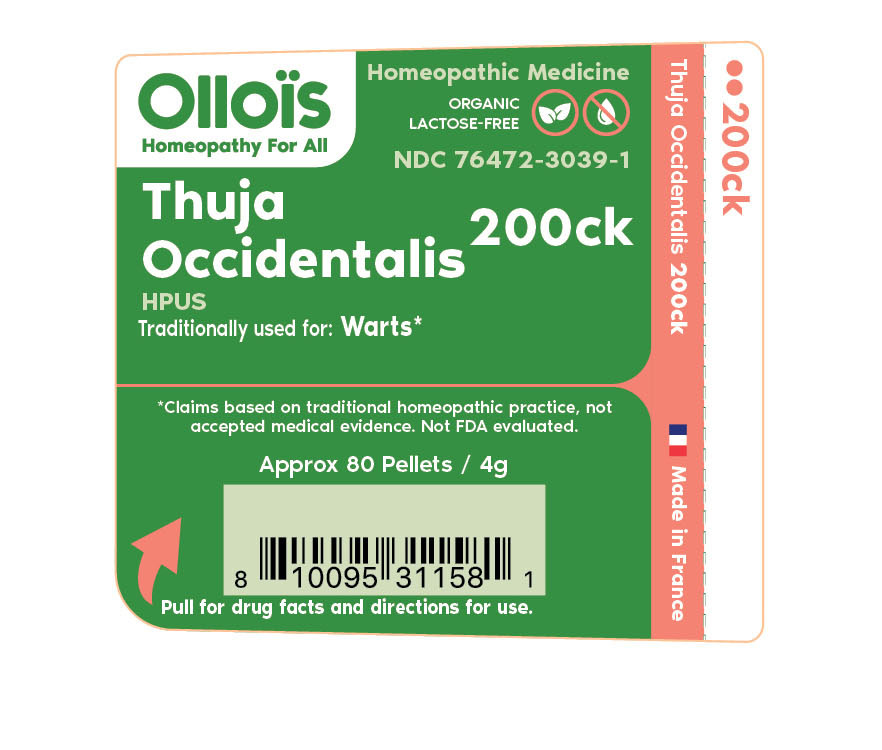
THUJA OCCIDENTALIS
200CK- thuja occidentalis pellet
SEVENE USA
Disclaimer: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
----------
ACTIVE INGREDIENT
HOMEOPATHIC DILUTION OF HPUS THUJA OCCIDENTALIS 200ck **C,K, CK, AND X ARE HOMEOPATHIC DILUTIONS.
TRADITIONALLY USED FOR
Stinging edemas from stings or allergies, relieved by cold*
DIRECTIONS
(adults/children) Dissolve 5 pellets under the tongue 3 times a day until symptoms are relieved or as directed by a physician.
USE
Condition listed above or as directed by a physician.
WARNINGS
Stop use and ask a physician if symptoms persist for more than 3 days or worsen.
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children.
OTHER INFORMATION
Store at room temperature.
Do not use if pellet-dispenser seal is broken.
INACTIVE INGREDIENT
Sucrose.
QUESTIONS?
INFO@OLLOIS.COM * www.ollois.com * MADE IN FRANCE. NOT REVIEWED BY THE FDA AND NOT GUARANTEED TO BE EFFECTIVE. THIS HOMEOPATHIC DILUTION MAY NOT BE SUSCEPTIBLE TO SCIENTIFIC MEASUREMENT.