FULL PRESCRIBING INFORMATION
1. INDICATIONS AND USAGE
1.1 Indication
Clobetasol Propionate Topical Spray, 0.05% is a super-high potent topical corticosteroid formulation indicated for the treatment of moderate to severe plaque psoriasis affecting up to 20% body surface area (BSA) in patients 18 years of age or older. The total dosage should not exceed 50 g (59 mL or 2 fl. oz.) per week. Do not use more than 26 sprays per application or 52 sprays per day. Treatment should be limited to 4 consecutive weeks.
Patients should be instructed to use Clobetasol Propionate Topical Spray, 0.05% for the minimum amount of time necessary to achieve the desired results [see Dosage and Administration (2)].
Use in patients under 18 years of age is not recommended because safety has not been established and because numerically high rates of HPA axis suppression were seen with other clobetasol propionate topical formulations.
1.2 Limitations of Use
Clobetasol Propionate Topical Spray, 0.05% should not be used on the face, axillae, or groin. Clobetasol Propionate Topical Spray, 0.05% should not be used if there is atrophy at the treatment site. Clobetasol Propionate Topical Spray, 0.05% should not be used in the treatment of rosacea or perioral dermatitis.
2. DOSAGE AND ADMINISTRATION
Clobetasol Propionate Topical Spray, 0.05% is for topical use only, and not for ophthalmic, oral or intravaginal use.
Clobetasol Propionate Topical Spray, 0.05% should be sprayed directly onto the affected skin areas twice daily and rubbed in gently and completely.
The total dosage should not exceed 50 g (59 mL or 2 fluid ounces) per week because of the potential for the drug to suppress the hypothalamic-pituitary-adrenal (HPA) axis. Do not use more than 26 sprays per application or 52 sprays per day.
Clobetasol Propionate Topical Spray, 0.05% contains a topical corticosteroid; therefore treatment should be limited to 4 weeks. Therapy should be discontinued when control has been achieved. Treatment beyond 2 weeks should be limited to localized lesions of moderate to severe plaque psoriasis that have not sufficiently improved after the initial 2 weeks of treatment with Clobetasol Propionate Topical Spray, 0.05%. If no improvement is seen within 2 weeks, reassessment of diagnosis may be necessary. Before prescribing for more than 2 weeks, any additional benefits of extending treatment to 4 weeks should be weighed against the risk of HPA axis suppression. Use in pediatric patients younger than 18 years is not recommended because of the potential for HPA axis suppression [see Use in Specific Populations (8.4)].
Unless directed by physician, Clobetasol Propionate Topical Spray, 0.05% should not be used with occlusive dressings.
3. DOSAGE FORMS AND STRENGTHS
Spray, 0.05% w/w. Each gram of Clobetasol Propionate Topical Spray, 0.05% contains 0.5 mg of clobetasol propionate in a clear, colorless liquid.
5. WARNINGS AND PRECAUTIONS
5.1 Effects on the Endocrine System
Clobetasol propionate is a highly potent topical corticosteroid that has been shown to suppress the HPA axis at the lowest doses tested. Systemic absorption of topical corticosteroids can produce reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for clinical glucocorticosteroid insufficiency. This may occur during treatment or upon withdrawal of the topical corticosteroid. In studies evaluating the potential for hypothalamic-pituitary-adrenal (HPA) axis suppression, using the Cosyntropin Stimulation Test, Clobetasol Propionate Topical Spray, 0.05% demonstrated rates of suppression that were comparable after 2 and 4 weeks of twice-daily use (19% and 15 to 20%, respectively), in adult patients with moderate to severe plaque psoriasis (≥ 20% BSA). In these studies, HPA axis suppression was defined as serum cortisol level ≤18 μg/dL 30-min post cosyntropin stimulation [see Clinical Pharmacology (12)].
Because of the potential for systemic absorption, use of topical corticosteroids may require that patients be periodically evaluated for HPA axis suppression. Factors that predispose a patient using a topical corticosteroid to HPA axis suppression include the use of more potent steroids, use over large surface areas, use over prolonged periods, use under occlusion, use on an altered skin barrier, and use in patients with liver failure.
