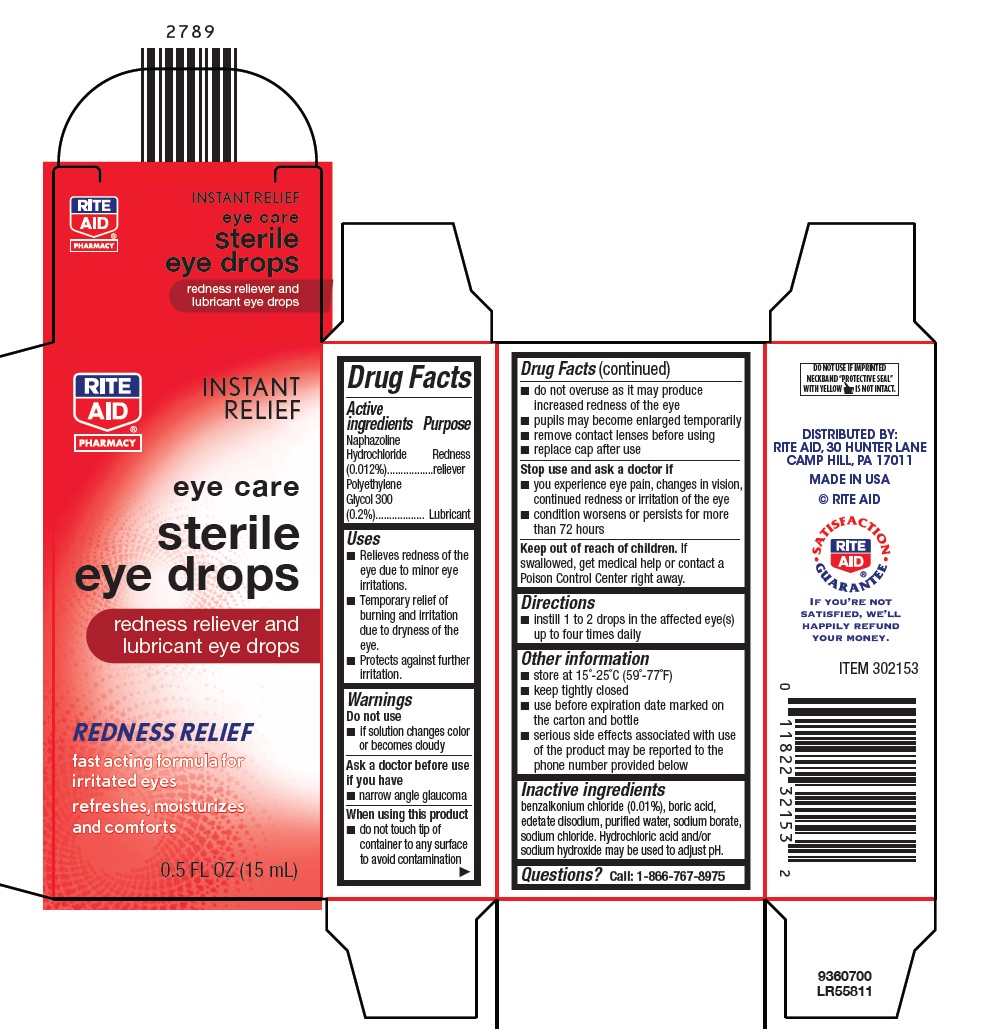
ORIGINAL REDNESS RELIEVER- naphazoline hydrochloride and polyethylene glycol 300 solution/ drops
Rite Aid Corporation
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Active ingredients
Naphazoline Hydrochloride (0.012%)
Polyethylene Glycol 300 (0.2%)
Purpose
Redness reliever
Lubricant
Uses
- •
- Relieves redness of the eye due to minor eye irritations.
- •
- Temporary relief of burning and irritation due to dryness of the eye.
- •
- Protects against further irritation.
Warnings
Do not use
- •
- If solution changes color or becomes cloudy
Ask a doctor before use if you have
- •
- narrow angle glaucoma
When using this product
- •
- do not touch tip of container to any surface to avoid contamination
- •
- do not overuse as it may produce increased redness of the eye
- •
- pupils may become enlarged temporarily
- •
- remove contact lenses before using
- •
- replace cap after use
Stop use and ask a doctor if
- •
- you experience eye pain, changes in vision, continued redness or irritation of the eye
- •
- condition worsens or persists for more than 72 hours
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Directions
- •
- instill 1 to 2 drops in the affected eye(s) up to four times daily
Other information
- •
- store at 15°-25°C (59°-77°F)
- •
- keep tightly closed
- •
- use before expiration date marked on the carton and bottle
- •
- serious side effects associated with use of the product may be reported to the phone number provided below
Inactive ingredients
benzalkonium chloride (0.01%), boric acid, edetate disodium, purified water, sodium borate, sodium chloride. Hydrochloric acid and/or sodium hydroxide may be used to adjust pH.
Questions?
Call: 1-866-767-8975
DISTRIBUTED BY:
RITE AID, 30 HUNTER LANE
CAMP HILL, PA 17011
MADE IN USA
© RITE AID
ITEM 302153
9360700
LR55811
Package/Label Principal Display Panel
RITE
AID®
PHARMACY
INSTANT RELIEF
eye care
sterile
eye drops
redness reliever and
lubricant eye drops
REDNESS RELIEF
fast acting formula for
irritated eyes
refreshes, moisturizes
and comforts
0.5 FL OZ (15 mL)