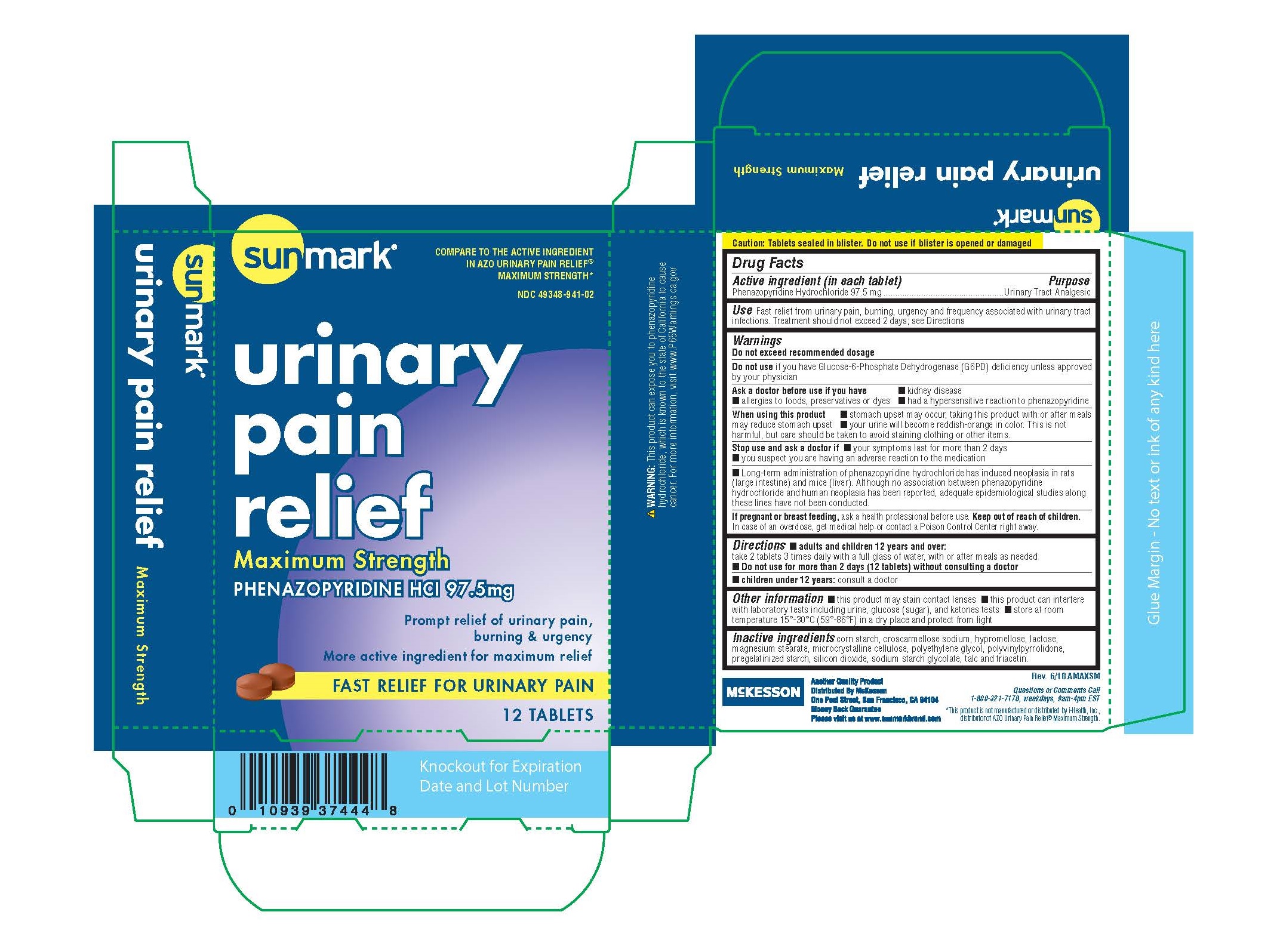
ASK DOCTOR
ASK DOCTOR BEFORE USE
IF YOU HAVE KIDNEY DISEASE
ALLERGIES TO FOODS,PRESERVATIVES OR DYES
HAD A HYPERSENSITIVE REACTION TO PHENAZOPYRIDINE
WHEN USING
WHEN USING THIS PRODUCT
STOMACH UPSET MAY OCCUR,TAKING THIS PRODUCT WITH OR AFTER MEALS MAY REDUCE STOMACH UPSET
YOUR URINE WILL BECOME REDDISH ORANGE IN COLOR.THIS IS NOT HARMFUL,BUT CARE SHOULD BE TAKEN TO
AVOID STAINING CLOTHING OR OTHER ITEMS.
STOP USE
STOP USE AND ASK A DOCTOR
IF YOUR SYMPTOMS LAST FOR MORE THAN 2 DAYS
YOU SUSPECT YOU ARE HAVING AN ADVERSE REACTION TO THE MEDICATION
KEEP OUT OF REACH OF CHILDREN
IN CASE OF OVERDOSE ,GET MEDICAL HELP OR CONTACT A POISON CONTROL CENTER RIGHT AWAY.
INDICATIONS & USAGE
Use; fast relief from urinary pain,burning,urgency and frequency associated with urinary tract infections.
INACTIVE INGREDIENT
Lactose, magnesium silicate, magnesium stearate, microcrystalline cellulose,
pharmaceutical glaze, and sodium starch glycolate.