An ACTH stimulation test may be helpful in evaluating patients for HPA axis suppression. If HPA axis suppression is documented, an attempt should be made to gradually withdraw the drug, to reduce the frequency of application, or to substitute a less potent steroid. Manifestations of adrenal insufficiency may require supplemental systemic corticosteroids. Recovery of HPA axis function is generally prompt and complete upon discontinuation of topical corticosteroids.
Cushing's syndrome, hyperglycemia, and unmasking of latent diabetes mellitus can also result from systemic absorption of topical corticosteroids.
Use of more than one corticosteroid-containing product at the same time may increase the total systemic corticosteroid exposure. Pediatric patients may be more susceptible to systemic toxicity from use of topical corticosteroids. [see Use in Specific Populations (8.4)]
5.2 Local Adverse Reactions with Topical Corticosteroids
The following additional local adverse reactions have been reported with topical corticosteroids. They may occur more frequently with the use of occlusive dressings and higher potency corticosteroids, including clobetasol propionate. These reactions are listed in an approximate decreasing order of occurrence: folliculitis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, secondary infection, striae and miliaria.
5.3 Allergic Contact Dermatitis
Allergic contact dermatitis to any component of topical corticosteroids is usually diagnosed by a failure to heal rather than a clinical exacerbation. Clinical diagnosis of allergic contact dermatitis can be confirmed by patch testing.
5.4 Concomitant Skin Infections
In the presence of dermatological infections, the use of an appropriate antifungal or antibacterial agent should be instituted. If a favorable response does not occur promptly, use of Clobetasol Propionate Topical Spray, 0.05% should be discontinued until the infection has been adequately controlled.
6. ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In controlled, clinical trials with Clobetasol Propionate Topical Spray, 0.05%, the most common adverse reaction was burning at the site of application [40% of subjects treated with Clobetasol Propionate Topical Spray, 0.05% and 47% of subjects treated with Spray Vehicle]. Other commonly reported adverse reactions for Clobetasol Propionate Topical Spray, 0.05% and Spray Vehicle, respectively, are noted in Table 1.
| Adverse Reaction | Clobetasol Propionate Topical Spray, 0.05% (N=120) | Vehicle Spray (N=120) |
|---|---|---|
| System Organ Class | ||
| General disorders and administration site conditions | 50 (42%) | 56 (47%) |
| Application site burning | 48 (40%) | 56 (47%) |
| Application site dryness | 2 (2%) | 0 (0%) |
| Application site irritation | 1 (1%) | 0 (0%) |
| Application site pain | 1 (1%) | 2 (2%) |
| Application site pigmentation changes | 1 (1%) | 0 (0%) |
| Application site pruritus | 4 (3%) | 3 (3%) |
| Infections and infestations | 17 (14%) | 12 (10%) |
| Nasopharyngitis | 6 (5%) | 3 (3%) |
| Pharyngitis streptococcal | 1 (1%) | 0 (0%) |
| Upper respiratory tract infection | 10 (8%) | 2 (2%) |
| Skin and subcutaneous tissue disorders | 4 (3%) | 2 (2%) |
| Eczema asteatotic | 2 (2%) | 0 (0%) |
Most local adverse reactions were rated as mild to moderate and they are not affected by age, race or gender.
Systemic absorption of topical corticosteroids has produced hypothalamic-pituitary-adrenal (HPA) axis suppression, manifestations of Cushing's syndrome, hyperglycemia, and glycosuria in some patients.
6.2 Postmarketing Experience
Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The following adverse reactions have been identified during post-approval use of Clobetasol Propionate Topical Spray, 0.05%.
Skin: Burning, pruritus, erythema, pain, irritation, rash, peeling, urticaria, and contact dermatitis.
8. USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Teratogenic Effects: Pregnancy Category C:
There are no adequate and well-controlled studies in pregnant women. Clobetasol Propionate Topical Spray, 0.05% should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Corticosteroids have been shown to be teratogenic in laboratory animals when administered systemically at relatively low dosage levels. Some corticosteroids have been shown to be teratogenic after dermal application to laboratory animals.
Clobetasol propionate is absorbed percutaneously, and when administered subcutaneously it was a significant teratogen in both the rabbit and the mouse.
Clobetasol propionate has greater teratogenic potential than steroids that are less potent.
