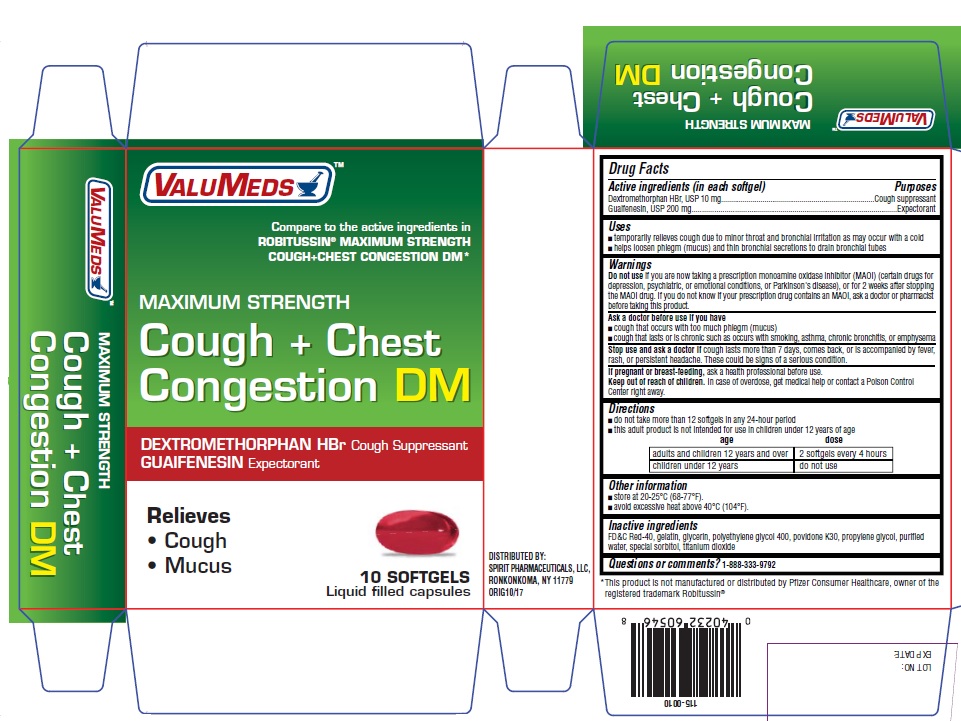
Uses
- temporarily relieves cough due to minor throat and bronchial irritation as may occur with a cold
- helps loosen phlegm (mucus) and thin bronchial secretions to drain bronchial tubes
Warnings
Do not use if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- cough that occurs with too much phlegm (mucus)
- cough that lasts or is chronic such as occurs with smoking, asthma, chronic bronchitis, or emphysema
Directions
- do not take more than 12 softgels in any 24-hour period
- this adult product is not intended for use in children under 12 years of age
| age | dose |
|---|---|
| adults and children 12 years and over | 2 softgels every 4 hours |
| children under 12 years | do not use |
Inactive ingredients
FD&C Red-40, gelatin, glycerin, polyethylene glycol 400, povidone K30, propylene glycol, purified water, special sorbitol, titanium dioxide
PRINCIPAL DISPLAY PANEL
VALUMEDS TM
Compare to the active ingredients in Robitussin® Maximum
Strength Cough+Chest Congestion DM*
Maximum Strength
Cough + Chest Congestion DM
DEXTROMETHORPHAN HBr / COUGH SUPPRESSANT
GUAIFENESIN/ EXPECTORANT
MAXIMUM STRENGTH
Relieves
- cough
- mucus
10 SOFTGELS
Liquid Filled capsules