FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
1.1 Clostridioides difficile-Associated Diarrhea
DIFICID® is indicated in adult and pediatric patients aged 6 months and older for the treatment of C. difficile-associated diarrhea (CDAD).
1.2 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of DIFICID and other antibacterial drugs, DIFICID should be used only to treat infections that are proven or strongly suspected to be caused by C. difficile. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Instructions
DIFICID is available for oral administration as 200 mg tablets and as granules for oral suspension (40 mg/mL (200 mg/5 mL) when reconstituted). DIFICID is administered orally with or without food.
2.2 Adult Patients
The recommended dosage for adults is one 200 mg DIFICID tablet orally twice daily for 10 days.
2.3 Pediatric Patients (6 Months to Less than 18 Years of Age)
Tablets
The recommended dosage for pediatric patients weighing at least 12.5 kg and able to swallow tablets is one 200 mg DIFICID tablet administered orally twice daily for 10 days. If unable to swallow tablets, pediatric patients may be dosed with DIFICID oral suspension as recommended in Table 1 below.
Oral Suspension
The recommended dosage for pediatric patients based on weight are shown in Table 1. Administer DIFICID oral suspension orally twice daily for 10 days using an oral dosing syringe [see Dosage and Administration (2.4)].
| Body Weight | Dose Administered Twice Daily | Volume of 40 mg/mL Suspension to be Administered Orally Twice Daily |
|---|---|---|
| 4 kg to less than 7 kg | 80 mg | 2 mL |
| 7 kg to less than 9 kg | 120 mg | 3 mL |
| 9 kg to less than 12.5 kg | 160 mg | 4 mL |
| 12.5 kg and above | 200 mg | 5 mL |
2.4 Preparation and Administration of DIFICID Oral Suspension
Preparation
- Shake the glass bottle to ensure the granules move around freely and no caking has occurred.
- Measure 130 mL of purified water, add to the glass bottle, and cap tightly.
- Hold bottle in a horizontal position and shake bottle vigorously in that position for at least 2 minutes.
- Verify that a homogeneous suspension is obtained. If not, repeat the shaking step.
- Once a homogeneous suspension is visually confirmed, shake an additional 30 seconds.
- Let bottle stand for 1 minute.
- Verify that the suspension is still homogeneous. If not, repeat steps 3 through 6.
- Once reconstituted, DIFICID oral suspension is white to yellowish white in color.
- Write discard date (current date plus 12 days) on the bottle [see How Supplied/Storage and Handling (16.1, 16.2)].
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
DIFICID is contraindicated in patients who have known hypersensitivity to fidaxomicin or any other ingredient in DIFICID [see Warnings and Precautions (5.1)].
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions
Acute hypersensitivity reactions, including dyspnea, rash, pruritus, and angioedema of the mouth, throat, and face have been reported with DIFICID. If a severe hypersensitivity reaction occurs, DIFICID should be discontinued and appropriate therapy should be instituted.
Some patients with hypersensitivity reactions to DIFICID also reported a history of allergy to other macrolides. Physicians prescribing DIFICID to patients with a known macrolide allergy should be aware of the possibility of hypersensitivity reactions.
5.2 Not for Use in Infections Other than C. difficile-Associated Diarrhea
DIFICID is not expected to be effective for the treatment of other types of infections due to minimal systemic absorption of fidaxomicin [see Clinical Pharmacology (12.3)]. DIFICID has not been studied for the treatment of infections other than CDAD. DIFICID should only be used for the treatment of CDAD.
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adults
The safety of DIFICID 200 mg tablets taken twice a day for 10 days was evaluated in 564 adult patients with CDAD in two active-controlled trials with 86.7% of patients receiving a full course of treatment.
Thirty-three adult patients receiving DIFICID (5.9%) withdrew from trials as a result of adverse reactions (AR). The types of AR resulting in withdrawal from the study varied considerably. Vomiting was the primary adverse reaction leading to discontinuation of dosing; this occurred at an incidence of 0.5% in both the DIFICID and vancomycin patients in Phase 3 trials. The most common selected adverse reactions occurring in ≥2% of adult patients treated with DIFICID are listed in Table 2.