The effect of clobetasol propionate on pregnancy outcome and development of offspring was studied in the rat. Clobetasol propionate was administered subcutaneously to female rats twice daily (0, 12.5, 25, and 50 μg/kg/day) from day 7 of presumed gestation through day 25 of lactation or day 24 presumed gestation for those rats that did not deliver a litter. The maternal NOEL for clobetasol propionate was less than 12.5 μg/kg/day due to reduced body weight gain and feed consumption during the gestation period. The reproductive NOEL in the dams was 25 μg/kg/day (ratio of animal dose to proposed human dose of 0.07 on a mg/m2/day basis) based on prolonged delivery at a higher dose level. The no-observed-adverse-effect-level (NOAEL) for viability and growth in the offspring was 12.5 μg/kg/day (ratio of animal dose to proposed human dose of 0.03 on a mg/m2/day basis) based on incidence of stillbirths, reductions in pup body weights on days 1 and 7 of lactation, increased pup mortality, increases in the incidence of umbilical hernia, and increases in the incidence of pups with cysts on the kidney at higher dose levels during the preweaning period. The weights of the epididymides and testes were significantly reduced at higher dosages. Despite these changes, there were no effects on the mating and fertility of the offspring.
8.3 Nursing Mothers
Systemically administered corticosteroids appear in human milk and could suppress growth, interfere with endogenous corticosteroid production, or cause other untoward effects. It is not known whether topical administration of corticosteroids could result in sufficient systemic absorption to produce detectable quantities in breast milk. Because many drugs are excreted in human milk, caution should be exercised when Clobetasol Propionate Topical Spray, 0.05% is administered to a nursing woman.
8.4 Pediatric Use
Use in patients under 18 years of age is not recommended, because safety has not been established and because numerically high rates of HPA axis suppression were seen with other clobetasol propionate topical formulations. Safety and effectiveness in pediatric patients treated with Clobetasol Propionate Topical Spray, 0.05% have not been established [see Warnings and Precautions (5.1)].
Because of higher ratio of skin surface area to body mass, pediatric patients are at a greater risk than adults of HPA axis suppression and Cushing's syndrome when they are treated with topical corticosteroids. They are therefore also at greater risk of glucocorticosteroid insufficiency during and/or after withdrawal of treatment. Adverse effects including striae have been reported with inappropriate use of topical corticosteroids in infants and children.
HPA axis suppression, Cushing's syndrome, linear growth retardation, delayed weight gain, and intracranial hypertension have been reported in children receiving topical corticosteroids. Manifestations of adrenal suppression in children include low plasma cortisol levels and absence of response to ACTH stimulation. Manifestations of intracranial hypertension include bulging fontanelles, headaches, and bilateral papilledema.
8.5 Geriatric Use
Clinical studies of Clobetasol Propionate Topical Spray, 0.05% did not include sufficient numbers of patients aged 65 and over to adequately determine whether they respond differently than younger patients. In two randomized, vehicle controlled clinical trials, 21 of the 240 patients (9%) were over the age of 65. In general, dose selection for an elderly patient should be made with caution, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal or cardiac function, and of concomitant disease or other drug therapy.
10. OVERDOSAGE
Topically applied Clobetasol Propionate Topical Spray, 0.05% can be absorbed in sufficient amount to produce systemic effects [see Warnings and Precautions (5.1)].
11. DESCRIPTION
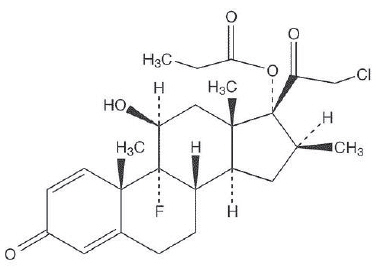
Clobetasol Propionate Topical Spray, 0.05% contains clobetasol propionate, a synthetic fluorinated corticosteroid, for topical use. The corticosteroids constitute a class of primarily synthetic steroids used topically as anti-inflammatory and antipruritic agents. Clobetasol propionate is 21-chloro-9-fluoro-11β, 17-dihydroxy-16β -methylpregna-1,4-diene-3,20-dione 17-propionate, with the empirical formula C25H32ClFO5, and a molecular weight of 466.97 (CAS Registry Number 25122-46-7). The following is the chemical structure:
Clobetasol propionate is a white to almost white crystalline powder that is practically insoluble in water. Each gram of Clobetasol Propionate Topical Spray, 0.05% contains 0.5 mg of clobetasol propionate, in a clear, colorless liquid composed of alcohol, isopropyl myristate, sodium lauryl sulfate, and undecylenic acid.
12. CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Like other topical corticosteroids Clobetasol Propionate Topical Spray, 0.05% has anti-inflammatory, antipruritic, and vasoconstrictive properties. The mechanism of the anti-inflammatory activity of the topical steroids in general is unclear. However, corticosteroids are thought to act by induction of phospholipase A2 inhibitory proteins, collectively called lipocortins. It is postulated that these proteins control the biosynthesis of potent mediators of inflammation such as prostaglandins and leukotrienes by inhibiting the release of their common precursor, arachidonic acid. Arachidonic acid is released from membrane phospholipids by phospholipase A2.
12.2 Pharmacodynamics
Vasoconstrictor Assay
Clobetasol Propionate Topical Spray, 0.05% is in the super-high range of potency as demonstrated in a vasoconstrictor study in healthy subjects when compared with other topical corticosteroids. However, similar blanching scores do not necessarily imply therapeutic equivalence.
Hypothalamic-Pituitary-Adrenal (HPA) Axis Suppression
The effect of Clobetasol Propionate Topical Spray, 0.05% on hypothalamic-pituitary-adrenal (HPA) axis function was investigated in adults in two studies. In the first study, patients with plaque psoriasis covering at least 20% of their body applied Clobetasol Propionate Topical Spray, 0.05% twice daily for up to 4 weeks. 15% (2 out of 13) of patients displayed adrenal suppression after 4 weeks of use based on the Cosyntropin Stimulation Test. The laboratory suppression was transient; all subjects returned to normal after cessation of drug use. In the second study, patients with plaque psoriasis covering at least 20% of their body applied Clobetasol Propionate Topical Spray, 0.05% twice daily for either 2 or 4 weeks. 19% (4 out of 21) of patients treated for 2 weeks and 20% (3 out of 15) of patients treated for 4 weeks displayed adrenal suppression at the end of treatment based on the Cosyntropin Stimulation Test. The laboratory suppression was transient; all subjects returned to normal after cessation of drug use. In these studies, HPA axis suppression was defined as serum cortisol level ≤ 18 μg/dL 30-min post cosyntropin (ACTH 1-24) stimulation [see Warnings and Precautions (5)].
12.3 Pharmacokinetics
The extent of percutaneous absorption of topical corticosteroids is determined by many factors, including the vehicle, the integrity of the epidermal barrier and occlusion.
Topical corticosteroids can be absorbed from normal intact skin. Inflammation and other disease processes in the skin may increase percutaneous absorption.
There are no human data regarding the distribution of corticosteroids to body organs following topical application. Nevertheless, once absorbed through the skin, topical corticosteroids are handled through metabolic pathways similar to systemically administered corticosteroids. They are metabolized, primarily in the liver, and are then excreted by the kidneys. In addition, some corticosteroids and their metabolites are also excreted in the bile.
13. NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Clobetasol propionate was not carcinogenic to rats when topically applied for 2 years at concentrations up to 0.005% which corresponded to doses up to 11 μg/kg/day (ratio of animal dose to proposed human dose of 0.03 on a mg/m2/day basis).
Clobetasol propionate at concentrations up to 0.001% did not increase the rate of formation of ultra violet light-induced skin tumors when topically applied to hairless mice 5 days per week for a period of 40 weeks. Clobetasol propionate was negative in the in vitro mammalian chromosomal aberration test and in the in vivo mammalian erythrocyte micronucleus test.
The effect of subcutaneously administered clobetasol propionate on fertility and general reproductive toxicity was studied in rats at doses of 0, 12.5, 25, and 50 μg/kg/day. Males were treated beginning 70 days before mating and females beginning 15 days before mating through day 7 of gestation. A dosage level of less than 12.5 μg/kg/day clobetasol propionate was considered to be the no-observed-effect-level (NOEL) for paternal and maternal general toxicity based on decreased weight gain and for male reproductive toxicity based on increased weights of the seminal vesicles with fluid. The female reproductive NOEL was 12.5 μg/kg/day (ratio of animal dose to proposed human dose of 0.03 on a mg/m2/day basis) based on reduction in the numbers of estrous cycles during the pre-cohabitation period and an increase in the number of nonviable embryos at higher doses.
