Warnings
For use in the ear only
Ask a Doctor before use if you have
- ear drainage or discharge
- ear pain
- irritation or rash in the ear
- dizziness
- an injury or perforation (hole) of the eardrum
- recently had ear surgery
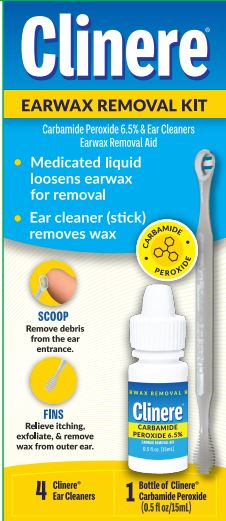
Directions
FOR USE IN THE EAR ONLY.
Adults and children over 12 years of age:
- tilt head sideways and place 5-10 drops into each ear canal.
- Tip of applicator should not enter ear canal.
- Keep drops in ear for several minutes by keeping head tilted or placing cotton in ear.
- Use twice daily for up to four days if needed, or as directed by a doctor.
- Any wax remaining in the ear may be removed by gently flushing the ear with warm water, using a soft rubber bulb ear syringe, or by use of the included Clinere® ear cleaning tool. (See separate directions)
Children under 12 years: Consult a doctor.
Other Information
- Store at 15° - 30°C (59° - 86°F)
- Avoid exposing bottle to excessive heat or direct sunlight
- Store bottle in the outer carton
- Keep cap on bottle when not in use
- Product foams on contact with ear wax due to release of oxygen. There may be an associated crackling sound.