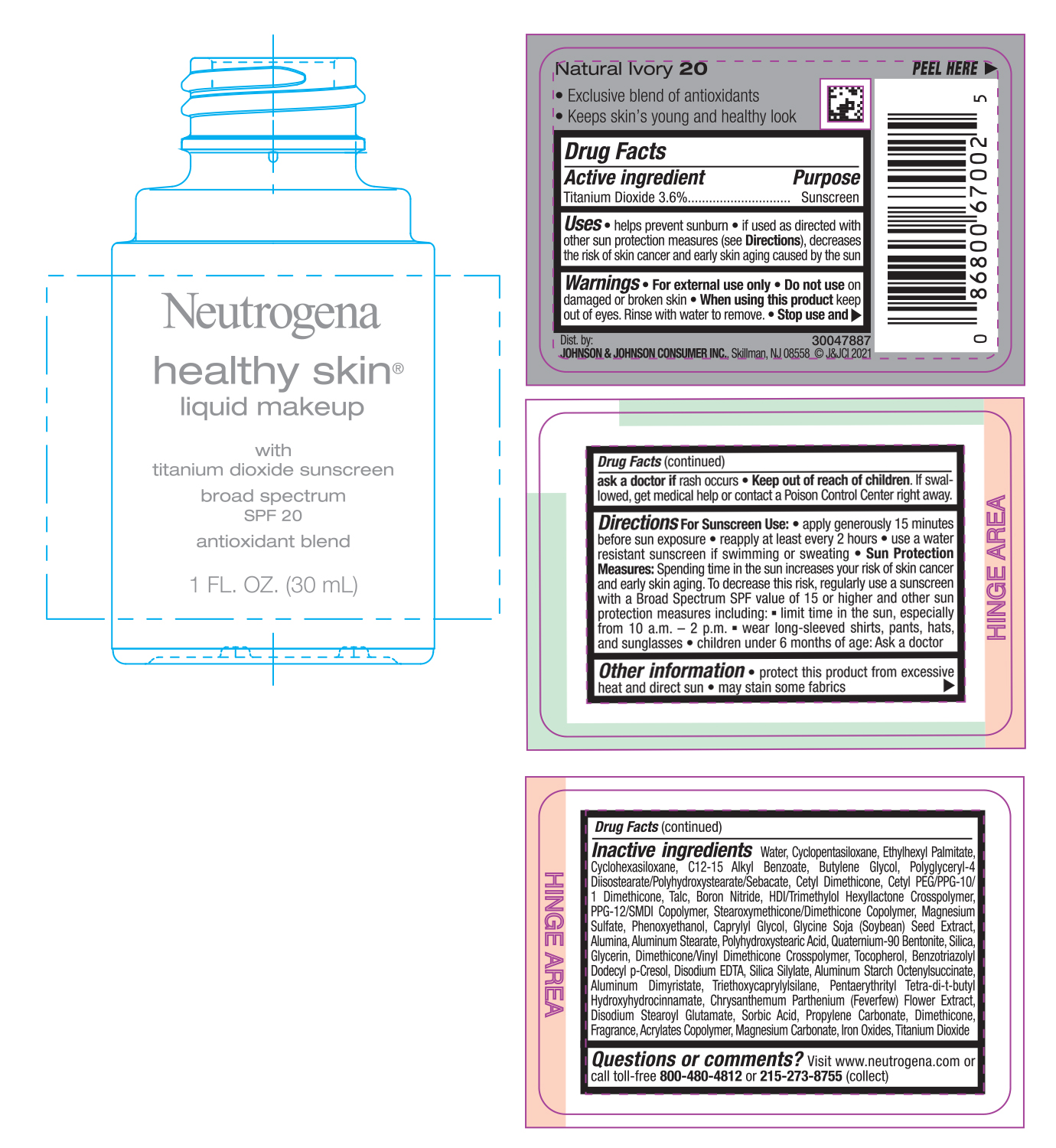
PRINCIPAL DISPLAY PANEL - 30 mL Bottle Label - Natural Ivory 20
Neutrogena
healthy skin®
liquid makeup
with
titanium dioxide sunscreen
broad spectrum
SPF 20
antioxidant blend
1 FL. OZ. (30mL)
PRINCIPAL DISPLAY PANEL - 30 mL Bottle Label - Buff 30
Neutrogena
healthy skin®
liquid makeup
with
titanium dioxide sunscreen
broad spectrum
SPF 20
antioxidant blend
1 FL. OZ. (30mL)

PRINCIPAL DISPLAY PANEL - 30 mL Bottle Label - Nude 40
Neutrogena
healthy skin®
liquid makeup
with
titanium dioxide sunscreen
broad spectrum
SPF 20
antioxidant blend
1 FL. OZ. (30mL)