14. CLINICAL STUDIES
The efficacy of Clobetasol Propionate Topical Spray, 0.05% in psoriasis has been demonstrated in two randomized, vehicle controlled clinical trials, which were identical in design. The studies were conducted in patients aged 18 years and older with moderate to severe plaque psoriasis. Patients were treated twice daily for up to 4 weeks with either Clobetasol Propionate Topical Spray, 0.05% or vehicle spray.
Patients were evaluated on their Overall Disease Severity, a 5-point scale based on scaling, erythema, and plaque elevation that classified subjects as clear, almost clear, mild, moderate, or severe/very severe. Only patients classified as moderate or severe/very severe at baseline were enrolled in the studies. The median percent body surface area (BSA) at baseline was 6% for the two studies. The numbers of patients scored as clear or almost clear at Weeks 2 and 4 are presented in Table 2.
| Study 1 | Study 2 | ||||
|---|---|---|---|---|---|
| Clobetasol Propionate Topical Spray, 0.05% | Vehicle | Clobetasol Propionate Topical Spray, 0.05% | Vehicle | ||
| N=60 | N=60 | N=60 | N=60 | ||
| Week 2 | Clear | 1 (2%) | 0 (0%) | 0 (0%) | 0 (0%) |
| Almost Clear | 32 (53%) | 1 (2%) | 28 (47%) | 0 (0%) | |
| Week 4 | Clear | 15 (25%) | 0 (0%) | 18 (30%) | 0 (0%) |
| Almost Clear | 32 (53%) | 2 (3%) | 31 (52%) | 1 (2%) | |
16. HOW SUPPLIED/STORAGE AND HANDLING
Clobetasol Propionate Topical Spray, 0.05% is a clear, colorless liquid, supplied in a white HDPE bottle with a white polypropylene cap and white LDPE liner in the following sizes:
2 fl oz/59 mL NDC 51672-1371-4
4.25 fl oz/125 mL NDC 51672-1371-8
17. PATIENT COUNSELING INFORMATION
[See FDA-approved patient labeling (Patient Information)]
17.1 Information for Patients
Patients using topical corticosteroids should receive the following information and instructions:
- This medication is to be used as directed by the physician and should not be used longer than the prescribed time period.
- This medication should not be used for any disorder other than that for which it was prescribed.
- Do not use other corticosteroid-containing products while using Clobetasol Propionate Topical Spray, 0.05% unless directed by your physician.
- The treated skin area should not be bandaged, otherwise covered, or wrapped so as to be occlusive unless directed by the physician.
- Patients should wash their hands after applying the medication.
- Patients should report any signs of local or systemic adverse reactions to the physician.
- Patients should inform their physicians that they are using Clobetasol Propionate Topical Spray, 0.05% if surgery is contemplated.
- If you go to another doctor for illness, injury or surgery, tell that doctor you are using Clobetasol Propionate Topical Spray, 0.05%.
- This medication is for external use only. It should not be used on the face, underarms, or groin area. Also avoid contact with the eyes and lips.
- As with other corticosteroids, therapy should be discontinued when control is achieved. If no improvement is seen within 2 weeks, contact the physician.
- Patients should not use more than 50 g (59 mL or 2 fl.oz.) per week of Clobetasol Propionate Topical Spray, 0.05%.
- Do not use more than 26 sprays per application or 52 sprays per day.
- This medication is flammable; avoid heat, flame or smoking when applying this product.
Mfd. by: Taro Pharmaceuticals Inc., Brampton, Ontario, Canada L6T 1C1
Dist. by: Taro Pharmaceuticals U.S.A., Inc., Hawthorne, NY 10532
Issued: November 2015
PPK-8022-0
68
PATIENT INFORMATION
Clobetasol Propionate (kloe bay' ta sol proe' pee oh nate) Topical Spray, 0.05%
| Important: For use on skin only. Do not get Clobetasol Propionate Topical Spray near or in your eyes, mouth or vagina. |
Read the Patient Information that comes with Clobetasol Propionate Topical Spray before you start using it and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or your treatment.
What is Clobetasol Propionate Topical Spray, 0.05%?
Clobetasol Propionate Topical Spray is a prescription corticosteroid medicine used to treat adults with moderate to severe plaque psoriasis that affects up to 20% of the body's skin surface. Clobetasol Propionate Topical Spray is for use on the skin only (topical).
- Clobetasol Propionate Topical Spray should only be used for the shortest amount of time needed to treat your plaque psoriasis.