| System Organ Class | DIFICID (N=564) | Vancomycin (N=583) |
|---|---|---|
| Adverse Reaction | n (%) | n (%) |
| Blood and Lymphatic System Disorders | ||
| Anemia | 14 (2%) | 12 (2%) |
| Neutropenia | 14 (2%) | 6 (1%) |
| Gastrointestinal Disorders | ||
| Nausea | 62 (11%) | 66 (11%) |
| Vomiting | 41 (7%) | 37 (6%) |
| Abdominal Pain | 33 (6%) | 23 (4%) |
| Gastrointestinal Hemorrhage | 20 (4%) | 12 (2%) |
The following adverse reactions were reported in <2% of adult patients taking DIFICID tablets in controlled trials:
Gastrointestinal Disorders: abdominal distension, abdominal tenderness, dyspepsia, dysphagia, flatulence, intestinal obstruction, megacolon
Investigations: increased blood alkaline phosphatase, decreased blood bicarbonate, increased hepatic enzymes, decreased platelet count
Metabolism and Nutrition Disorders: hyperglycemia, metabolic acidosis
Skin and Subcutaneous Tissue Disorders: drug eruption, pruritus, rash
Pediatrics
The safety of DIFICID in pediatric patients 6 months to less than 18 years of age was evaluated in a Phase 2 single-arm trial in 38 patients and a Phase 3 randomized, active-controlled trial in 98 patients treated with DIFICID and 44 patients treated with vancomycin [see Clinical Studies (14.2)]. In both studies, patients received DIFICID orally twice daily for 10 days. Patients <2 years of age, or weighing <12.5 kg, or unable to swallow tablets received weight-based doses of DIFICID oral suspension. Patients weighing at least 12.5 kg and able to swallow tablets received the 200 mg DIFICID tablet. The age range in the Phase 2 trial was 11 months to 17 years and in the Phase 3 trial was 1 month to 17 years (one patient was less than 6 months of age).
One death occurred in the Phase 2 single-arm trial. In the Phase 3 trial, there were 3 deaths in DIFICID-treated patients and no deaths in vancomycin-treated patients during the study period (40 days). All deaths occurred in patients less than 2 years of age and appeared to be related to underlying comorbidities [see Clinical Studies (14.2)].
Treatment discontinuation due to adverse reactions occurred in 7.9% (3/38) of patients in the Phase 2 trial, and in 1% (1/98) and 2.3% (1/44) of DIFICID- and vancomycin-treated patients, respectively, in the Phase 3 trial. The most common selected adverse reactions occurring in ≥5% of pediatric patients treated with DIFICID in the Phase 3 trial are listed in Table 3.
| System Organ Class | DIFICID (N=98) | Vancomycin (N=44) |
|---|---|---|
| Adverse Reaction | n (%) | n (%) |
| Gastrointestinal Disorders | ||
| Abdominal pain* | 8 (8.2) | 9 (20.5) |
| Vomiting | 7 (7.1) | 6 (13.6) |
| Diarrhea | 7 (7.1) | 5 (11.4) |
| Constipation | 5 (5.1) | 1 (2.3) |
| General Disorders and Administration Site Conditions | ||
| Pyrexia | 13 (13.3) | 10 (22.7) |
| Investigations | ||
| Aminotransferases increased† | 5 (5.1) | 1 (2.3) |
| Skin and Subcutaneous Tissue Disorders | ||
| Rash‡ | 5 (5.1) | 1 (2.3) |
The following adverse reactions were reported in <5% of pediatric patients taking DIFICID in clinical trials:
Skin and Subcutaneous Tissue Disorders: urticaria, pruritus
6.2 Post Marketing Experience
The following adverse reactions have been identified during post-approval use of DIFICID. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hypersensitivity reactions (dyspnea, angioedema, rash, pruritus)
7 DRUG INTERACTIONS
Fidaxomicin and its main metabolite, OP-1118, are substrates of the efflux transporter, P-glycoprotein (P-gp), which is expressed in the gastrointestinal tract.
7.1 Cyclosporine
Cyclosporine is an inhibitor of multiple transporters, including P-gp. When cyclosporine was co-administered with DIFICID, plasma concentrations of fidaxomicin and OP-1118 were significantly increased but remained in the ng/mL range [see Clinical Pharmacology (12.3)]. Concentrations of fidaxomicin and OP-1118 may also be decreased at the site of action (i.e., gastrointestinal tract) via P-gp inhibition; however, concomitant P-gp inhibitor use had no attributable effect on safety or treatment outcome of fidaxomicin-treated adult patients in controlled clinical trials. Based on these results, fidaxomicin may be co-administered with P-gp inhibitors and no dose adjustment is recommended.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
The limited available data on use of DIFICID in pregnant women are insufficient to inform any drug-associated risk for major birth defects, miscarriage or adverse maternal or fetal outcomes. Embryo-fetal reproduction studies in rats and rabbits dosed intravenously during organogenesis revealed no evidence of harm to the fetus at fidaxomicin and OP-1118 (its main metabolite) exposures 65-fold or higher than the clinical exposure at the DIFICID recommended dose [see Data].
