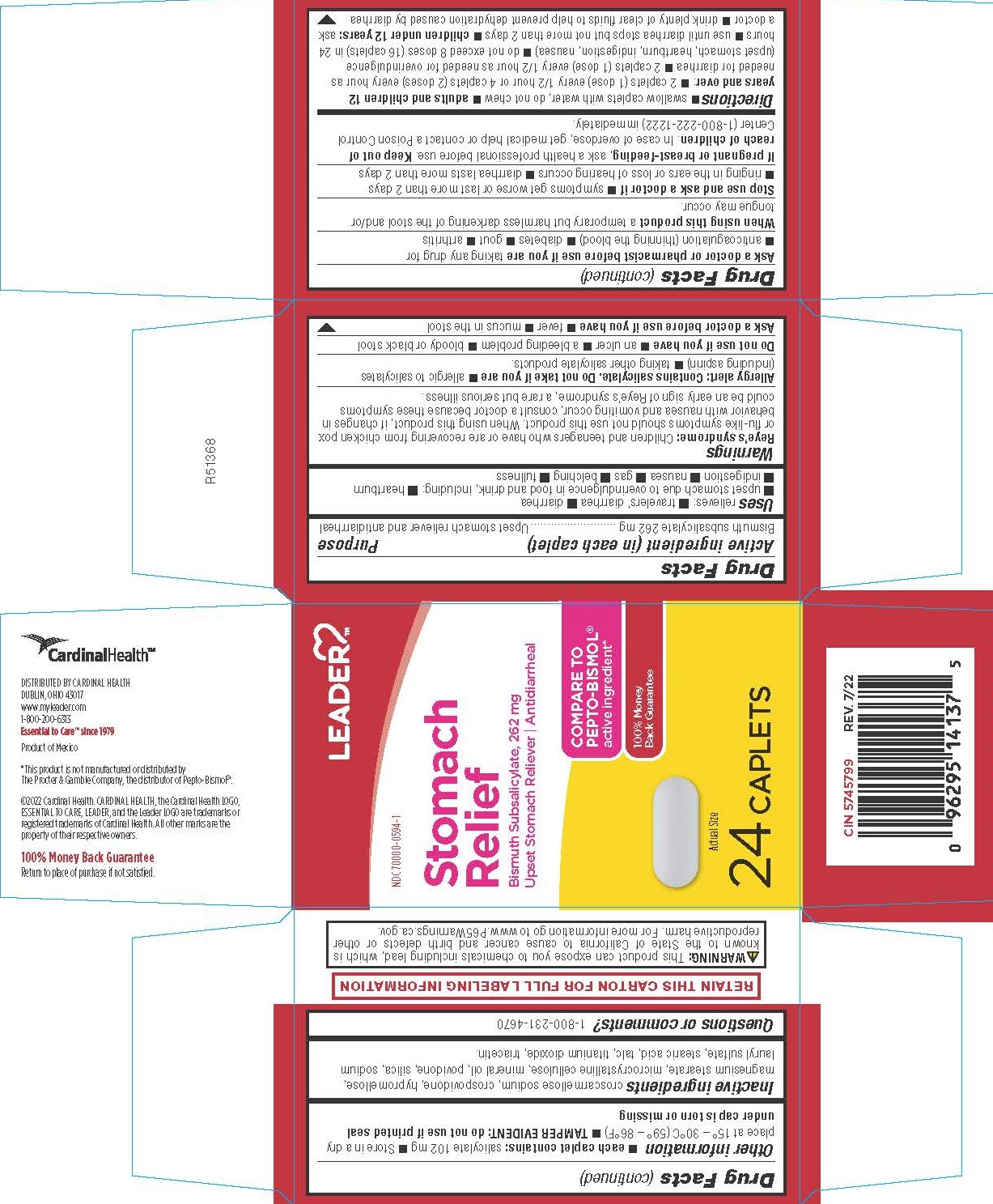
Uses
relieves: ■ travelers’ diarrhea ■ diarrhea
■ upset stomach due to overindulgence in food and drink, including: ■ heartburn ■ indigestion ■ nausea ■ gas ■ belching ■ fullness
Reye’s syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye’s syndrome, a rare but serious illness.
Allergy alert: Contains salicylate. Do not take if you are
- allergic to salicylates (including aspirin)
- taking other salicylate products.
Ask a doctor or pharmacist before use if you are taking any drug for
- anticoagulation (thinning the blood)
- diabetes
- gout
- arthritis
Directions
- swallow caplets with water, do not chew
adults and children 12 years and over:
- 2 caplets (1 dose) every 1/2 hour or 4 caplets (2 doses) every hour as needed for diarrhea
- 2 caplets (1 dose) every 1/2 hour as needed for overindulgence (upset stomach, heartburn, indigestion, nausea)
- do not exceed 8 doses (16 caplets) in 24 hours
- use until diarrhea stops but not more than 2 days
children under 12 years: ask a doctor
- drink plenty of clear fluids to help prevent dehydration caused by diarrhea
Other information
- each caplet contains: salicylate 102 mg
- Store in a dry place at 15° – 30°C (59° – 86°F)
- TAMPER EVIDENT: do not use if printed seal under cap is torn or missing
croscarmellose sodium, crospovidone, hypromellose, magnesium stearate, microcrystalline cellulose, mineral oil, povidone, silica, sodium lauryl sulfate, stearic acid, talc, titanium dioxide, triacetin.
RETAIN THIS CARTON FOR FULL LABELING INFORMATION
WARNING: This product can expose you to chemicals including lead, which is known to the State of California to cause cancer and birth defects or other reproductive harm. For more information go to www.P65Warnings.ca.gov.
COMPARE TO PEPTO-BISMOL active ingredient*
*This product is not manufactured or distributed by Procter & Gamble Inc., the distributor of Pepto-Bismol®.
DISTRIBUTED BY CARDINAL HEALTH
DUBLIN, OHIO 43017
www.myleader.com
1-800-200-6313
Essential to Care™ since 1979
Product of Mexico