- Do not use more than 26 sprays for each application or more than 52 sprays in 1 day.
- You should not apply more than 59 mL (2 fluid ounces) of Clobetasol Propionate Topical Spray to your skin in 1 week.
You should not use Clobetasol Propionate Topical Spray, 0.05%:
- on your face, underarms (armpits), or groin areas
- if you have thinning of the skin (atrophy) at the treatment site
- to treat rosacea or a rash around the mouth (perioral dermatitis)
It is not known if Clobetasol Propionate Topical Spray is safe and effective in children under 18 years of age. Clobetasol Propionate Topical Spray should not be used in children under 18 years of age.
What should I tell my doctor before using Clobetasol Propionate Topical Spray, 0.05%?
Before you use Clobetasol Propionate Topical Spray, tell your doctor if you:
- have a skin infection. You may need medicine to treat the skin infection before you use Clobetasol Propionate Topical Spray.
- plan to have surgery
- have any other medical conditions
- are pregnant or plan to become pregnant. It is not known if Clobetasol Propionate Topical Spray will harm your unborn baby.
- are breast-feeding or plan to breast-feed. It is not known if Clobetasol Propionate Topical Spray passes into your breast milk. Talk to your doctor about the best way to feed your baby if you use Clobetasol Propionate Topical Spray.
Tell your doctor about all the medicines you take, including prescription and non-prescription medicines, vitamins and herbal supplements. Especially tell your doctor if you take other corticosteroid medicines by mouth or use other products on your skin that contain corticosteroids. Ask your doctor or pharmacist if you are not sure.
Know the medicines you take. Keep a list of them to show your doctor and pharmacist when you get a new medicine.
How should I use Clobetasol Propionate Topical Spray, 0.05%?
- Use Clobetasol Propionate Topical Spray exactly as your doctor tells you to use it.
- Your doctor should tell you how much Clobetasol Propionate Topical Spray to use and where to apply it.
- Clobetasol Propionate Topical Spray is for use on skin only. Do not get Clobetasol Propionate Topical Spray near or in your eyes, mouth, or vagina.
- You should not use Clobetasol Propionate Topical Spray on your face, underarms (armpits), or groin areas.
- Apply Clobetasol Propionate Topical Spray 2 times each day.
- Apply only enough Clobetasol Propionate Topical Spray to cover the affected skin areas. Rub in gently and completely.
- Wash your hands after using Clobetasol Propionate Topical Spray.
- Throw away any unused Clobetasol Propionate Topical Spray.
- Do not bandage or cover your treated areas unless your doctor tells you to.
- Tell your doctor if your skin condition is not getting better after 2 weeks of using Clobetasol Propionate Topical Spray. Your doctor may tell you to apply Clobetasol Propionate Topical Spray to certain areas of your skin for up to 2 more weeks if needed. You should not use Clobetasol Propionate Topical Spray for more than 4 weeks unless your doctor tells you to. This can increase your risk of serious side effects.
See the "Instructions for Use" at the end of the Patient Information for detailed information about the right way to apply Clobetasol Propionate Topical Spray.
What should I avoid while using Clobetasol Propionate Topical Spray, 0.05%?
- Clobetasol Propionate Topical Spray is flammable. Avoid heat, flames or smoking while applying Clobetasol Propionate Topical Spray to your skin.
What are the possible side effects of Clobetasol Propionate Topical Spray, 0.05%?
- Clobetasol Propionate Topical Spray can pass through your skin. Too much Clobetasol Propionate Topical Spray passing through your skin can cause your adrenal glands to stop working. Your doctor may do blood tests to check how well your adrenal glands are working.
The most common side effects of Clobetasol Propionate Topical Spray include:
- burning at treated site
- upper respiratory tract infection
- runny nose
- sore throat
- dry, itchy, and reddened skin
If you go to another doctor for illness, injury or surgery, tell that doctor you are using Clobetasol Propionate Topical Spray.
Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of Clobetasol Propionate Topical Spray. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
You may also report side effects to Taro Pharmaceuticals U.S.A., Inc. at 1-866-923-4914.
How should I store Clobetasol Propionate Topical Spray, 0.05%?
- Store Clobetasol Propionate Topical Spray at room temperature between 68˚F to 77˚F (20˚C to 25˚C).