The estimated background risk of major birth defects and miscarriage for the indicated populations is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Data
Animal Data
In pregnant rats, fidaxomicin was administered intravenously at doses of 4, 8, and 15 mg/kg/day from gestation day 6 through 17 (during the period of organogenesis). No embryo/fetal effects were noted in this study at exposures (AUC) 193-fold higher for fidaxomicin, and 65-fold higher for OP-1118 than the clinical exposure at the DIFICID recommended dose.
In pregnant rabbits, fidaxomicin was administered intravenously at doses of 2, 4, and 7.5 mg/kg/day from gestation day 6 through 18 (during the period of organogenesis). No embryo/fetal effects were noted in this study at exposures 66-fold higher for fidaxomicin, and 245-fold higher for OP-1118 than the clinical exposure at the DIFICID recommended dose.
8.2 Lactation
Risk Summary
There is no information on the presence of fidaxomicin or its main metabolite, OP-1118, in human milk, the effects on the breastfed infant, or the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for DIFICID and any potential adverse effects on the breastfed infant from DIFICID or from the underlying maternal condition.
8.4 Pediatric Use
The safety and effectiveness of DIFICID for the treatment of CDAD have been established in pediatric patients 6 months to less than 18 years of age. Use of DIFICID in these age groups is supported by evidence from adequate and well-controlled trials of DIFICID in adults with CDAD and pharmacokinetic, safety and efficacy data from pediatric trials [see Clinical Pharmacology (12.3), Clinical Studies (14.2)]. No new safety signals associated with the use of DIFICID in pediatric patients were identified in the pediatric trials [see Adverse Reactions (6.1)].
The safety and effectiveness of DIFICID have not been established in pediatric patients younger than 6 months of age.
8.5 Geriatric Use
Of the total number of patients in controlled trials of DIFICID, 50% were 65 years of age and over, while 31% were 75 and over. No overall differences in safety or effectiveness of DIFICID compared to vancomycin were observed between these subjects and younger subjects.
In controlled trials, elderly patients (≥65 years of age) had higher plasma concentrations of fidaxomicin and its main metabolite, OP-1118, versus non-elderly patients (<65 years of age) [see Clinical Pharmacology (12.3)]. However, greater exposures in elderly patients were not considered to be clinically significant. No dose adjustment is recommended for elderly patients.
10 OVERDOSAGE
No cases of acute overdose have been reported in humans. No drug-related adverse effects were seen in dogs dosed with fidaxomicin tablets at 9600 mg/day (over 100 times the human dose, scaled by weight) for 3 months.
11 DESCRIPTION
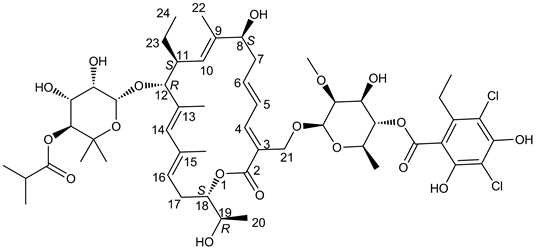
DIFICID (fidaxomicin) is a macrolide antibacterial drug for oral administration. Its CAS chemical name is Oxacyclooctadeca-3,5,9,13,15-pentaen-2-one, 3-[[[6-deoxy-4-O-(3,5-dichloro-2-ethyl-4,6-dihydroxybenzoyl)-2-O-methyl-β-D-mannopyranosyl]oxy]methyl]-12-[[6-deoxy-5-C-methyl-4-O-(2-methyl-1-oxopropyl)-β-D-lyxo-hexopyranosyl]oxy]-11-ethyl-8-hydroxy-18-[(1R)-1-hydroxyethyl]-9,13,15-trimethyl-, (3E,5E,8S,9E,11S,12R,13E,15E,18S)-. The structural formula of fidaxomicin is shown in Figure 1.
|
|
|
| Figure 1: Structural Formula of Fidaxomicin |
DIFICID tablets are film-coated and contain 200 mg of fidaxomicin per tablet and the following inactive ingredients: butylated hydroxytoluene, hydroxypropyl cellulose, lecithin (soy), magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, pregelatinized starch, sodium starch glycolate, talc, and titanium dioxide.