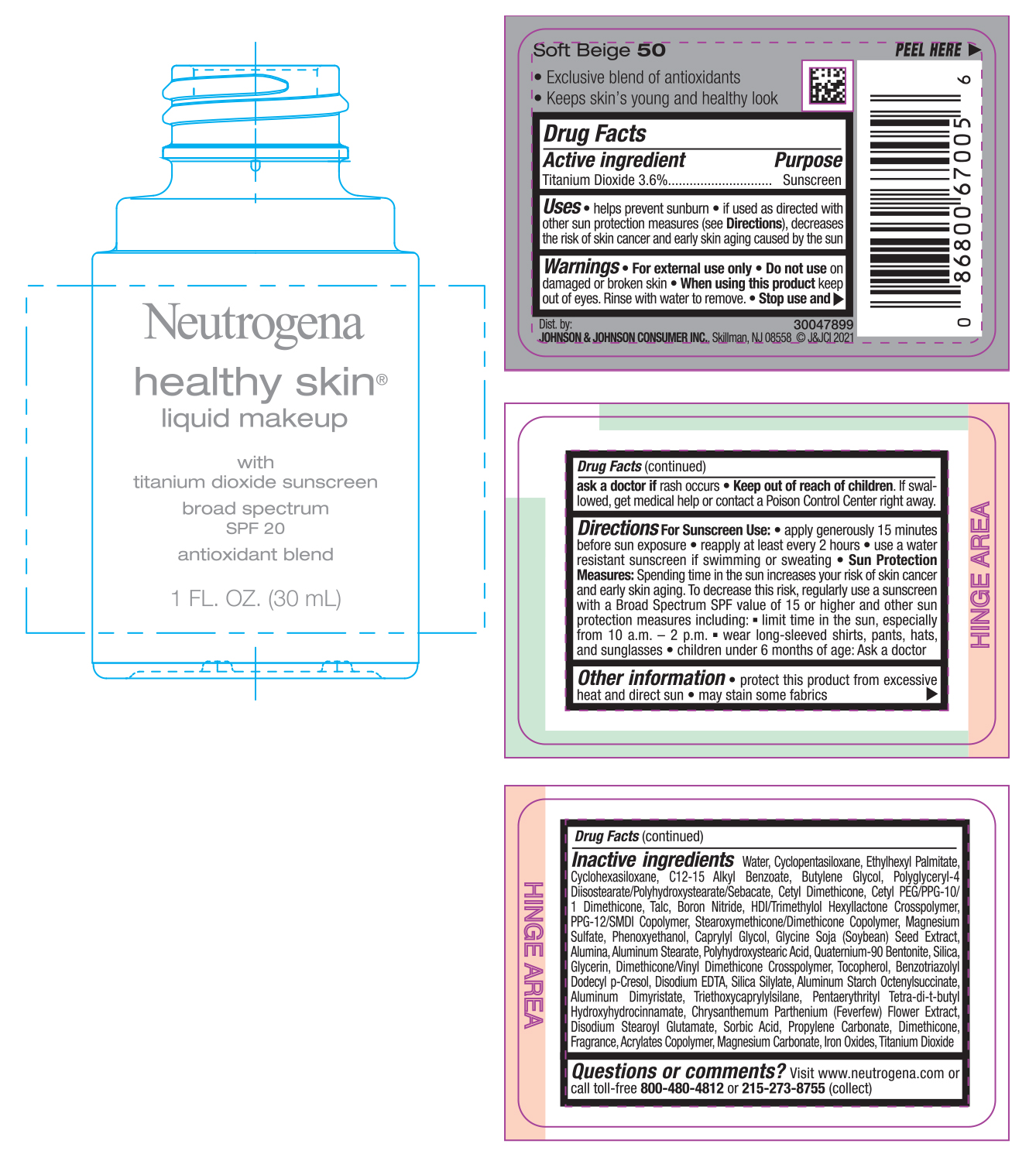
PRINCIPAL DISPLAY PANEL - 30 mL Bottle Label - Soft Beige 50
Neutrogena
healthy skin®
liquid makeup
with
titanium dioxide sunscreen
broad spectrum
SPF 20
antioxidant blend
1 FL. OZ. (30mL)
PRINCIPAL DISPLAY PANEL - 30 mL Bottle Label - Natural Beige 60
Neutrogena
healthy skin®
liquid makeup
with
titanium dioxide sunscreen
broad spectrum
SPF 20
antioxidant blend
1 FL. OZ. (30mL)

PRINCIPAL DISPLAY PANEL - 30 mL Bottle Label - Fresh Beige 70
Neutrogena
healthy skin®
liquid makeup
with
titanium dioxide sunscreen
broad spectrum
SPF 20
antioxidant blend
1 FL. OZ. (30mL)

PRINCIPAL DISPLAY PANEL - 30 mL Bottle Label - Medium Beige 80
Neutrogena
healthy skin®
liquid makeup
with
titanium dioxide sunscreen
broad spectrum
SPF 20
antioxidant blend
1 FL. OZ. (30mL)