- Do not store Clobetasol Propionate Topical Spray above 86°F (30°C).
- Do not freeze or refrigerate Clobetasol Propionate Topical Spray.
- Keep the bottle of Clobetasol Propionate Topical Spray tightly closed.
- Keep away from heat or flame
Keep Clobetasol Propionate Topical Spray and all medicines out of the reach of children.
General information about Clobetasol Propionate Topical Spray, 0.05%.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Clobetasol Propionate Topical Spray for a condition for which it was not prescribed. Do not give Clobetasol Propionate Topical Spray to other people, even if they have the same symptoms that you have. It may harm them.
This Patient Information leaflet summarizes the most important information about Clobetasol Propionate Topical Spray. If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for information about Clobetasol Propionate Topical Spray that is written for health professionals.
What are the ingredients in Clobetasol Propionate Topical Spray, 0.05%?
Active ingredient: clobetasol propionate
Inactive ingredients: alcohol, isopropyl myristate, sodium lauryl sulfate, and undecylenic acid
Instructions for Use
Clobetasol Propionate (kloe bay' ta sol proe' pee oh nate) Topical Spray, 0.05%
| Important: For use on skin only. Do not get Clobetasol Propionate Topical Spray near or in your mouth, eyes or vagina. |
Read the Instructions for Use that comes with Clobetasol Propionate Topical Spray before you start using it and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or your treatment.
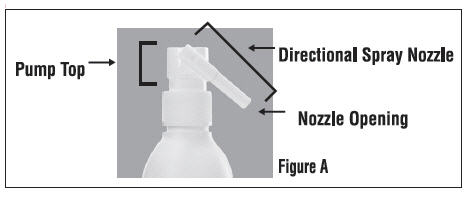
Parts of Clobetasol Propionate Topical Spray, 0.05% (See Figure A)
When you receive Clobetasol Propionate Topical Spray the Directional Spray Nozzle is in the "locked" position with the nozzle pointing downwards. (See Figure B)
 |  |
| How to apply Clobetasol Propionate Topical Spray, 0.05%: | ||||
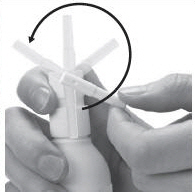 Figure C | Step 1: To unlock the Directional Spray Nozzle, hold the Clobetasol Propionate Topical Spray bottle and sides of the Pump Top with one hand. Use your other hand to turn the Directional Spray Nozzle to either the right or the left (See Figure C). The spray will come out through the opening at the end of the Directional Spray Nozzle. | 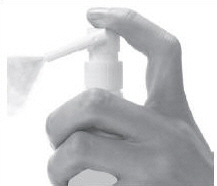 Figure D | Step 2: To apply Clobetasol Propionate Topical Spray, point the Directional Spray Nozzle to the affected area. To spray, push down on the Pump Top. Apply Clobetasol Propionate Topical Spray to the affected area as instructed by your doctor. (See Figure D) | |
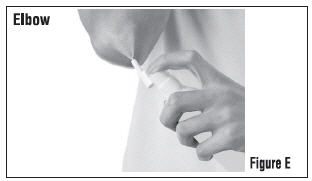
Step 3: Spray only enough Clobetasol Propionate Topical Spray to cover the affected area, for example, the elbow (See Figure E). Rub in Clobetasol Propionate Topical Spray gently and completely.
- You should not apply Clobetasol Propionate Topical Spray to your face, underarms or groin. Avoid contact with your eyes and lips.
- To apply to your scalp, part your hair at the area where Clobetasol Propionate Topical Spray is to be applied. Spray directly on the affected area, and then rub in gently and completely. (See Figure F)
- Repeat Steps 2 and 3 to apply Clobetasol Propionate Topical spray to other affected areas as instructed by your doctor.
 Figure G | Step 4: After applying Clobetasol Propionate Topical Spray, return the Directional Spray Nozzle to the "locked" position. (See Figure G) | Step 5: Wash your hands after applying Clobetasol Propionate Topical Spray. | ||
This Patient Information and Instructions for Use have been approved by the U.S. Food and Drug Administration.
Mfd. by: Taro Pharmaceuticals Inc., Brampton, Ontario, Canada L6T 1C1
Dist. by: Taro Pharmaceuticals U.S.A., Inc., Hawthorne, NY 10532
Issued: November 2015
PPK-8022-0
68