DIFICID for oral suspension is supplied as granules in bottles containing 5.45 g of fidaxomicin (40 mg of fidaxomicin per mL after reconstitution) and the following inactive ingredients: citric acid, microcrystalline cellulose, mixed berry flavor, sodium benzoate, sodium citrate, sodium starch glycolate, sucralose, and xanthan gum.
12 CLINICAL PHARMACOLOGY
12.2 Pharmacodynamics
Fidaxomicin acts locally in the gastrointestinal tract on C. difficile. In a dose-ranging trial (N=48) of fidaxomicin using 50 mg, 100 mg, and 200 mg twice daily for 10 days, a dose-response relationship was observed for efficacy.
12.3 Pharmacokinetics
The pharmacokinetic parameters of fidaxomicin and its main metabolite OP-1118 following a single dose of 200 mg in healthy adult males (N=14) are summarized in Table 4.
| Parameter | Fidaxomicin | OP-1118 | ||
|---|---|---|---|---|
| N | Value | N | Value | |
|
||||
| Cmax (ng/mL) | 14 | 5.20 ± 2.81 | 14 | 12.0 ± 6.06 |
| Tmax (h)* | 14 | 2.00 (1.00-5.00) | 14 | 1.02 (1.00-5.00) |
| AUC0-t (ng-h/mL) | 14 | 48.3 ± 18.4 | 14 | 103 ± 39.4 |
| AUC0-∞ (ng-h/mL) | 9 | 62.9 ± 19.5 | 10 | 118 ± 43.3 |
| t1/2 (h) | 9 | 11.7 ± 4.80 | 10 | 11.2 ± 3.01 |
Absorption
Fidaxomicin has minimal systemic absorption following oral administration, with plasma concentrations of fidaxomicin and OP-1118 in the ng/mL range at the therapeutic dose. In fidaxomicin-treated patients from controlled trials, plasma concentrations of fidaxomicin and OP-1118 obtained within the Tmax window (1-5 hours) were approximately 2- to 6-fold higher than Cmax values in healthy adults. Following administration of DIFICID 200 mg twice daily for 10 days, OP-1118 plasma concentrations within the Tmax window were approximately 50%-80% higher than on Day 1, while concentrations of fidaxomicin were similar on Days 1 and 10.
In a food-effect study involving administration of DIFICID to healthy adults (N=28) with a high-fat meal versus under fasting conditions, Cmax of fidaxomicin and OP-1118 decreased by 21.5% and 33.4%, respectively, while AUC0-t remained unchanged. This decrease in Cmax is not considered clinically significant, and thus, DIFICID may be administered with or without food.
Distribution
Fidaxomicin is mainly confined to the gastrointestinal tract following oral administration. In selected patients (N=8) treated with DIFICID 200 mg twice daily for 10 days from controlled trials, fecal concentrations of fidaxomicin and OP-1118 obtained within 24 hours of the last dose ranged from 639-2710 µg/g and 213-1210 µg/g, respectively. In contrast, plasma concentrations of fidaxomicin and OP-1118 within the Tmax window (1-5 hours) ranged 2-179 ng/mL and 10-829 ng/mL, respectively.
Elimination
Metabolism
Fidaxomicin is primarily transformed by hydrolysis at the isobutyryl ester to form its main and microbiologically active metabolite, OP-1118. Metabolism of fidaxomicin and formation of OP-1118 are not dependent on cytochrome P450 (CYP) enzymes.
At the therapeutic dose, OP-1118 was the predominant circulating compound in healthy adults, followed by fidaxomicin.
Excretion
Fidaxomicin is mainly excreted in feces. In one trial of healthy adults (N=11), more than 92% of the dose was recovered in the stool as fidaxomicin and OP-1118 following single doses of 200 mg and 300 mg. In another trial of healthy adults (N=6), 0.59% of the dose was recovered in urine as OP-1118 only following a single dose of 200 mg.
Specific Populations
Geriatric Patients
In controlled trials of patients treated with DIFICID 200 mg twice daily for 10 days, mean and median values of fidaxomicin and OP-1118 plasma concentrations within the Tmax window (1-5 hours) were approximately 2- to 4-fold higher in elderly patients (≥65 years of age) versus non-elderly patients (<65 years of age). Despite greater exposures in elderly patients, fidaxomicin and OP-1118 plasma concentrations remained in the ng/mL range [see Use in Specific Populations (8.5)].