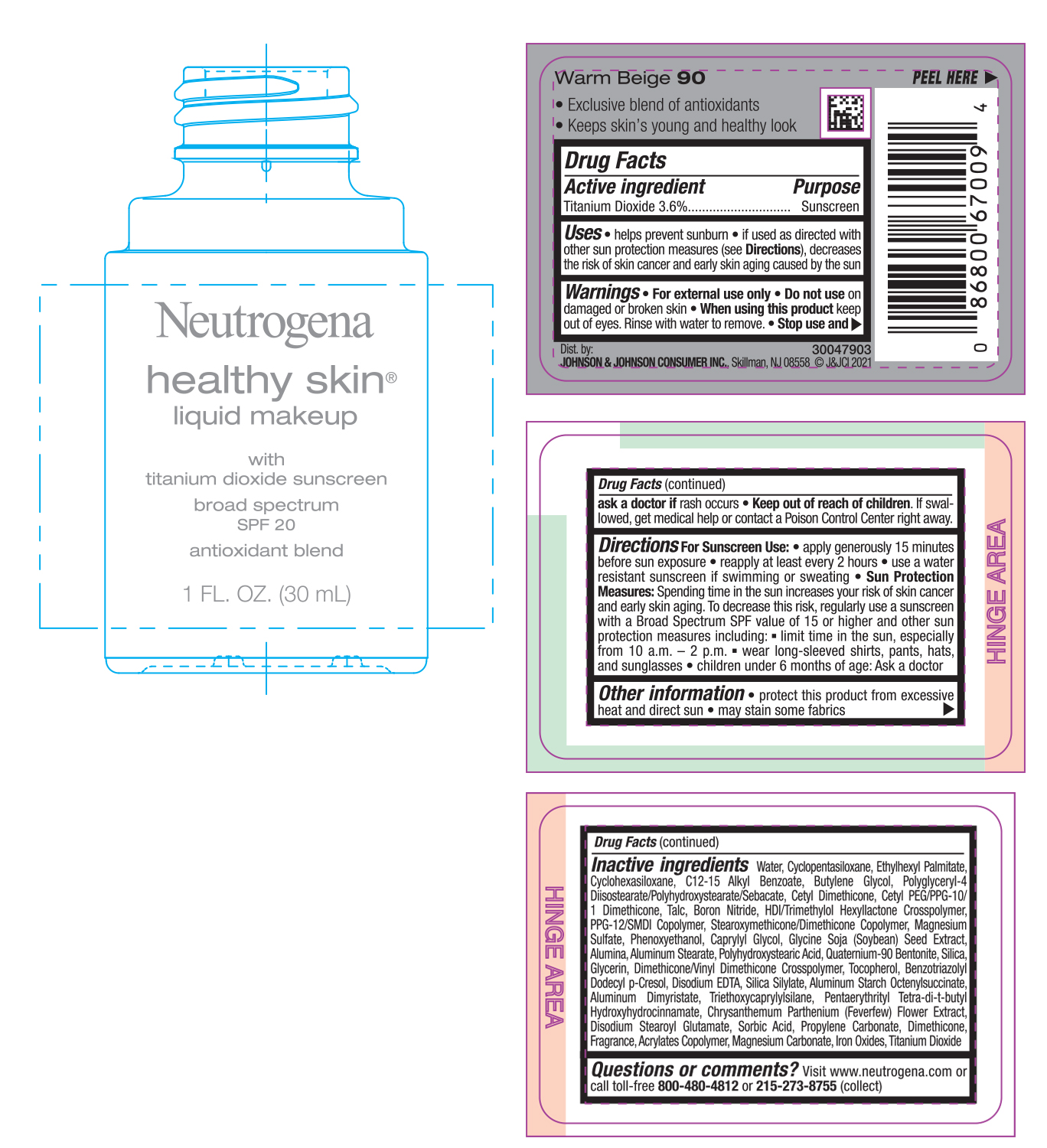
PRINCIPAL DISPLAY PANEL - 30 mL Bottle Label - Warm Beige 90
Neutrogena
healthy skin®
liquid makeup
with
titanium dioxide sunscreen
broad spectrum
SPF 20
antioxidant blend
1 FL. OZ. (30mL)
PRINCIPAL DISPLAY PANEL - 30 mL Bottle Label - Natural Tan 100
Neutrogena
healthy skin®
liquid makeup
with
titanium dioxide sunscreen
broad spectrum
SPF 20
antioxidant blend
1 FL. OZ. (30mL)

PRINCIPAL DISPLAY PANEL - 30 mL Bottle Label - Honey 85
Neutrogena
healthy skin®
liquid makeup
with
titanium dioxide sunscreen
broad spectrum
SPF 20
antioxidant blend
1 FL. OZ. (30mL)
PRINCIPAL DISPLAY PANEL - 30 mL Bottle Label - Caramel 105
Neutrogena
healthy skin®
liquid makeup
with
titanium dioxide sunscreen
broad spectrum
SPF 20
antioxidant blend
1 FL. OZ. (30mL)