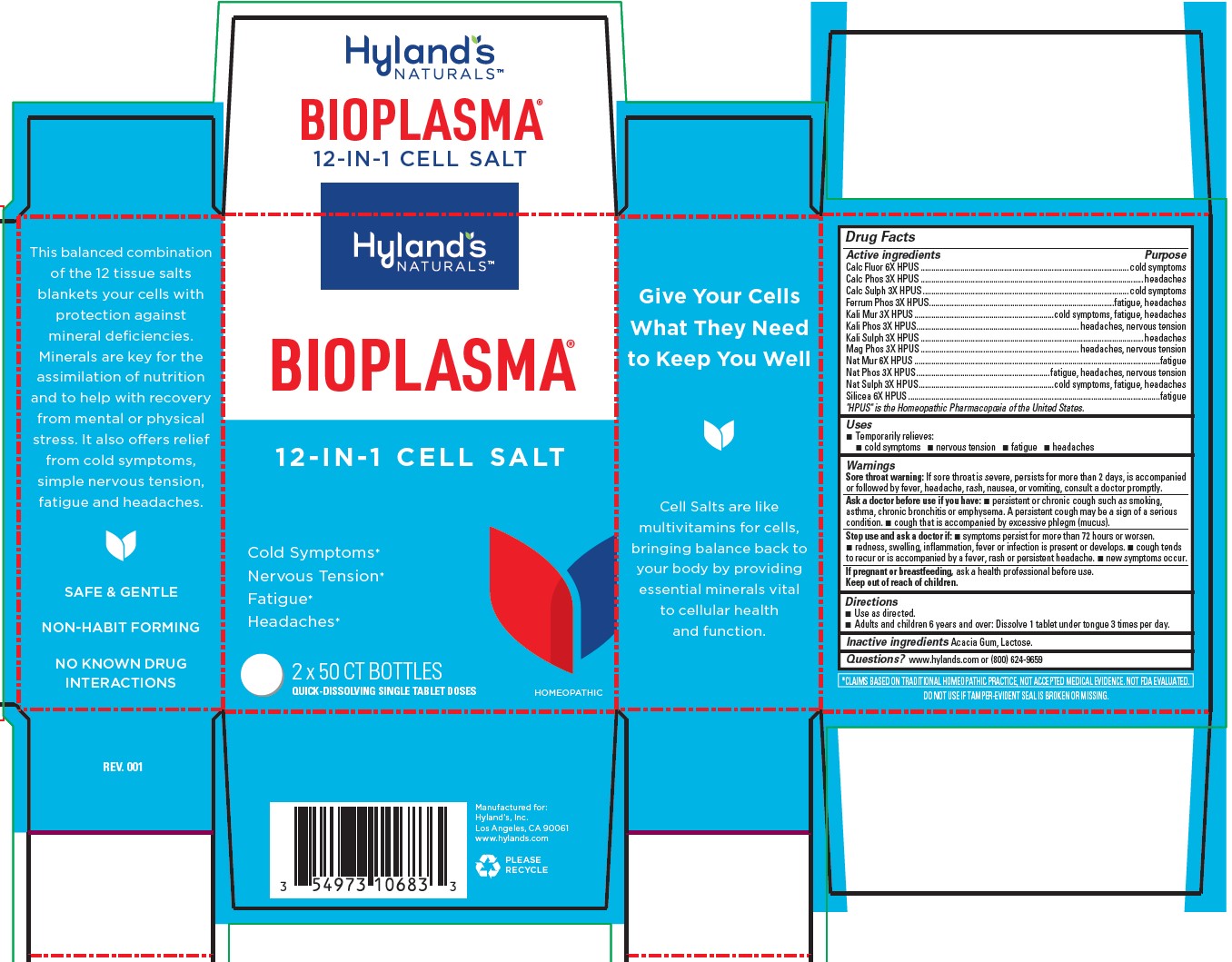
BIOPLASMA- sodium phosphate, dibasic, heptahydrate, calcium sulfate anhydrous, ferrosoferric phosphate, potassium phosphate, dibasic, potassium sulfate, magnesium phosphate, dibasic trihydrate, sodium chloride, sodium sulfate, silicon dioxide, potassium chloride, calcium fluoride, tribasic calcium phosphate tablet
Hyland's Inc.
Disclaimer: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
----------
Temporarily relieves: cold symptoms, nervous tension, fatigue, headaches
Drug Facts
Active ingredients
| Active ingredients | Purpose |
| Calc Fluor 6X HPUS | cold symptoms |
| Calc Phos 3X HPUS | headaches |
| Calc Sulph 3X HPUS | cold symptoms |
| Ferrum Phos 3X HPUS | fatigue, headaches |
| Kali Mur 3X HPUS | cold symptoms, fatigue, headaches |
| Kali Phos 3X HPUS | headaches, nervous tension |
| Kali Sulph 3X HPUS | headaches |
| Mag Phos 3X HPUS | headaches, nervous tension |
| Nat Mur 6X HPUS | fatigue |
| Nat Phos 3X HPUS | fatigue, headaches, nervous tension |
| Nat Sulph 3X HPUS | cold symptoms, fatigue, headaches |
| Silicea 6X HPUS | fatigue |
"HPUS" is the Homeopathic Pharmacopoeia of the United States.
Uses
Temporarily relieves: ■ cold symptoms ■ nervous tension ■ fatigue ■ headaches
Warnings
Sore throat warning
If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
Ask a doctor before use if you have
■ persistent or chronic cough such as smoking, asthma, chronic bronchitis or emphysema. A persistent cough may be a sign of a serious condition. ■ cough that is accompanied by excessive phlegm (mucus).
Stop use and ask a doctor if
■ symptoms persist for more than 72 hours or worsen. ■ redness, swelling, inflammation, fever or infection is present or develops. ■ cough tends to recur or is accompanied by a fever, rash or persistent headache. ■ new symptoms occur.
If pregnant or breastfeeding
ask a health professional before use.
Keep out of reach of children.
Directions
■ Use as directed.
■ Adults and children 6 years and over: Dissolve 1 tablet under tongue 3 times per day.
Inactive ingredients
Acacia Gum, Lactose.
Questions?
www.hylands.com or (800) 624-9659
*CLAIMS BASED ON TRADITIONAL HOMEOPATHIC PRACTICE, NOT ACCEPTED MEDICAL EVIDENCE. NOT FDA EVALUATED.
DO NOT USE IF TAMPER-EVIDENT SEAL IS BROKEN OR MISSING.
PACKAGE LABEL - 2 x 50 TABLETS BOTTLE CARTON
Hyland's
NATURALS™
BIOPLASMA
®
12 - IN - 1 CELL SALT
Cold Symptoms*
Nervous Tension*
Fatigue*
Headaches*
2 x 50 CT BOTTLES
QUICK-DISSOLVING SINGLE TABLET DOSES
HOMEOPATHIC
Hyland's Inc.