Pediatric Patients
Similar to adults, fidaxomicin has minimal systemic absorption following oral administration across all age groups in pediatric patients. Plasma concentrations remained in the ng/mL range at the therapeutic dose in pediatric patients with mean (± standard deviation) plasma concentrations of 39.41 (±62.15) ng/mL of fidaxomicin and 116.64 (±259.10) ng/mL of OP-1118 at 1 to 5 hours post-dose.
Male and Female Patients
Plasma concentrations of fidaxomicin and OP-1118 within the Tmax window (1-5 hours) did not vary by gender in patients treated with DIFICID 200 mg twice daily for 10 days from controlled trials. No dose adjustment is recommended based on gender.
Patients with Renal Impairment
In controlled trials of patients treated with DIFICID 200 mg twice daily for 10 days, plasma concentrations of fidaxomicin and OP-1118 within the Tmax window (1-5 hours) did not vary by severity of renal impairment (based on creatinine clearance) between mild (51-79 mL/min), moderate (31-50 mL/min), and severe (≤30 mL/min) categories. No dose adjustment is recommended based on renal function.
Patients with Hepatic Impairment
The impact of hepatic impairment on the pharmacokinetics of fidaxomicin has not been evaluated. Because fidaxomicin and OP-1118 do not appear to undergo significant hepatic metabolism, elimination of fidaxomicin and OP-1118 is not expected to be significantly affected by hepatic impairment.
Drug Interaction Studies
In vivo studies were conducted to evaluate intestinal drug-drug interactions of fidaxomicin as a P-gp substrate, P-gp inhibitor, and inhibitor of major CYP enzymes expressed in the gastrointestinal tract (CYP3A4, CYP2C9, and CYP2C19).
Table 5 summarizes the impact of a co-administered drug (P-gp inhibitor) on the pharmacokinetics of fidaxomicin [see Drug Interactions (7.1)].
| Parameter | Cyclosporine 200 mg + Fidaxomicin 200 mg*
(N=14) | Fidaxomicin 200 mg Alone (N=14) | Mean Ratio of Parameters With/Without Co-Administered Drug (90% CI †) No Effect = 1.00 |
||
|---|---|---|---|---|---|
| N | Mean | N | Mean | ||
| Fidaxomicin | |||||
| Cmax (ng/mL) | 14 | 19.4 | 14 | 4.67 | 4.15 (3.23-5.32) |
| AUC0-∞ (ng-h/mL) | 8 | 114 | 9 | 59.5 | 1.92 (1.39-2.64) |
| OP-1118 | |||||
| Cmax (ng/mL) | 14 | 100 | 14 | 10.6 | 9.51 (6.93-13.05) |
| AUC0-∞ (ng-h/mL) | 12 | 438 | 10 | 106 | 4.11 (3.06-5.53) |
Fidaxomicin had no significant impact on the pharmacokinetics of the following co-administered drugs: digoxin (P-gp substrate), midazolam (CYP3A4 substrate), warfarin (CYP2C9 substrate), and omeprazole (CYP2C19 substrate). No dose adjustment is warranted when fidaxomicin is co-administered with substrates of P-gp or CYP enzymes.
12.4 Microbiology
Mechanism of Action
Fidaxomicin is a fermentation product obtained from the Actinomycete Dactylosporangium aurantiacum. Fidaxomicin is a macrolide antibacterial drug that inhibits RNA synthesis by binding to RNA polymerases. Fidaxomicin is bactericidal against C. difficile in vitro, and demonstrates a post-antibiotic effect vs. C. difficile of 6-10 hrs.
Resistance
Fidaxomicin demonstrates no in vitro cross-resistance with other classes of antibacterial drugs. In vitro studies indicate a low frequency of spontaneous resistance to fidaxomicin in C. difficile (ranging from <1.4 × 10-9 to 12.8 × 10-9). A specific mutation (Val-ll43-Gly) in the beta subunit of RNA polymerase is associated with reduced susceptibility to fidaxomicin. This mutation was created in the laboratory and seen during clinical trials in a C. difficile isolate obtained from an adult subject treated with DIFICID who had recurrence of CDAD. The fidaxomicin minimum inhibitory concentration (MIC) of the C. difficile isolate from this subject increased from a baseline of 0.06 µg/mL to 16 µg/mL at the time of CDAD recurrence.
Interaction With Other Antimicrobials
Fidaxomicin and its main metabolite OP-1118 do not exhibit any antagonistic interaction with other classes of antibacterial drugs. Synergistic interactions of fidaxomicin and OP-1118 have been observed in vitro with rifampin and rifaximin against C. difficile.
Antimicrobial Activity
Fidaxomicin has been shown to be active against most isolates of Clostridioides (formerly Clostridium) difficile, both in vitro and in clinical infections [see Indications and Usage (1)].
Susceptibility Testing
For specific information regarding susceptibility test interpretive criteria, and associated test methods and quality control standards recognized by FDA for this drug, please see: https://www.fda.gov/STIC .
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term carcinogenicity studies have not been conducted to evaluate the carcinogenic potential of fidaxomicin.
Neither fidaxomicin nor OP-1118 was mutagenic in the Ames assay. Fidaxomicin was also negative in the rat micronucleus assay. However, fidaxomicin was clastogenic in Chinese hamster ovary cells.
Fidaxomicin did not affect the fertility of male and female rats at intravenous doses of 6.3 mg/kg. The exposure (AUC0-t) was approximately 100 times that in humans.
14 CLINICAL STUDIES
14.1 Clinical Studies of DIFICID in Adult Patients with CDAD
In two randomized, double-blinded trials, a non-inferiority design was utilized to demonstrate the efficacy of DIFICID (200 mg tablets twice daily for 10 days) compared to vancomycin (125 mg four times daily for 10 days) in adults with CDAD.
Enrolled patients were 18 years of age or older and received no more than 24 hours of pretreatment with vancomycin or metronidazole. CDAD was defined by >3 unformed bowel movements (or >200 mL of unformed stool for subjects having rectal collection devices) in the 24 hours before randomization, and presence of either C. difficile toxin A or B in the stool within 48 hours of randomization. Enrolled patients had either no prior CDAD history or only one prior CDAD episode in the past three months. Subjects with life-threatening/fulminant infection, hypotension, septic shock, peritoneal signs, significant dehydration, or toxic megacolon were excluded.
The demographic profile and baseline CDAD characteristics of enrolled subjects were similar in the two trials. Patients had a median age of 64 years, were mainly white (90%), female (58%), and inpatients (63%). The median number of bowel movements per day was 6, and 37% of subjects had severe CDAD (defined as 10 or more unformed bowel movements per day or WBC ≥15000/mm3). Diarrhea alone was reported in 45% of patients and 84% of subjects had no prior CDAD episode.
The primary efficacy endpoint was the clinical response rate at the end of treatment, based upon improvement in diarrhea or other symptoms such that, in the investigator's judgment, further CDAD treatment was not needed. An additional efficacy endpoint was sustained clinical response 25 days after the end of treatment. Sustained response was evaluated only for patients who were clinical successes at the end of treatment. Sustained response was defined as clinical response at the end of treatment, and survival without proven or suspected CDAD recurrence through 25 days beyond the end of treatment.
The results for clinical response at the end of treatment in both trials, shown in Table 6, indicate that DIFICID is non-inferior to vancomycin based on the 95% confidence interval (CI) lower limit being greater than the non-inferiority margin of -10%.
The results for sustained clinical response at the end of the follow-up period, also shown in Table 6, indicate that DIFICID is superior to vancomycin on this endpoint. Since clinical success at the end of treatment and mortality rates were similar across treatment arms (approximately 6% in each group), differences in sustained clinical response were due to lower rates of proven or suspected CDAD during the follow-up period in DIFICID patients.
| Clinical Response at End of Treatment | Sustained Response at 25 days Post-Treatment | |||||
|---|---|---|---|---|---|---|
| DIFICID % (N) | Vancomycin % (N) | Difference (95% CI)* | DIFICID % (N) | Vancomycin % (N) | Difference (95% CI)* |
|
|
||||||
| Trial 1 | 88% (N=289) | 86% (N=307) | 2.6% (-2.9%, 8.0%) | 70% (N=289) | 57% (N=307) | 12.7% (4.4%, 20.9%) |
| Trial 2 | 88% (N=253) | 87% (N=256) | 1.0% (-4.8%, 6.8%) | 72% (N=253) | 57% (N=256) | 14.6% (5.8%, 23.3%) |
Restriction Endonuclease Analysis (REA) was used to identify C. difficile baseline isolates in the BI group, isolates associated with increasing rates and severity of CDAD in the US in the years prior to the clinical trials. Similar rates of clinical response at the end of treatment and proven or suspected CDAD during the follow-up period were seen in fidaxomicin-treated and vancomycin-treated patients infected with a BI isolate. However, DIFICID did not demonstrate superiority in sustained clinical response when compared with vancomycin (Table 7).
|
|||
| Trial 1 | |||
| Initial C. difficile Group | DIFICID n/N (%) | Vancomycin n/N (%) | Difference (95% CI)* |
| BI Isolates | 44/76 (58%) | 52/82 (63%) | -5.5% (-20.3%, 9.5%) |
| Non-BI Isolates | 105/126 (83%) | 87/131 (66%) | 16.9% (6.3%, 27.0%) |
| Trial 2 | |||
| Initial C. difficile Group | DIFICID n/N (%) | Vancomycin n/N (%) | Difference (95% CI)* |
| BI Isolates | 42/65 (65%) | 31/60 (52%) | 12.9% (-4.2%, 29.2%) |
| Non-BI Isolates | 109/131 (83%) | 77/121 (64%) | 19.6% (8.7%, 30.0%) |
14.2 Clinical Studies of DIFICID in Pediatric Patients with CDAD
The safety and efficacy of DIFICID in pediatric patients 6 months to less than 18 years of age was investigated in a Phase 3, multicenter, investigator-blinded, randomized, comparative trial (NCT02218372). In this trial, 148 patients were randomized, of whom 142 received either DIFICID or vancomycin in a 2:1 ratio. Randomized patients were stratified by age group as follows: 30 aged 6 months to <2 years, 49 aged 2 to <6 years, 40 aged 6 to <12 years, and 29 aged 12 to <18 years (one patient <6 months of age was enrolled in the trial). Treatment arms were balanced regarding demographics and other baseline characteristics.
Clinical response for patients <2 years of age was defined as the absence of watery stools for at least 2 consecutive days while on treatment and the patient remained well with no requirement for further CDAD therapy through 2 days after completing treatment as assessed by the Investigator. Clinical response for patients ≥2 to <18 years of age was defined as <3 unformed bowel movements for at least 2 consecutive days while on treatment and the patient remained well with no requirement for further CDAD therapy through 2 days after completing treatment as assessed by the Investigator. Sustained clinical response was defined as the proportion of treated patients with confirmed clinical response and no CDAD recurrence through 30 days after end of treatment. The clinical response and sustained clinical response overall and by age groups are presented in Table 8.
| Clinical Response | Sustained Response at 30 days Post-Treatment | |||||
|---|---|---|---|---|---|---|
| DIFICID n/N (%) | Vancomycin n/N (%) | Difference (95% CI) | DIFICID n/N (%) | Vancomycin n/N (%) | Difference (95% CI) |
|
| Overall | 76/98 (77.6) | 31/44 (70.5) | 7.5 (-7.4, 23.9) | 67/98 (68.4) | 22/44 (50.0) | 18.4 (1.5, 35.3) |
| <2 years | 13/20 (65.0) | 9/10 (90.0) | 11/20 (55.0) | 7/10 (70.0) | ||
| ≥2 to <6 years | 25/32 (78.1) | 12/16 (75.0) | 21/32 (65.6) | 8/16 (50.0) | ||
| ≥6 to <12 years | 23/26 (88.5) | 5/10 (50.0) | 22/26 (84.6) | 4/10 (40.0) | ||
| ≥12 to <18 years | 15/20 (75.0) | 5/8 (62.5) | 13/20 (65.0) | 3/8 (37.5) | ||
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
Tablets
DIFICID tablets are white to off-white film-coated, oblong tablets containing 200 mg of fidaxomicin per tablet; each tablet is debossed with "FDX" on one side and "200" on the other side.
DIFICID tablets are supplied as bottles of 20 tablets (NDC 52015-080-01).
Granules for Oral Suspension
DIFICID granules for oral suspension are white to yellowish white.
DIFICID granules for oral suspension (NDC 52015-700-22) is supplied as 150 mL amber glass bottles of 9.53 g of granules that contain 5.45 g of fidaxomicin. Each glass bottle has a child-resistant cap and is sealed in a laminated aluminum foil pouch. After reconstitution, the total oral suspension volume is 136 mL. Discard unused suspension after 12 days. The concentration of fidaxomicin is 40 mg/mL (200 mg per 5 mL) in the reconstituted oral suspension.
16.2 Storage
Tablets
Store DIFICID tablets at 20°C-25°C (68°F-77°F); excursions permitted to 15°C-30°C (59°F-86°F). See USP controlled room temperature. Store in the original bottle.
Granules for oral suspension
Store DIFICID granules for oral suspension at 20°C-25°C (68°F-77°F); excursions permitted to 15°C-30°C (59°F-86°F). Store in the original package. Do not open pouch until time of use.
Once reconstituted, store DIFICID oral suspension refrigerated at 2°C-8°C (36°F-46°F) for up to 12 days. Store capped in the original bottle.
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Oral Suspension
Remove the bottle from the refrigerator 15 minutes prior to each administration.
Instruct patients or caregivers to use an oral dosing syringe to correctly measure the prescribed amount of medication. Inform patients or caregivers that oral dosing syringes may be obtained from their pharmacy.
Inform the patients or caregivers that DIFICID oral suspension should be prepared by a healthcare professional. Advise them to contact a healthcare professional for any questions regarding administration of DIFICID oral suspension.
Administration with Food
Inform patients and caregivers that DIFICID tablets and oral suspension may be taken with or without food.
Antibacterial Resistance
Patients should be counseled that antibacterial drugs, including DIFICID, should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When DIFICID is prescribed to treat a C. difficile infection, patients should be told that, although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by DIFICID or other antibacterial drugs in the future.
Manufactured for: Merck Sharp & Dohme LLC
Rahway, NJ 07065, USA
For patent information: www.msd.com/research/patent
Copyright © 2015-2022 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates.
All rights reserved.
uspi-mk5119-mf-2206r007
| Patient Information DIFICID® (dih-fih-sid) (fidaxomicin) tablets, for oral use DIFICID® (dih-fih-sid) (fidaxomicin) for oral suspension |
||
What You Need to Know About Your Medicine
|
||
| What is DIFICID? | ||
DIFICID is an antibiotic medicine used to treat an infection called Clostridioides difficile-associated diarrhea (CDAD) in adults and children 6 months of age and older. Clostridioides difficile (C-diff) is a bacterium that can cause an infection that can damage your colon and cause stomach pain and severe diarrhea.
|
||
| Who should not take DIFICID? | ||
| Do not take DIFICID if you are allergic to fidaxomicin, or any other ingredient in DIFICID. See the end of this Patient Information for a complete list of ingredients in DIFICID. | ||
| What should I tell my doctor before taking DIFICID? | ||
Pregnancy
|
||
| How do I take DIFICID? | ||
|
||
| What are the possible side effects of DIFICID? | ||
DIFICID can cause serious side effects, including:
The most common side effects of DIFICID in adults include: |
||
|
|
|
| The most common side effects of DIFICID in children include: | ||
|
|
|
| Other less common side effects of DIFICID may include: | ||
|
||
|
|
|
| If you have any side effect that bothers you or does not go away, tell your doctor. | ||
| There may be other side effects to DIFICID that are not listed. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | ||
| How should I store DIFICID? | ||
DIFICID tablets
|
||
| General information about the safe and effective use of DIFICID. | ||
| Medicines are sometimes prescribed for purposes other than those listed in the Patient Information. Do not take DIFICID for a condition for which it was not prescribed. Do not give DIFICID to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or doctor for information about DIFICID that is written for health professionals. | ||
| What if I have questions? | ||
|
||
| What are the ingredients in DIFICID? | ||
DIFICID tablets
|
||
Manuf. for: Merck Sharp & Dohme LLC
Rahway, NJ 07065, USA
For patent information: www.msd.com/research/patent. The trademarks depicted herein are owned by their respective companies.
Copyright © 2019-2022 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates.
All rights reserved.
usppi-mk5119-mf-2206r002
This Patient Information has been approved by the U.S. Food and Drug Administration.
Revised: 06/2022
PRINCIPAL DISPLAY PANEL - 200 mg Tablet Bottle Carton
NDC 52015-080-01 20 tablets
DIFICID®
(fidaxomicin) tablets
200 mg per tablet
Rx only

PRINCIPAL DISPLAY PANEL - 40 mg/mL Bottle Pouch Carton
NDC 52015-700-22
DIFICID®
(fidaxomicin) for oral suspension
40 mg/mL
MUST BE RECONSTITUTED BY A PHARMACIST
BEFORE DISPENSING
See enclosed package insert for preparation instructions.
For pediatric use
For oral administration only
Each mL contains 40 mg fidaxomicin after reconstitution
with 130 mL purified water. (136 mL total volume
after reconstitution)
Mixed berry flavored Rx only